† Corresponding author. E-mail:
We report the growth of AlN epilayers on c-plane sapphire substrates by pulsed metal organic chemical vapor deposition (MOCVD). The sources of trimethylaluminium (TMAl) and ammonia were pulse introduced into the reactor to avoid the occurrence of the parasitic reaction. Through adjusting the duty cycle ratio of TMAl to ammonia from 0.8 to 3.0, the growth rate of AlN epilayers could be controlled in the range of 0.24 m/h to 0.93 m/h. The high-resolution x-ray diffraction (HRXRD) measurement showed that the full width at half maximum (FWHM) of the (0002) and (10-12) reflections for a sample would be 194 arcsec and 421 arcsec, respectively. The step-flow growth mode was observed in the sample with the atomic level flat surface steps, in which a root-mean-square (RMS) roughness was lower to 0.2 nm as tested by atomic force microscope (AFM). The growth process of AlN epilayers was discussed in terms of crystalline quality, surface morphology, and residual stress.
AlN and AlGaN based materials have potential applications in power electronic and optoelectronic fields due to their wide energy bandgaps, high electron mobilities, high breakdown voltages, and strong resistance to radiation.[1] For optoelectronic devices, an AlN layer can be usually used as a template layer or window layer for light-emitting and photo-detective devices in the ultra-visible (UV) spectral region from 200 nm to 365 nm. These UV devices can be used in areas such as disinfection/sterilization, homeland security, and secure space-to-space communication.[2,3] For application in power electronic devices such as the field effect transistor (FET) and high electronic mobility transistor (HEMT), an AlN layer can be used both as a spacer layer, which significantly influences the electron mobility in the two-dimensional electron gases, and a buffer layer when the devices are grown on semi-insulating SiC or Si substrates.[4] However, growing high quality AlN films on foreign substrates using a routine metal organic chemical vapor deposition (MOCVD) method at low temperature (
In this study, we grow good quality AlN epilayers on sapphire by a pulsed MOCVD method at a relative low temperature (1150 °C–1190 °C) and find three kinds of growth modes (step-flowing mode, step-bunching mode, and island growth mode) depending on the process parameters. The crystalline quality, surface morphology, and residual stress are characterized and the growth kinetics is discussed. The realization of AlN epilayers with good crystalline quality would be beneficial to the development of the UV optoelectronic and power electronic devices.
AlN samples were grown on 2-inch c-plane sapphire substrates in a low-pressure AIX 200/4 RS-F MOCVD reactor. Trimethylaluminium (TMAl) and ammonia (NH3) were used as Al and N sources, respectively. Prior to growth, the sapphire surface was thermally cleaned in hydrogen ambient for 10 min, followed by AlN growth in a pressure of 50 mbar. To avoid the gas parasitic reactions of TMAl and NH3, TMAl and NH3 were pulse introduced into the reactor by a pulsed MOCVD method. Here we used the duty cycle ratio of TMAl to NHβ, to define the growth atmosphere of AlN for the chamber pressure and the carrier gas flow was constantly maintained for all the samples. The value of β changed from 0.8 to 3.0 so that the growth rate of the AlN epilayers could be controlled, as schematically shown in Fig. 
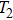
| Table 1.
Growth parameters of AlN samples. . |
The crystalline quality of AlN epilayers was examined by a high resolution x-ray diffractometer (HRXRD) installed at a diffuse x-ray scattering station of the Beijing Synchrotron Radiation Facility, and a Huber five-circle diffractometer was used. The radiation energy of the x-ray beam was 8.05 keV with 0.7 mm×0.4 mm (
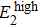
Under high temperature conditions and free of the parasitic reaction, AlN growth rate G is mass transport limited, which means that the growth rate should be proportional to the TMAl flow rate. However, due to the strong parasitic reaction of TMAl and ammonia, the growth rate is very sensitive to the growth atmosphere like the V/III ratio. The possible reaction pathways are described in Fig.
 | Fig. 2. Schematic of the AlN deposition and reaction pathways.[13] |
The reflectance versus time curves of AlN growth in-situ optically monitored by Filmetrics F30 with selective wavelength at 650 nm are shown in Fig.
 |
| Table 2.
