† Corresponding author. E-mail:
Single-crystal GaN layers have been obtained by nitriding β-Ga2O3 films in NH3 atmosphere. The effect of the temperature and time on the nitridation and conversion of Ga2O3 films have been investigated. The nitridation process results in lots of holes in the surface of films. The higher nitridation temperature and longer time can promote the nitridation and improve the crystal quality of GaN films. The converted GaN porous films show the single-crystal structures and low-stress, which can be used as templates for the epitaxial growth of high-quality GaN.
GaN-based nitrides have been widely used in optical devices and high power or high temperature microelectronic devices.[1–3] However, one of the main problems for high quality GaN growth is lack of a suitable lattice-match substrate. Foreign substrates, such as sapphire and Si, bring about a large number of defects in GaN epilayers, although they are the most popular substrates.[4,5] The ideal substrate would be GaN for the homoepitaxy.
β-Ga2O3 is an ultra-wide band gap semiconductor and has excellent properties, such as high transparency in the region from visible to UV regions, high electrical conductivity and thermal stability at high temperature.[6–8] The lattice mismatch between GaN and β-Ga2O3 is relatively small, as low as 2.6%.[9] So, β-Ga2O3 can also be applied as a substrate for GaN epitaxial growth.[10,11] It has been reported that the (100) cleavage plane of β-Ga2O3 undergoes a surface reconstruction under nitridation at high temperature and NH3 gas, which causes the surface to match GaN naturally.[12] The surface of β-Ga2O3 film can be nitridated to acquire good lattice-matching of GaN for the direct growth of GaN. And, GaN films converted from β-Ga2O3 by nitridation with NH3 can be also used as a substrate or template for GaN homogeneous epitaxial growth. In this work, we report the influences of temperature and time on the nitridation to better understand the process.
The 2-inch (1 inch = 2.54 cm) β-Ga2O3 films have been grown by self-made halide vapor phase epitaxy (HVPE) system.[13] The as-grown β-Ga2O3 is transparent and smooth, the surface roughness is about 6.2 nm obtained by the analysis of atomic force microscope. The as-grown β-Ga2O3 films have been cut into 1 cm 
Figure
 | Fig. 1. The SEM images of β-Ga2O3 film and the samples after nitridation at different temperatures for 0.5 h. The insets are SEM images in the higher magnification. |
The XRD patterns (
 | Fig. 2. XRD patterns of β-Ga2O3 film and the samples after nitridation at different temperatures for 0.5 h. |
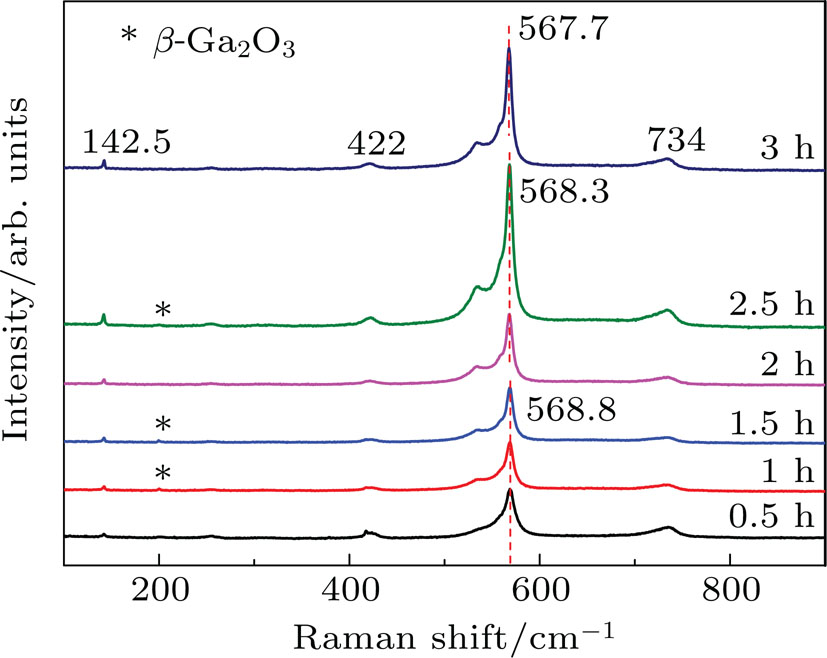
Raman spectra has been measured at room temperature by using an argon ion laser (**
Figure
 | Fig. 4. The SEM images of β-Ga2O3 films after nitridation at 1050 °C for different times. The insets are SEM images in the higher magnification. |
Figure
 | Fig. 5. XRD patterns of β-Ga2O3 films after nitridation at 1050 °C for different times (a). The XRC patterns of GaN (002) and (102) formed at 1050 °C for different times (b). |
In order to characterize the crystal quality, the x-ray rocking curves (XRC) of the GaN (002) and (102) planes were obtained from as-nitridated β-Ga2O3 films at 1050 °C for different times. As shown in Fig.
Figure
In summary, GaN films have been obtained from the β-Ga2O3 films by the nitridation under the NH3 atmosphere. The nitridated films show a porous network surface morphology. The structural analysis indicates that the nitridated surface layer is single-crystal GaN layer. After full nitridation at 1050 °C for 3 h, GaN layers show no significant stress. The nitridation temperature and time can promote and improve the crystal quality of GaN films. The porous GaN films obtained by the nitridation of β-Ga2O3 can be used as templates for the epitaxial growth of GaN instead of the expensive bulk GaN substrates.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] |