Growth rate G and the other correlative parameters. . |
The diffusion coefficient D and the rate of adsorption and desorption of Al atoms follow the Arrhenius equation
 |
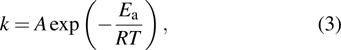 |
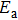
To determine the extent of the reaction of TMAl, the relative efficiency value γ is introduced as
 |
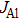
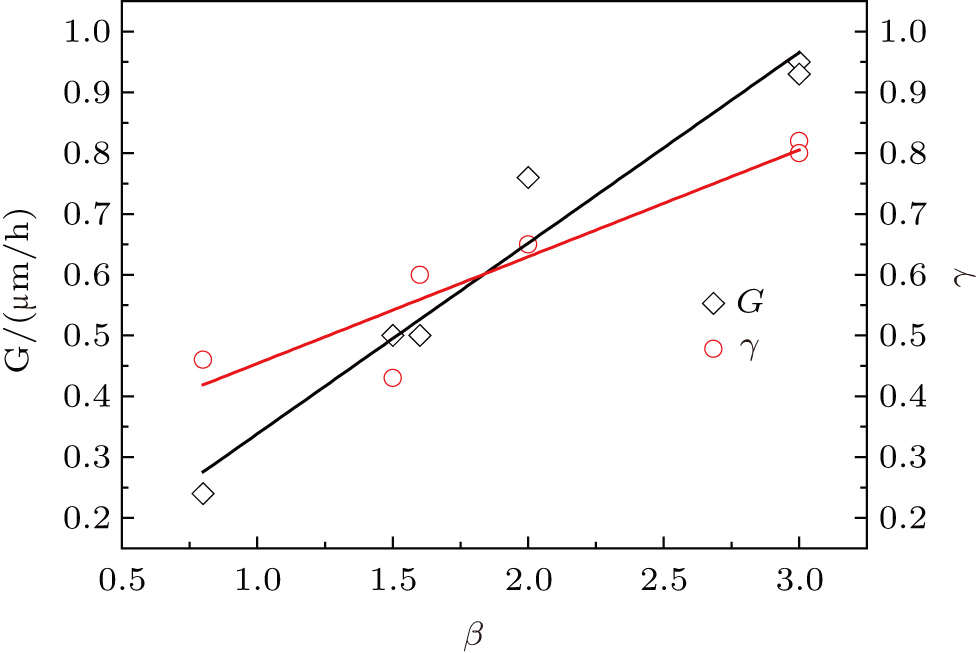
To confirm the validity and examine the quantitative relationships of β with 
Three key factors, the actual TMAl flow, growth temperature T, and growth atmosphere, that may influence the growth rate of AlN are considered. It can be easily concluded that the value of 
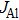

A linear fitting of β with growth rate G and relative efficiency value γ is performed and the results are shown in Fig.
To explain the nature of the approximate proportional relationship of β with growth rate G and the relative efficiency value γ, we speculate that it can be attributed to the change of the growth atmosphere, i.e., the ammonia flow is switched to hydrogen flow corresponding to the transition from the N-rich condition to the H-rich condition. Gherasoiu et al.[15] pointed out that the AlN growth rate is limited by the degree of hydrogen passivation and the nitrogen bonds available at the growth surface. Under the N-rich condition (when the ammonia flow is switched on), the hydrogen surface coverage increases, which means that Al has to compete with residual hydrogen for N bonds, thus resulting in a reduced growth rate. To conclude, it is a feasible method to modify the growth rate of AlN growth by adjusting the duty cycle ratio of TMAl to ammonia when the TMAl flow has reached the saturation point.
The nucleation layer in sample A was directly grown on the sapphire substrate at 1150 °C, while the other samples were grown at 800 °C. As shown in Fig.
Symmetrical (0002) and asymmetrical (10-12) x-ray rocking curve measurements (XRC) were carried out to evaluate the crystalline quality of the AlN samples as shown in Fig.
 |
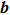
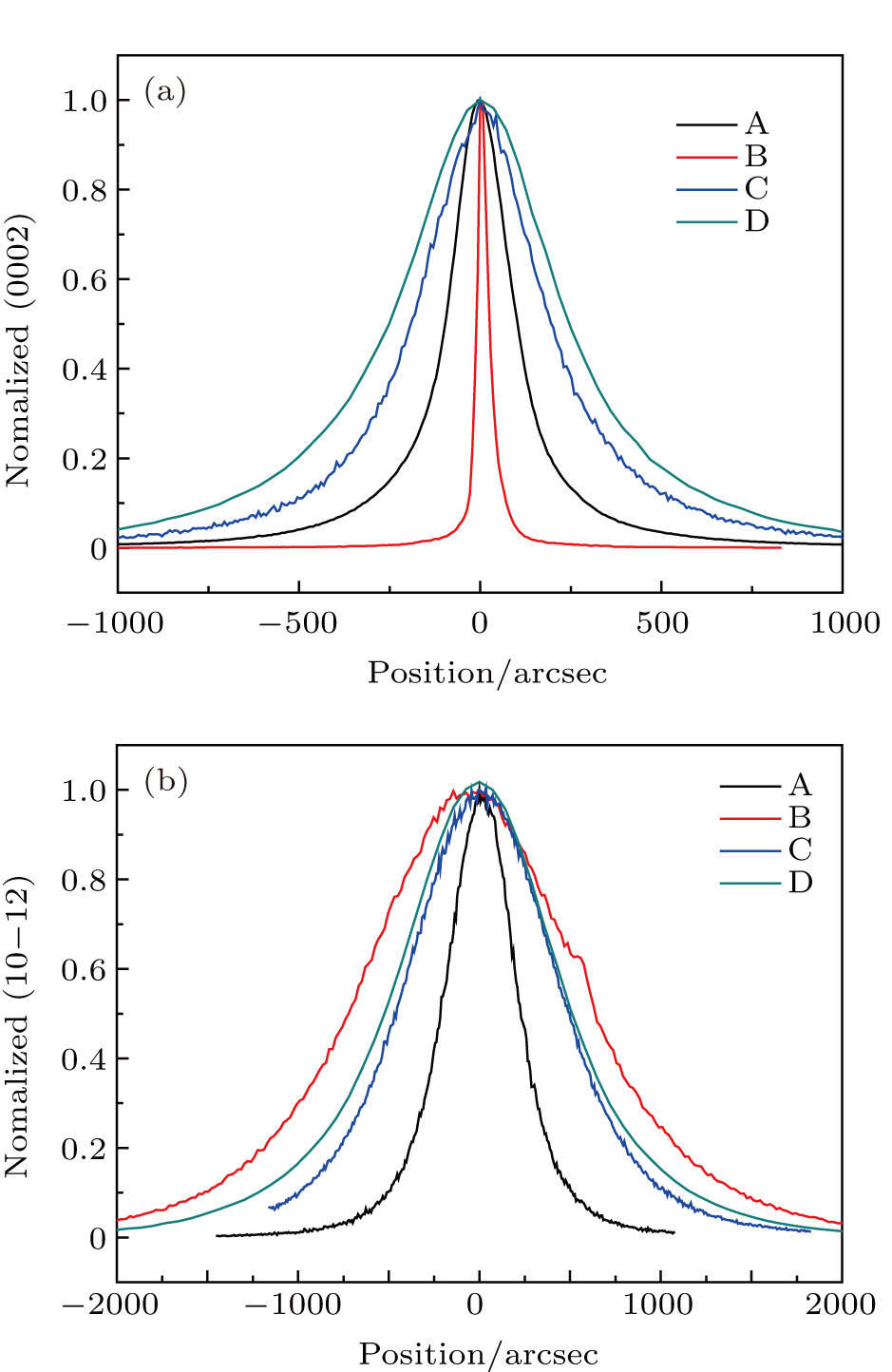 | Fig. 5. (color online) X-ray rocking curves of AlN (0002) and (10-12) reflections measured by HRXRD. |
| Table 3.
XRD, AFM, and Raman measurement results of AlN samples. . |
Raman scattering measurements were performed to evaluate the residual stress in the AlN layer[19] as shown in Fig. 
Figure
 | Fig. 7. (color online) AFM micrographs of AlN layers of (a) sample A, (b) sample B, (c) sample C and (d) sample D. |
In sample C, there is a step-bunched morphology with 5–10 ML-height steps and 
We studied the growth of AlN layers using pulse MOCVD methods and obtained a high quality AlN layer under a relative low temperature. The growth rate of AlN was adjusted by the on-duty time ratio of TMAl flow to ammonia. We found that a low temperature nucleation layer (about 800 °C) can improve the uniformity of the AlN layer while there is a narrow growth window to grow a high quality AlN film with a high temperature nucleation layer (about 1150 °C). Three different growth modes were observed in our samples including step-flow mode, step-bunching mode, and island growth mode.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] |