† Corresponding author. E-mail:
Project supported by the Fund from the Science and Technology Department of Jilin Province, China (Grant No. 20170520108JH), the Beihua University Youth Nurtural Fund, China (Grant No. 2017QNJJL15), the Beihua University PhD Research Start-up Fund, China (Grant No. 202116140), and the Undergraduate Innovation Project, China (Grant No. 220718100).
We describe the synthesis of three-dimensional (3D) multilayer ZnO@Ag/SiO2@Ag nanorod arrays by the physico–chemical method. The surface-enhanced Raman scattering (SERS) performance of the 3D multilayer ZnO@Ag/SiO2@Ag nanorod arrays is studied by varying the thickness of dielectric layer SiO2 and outer-layer noble Ag. The 3D ZnO@Ag/SiO2@Ag nanorod arrays create a huge number of SERS “hot spots” that mainly contribute to the high SERS sensitivity. The great enhancement of SERS results from the electron transfer between ZnO and Ag and different electromagnetic enhancements of Ag nanoparticles (NPs) with different thicknesses. Through the finite-difference time-domain (FDTD) theoretical simulation, the enhancement of SERS signal can be ascribed to a strong electric field enhancement produced in the 3D framework. The simplicity and generality of our method offer great advantages for further understanding the SERS mechanism induced by the surface plasmon resonance (SPR) effect.
Owing to the urgent need of spectrum analysis, surface-enhanced Raman scattering (SERS) has been developed rapidly in chemical and biological fields, which is a powerful spectrum analysis technique to detect single-molecule and identify specific analytes. The sensitivity of an SERS substrate is mainly based on the surface plasmon resonance (SPR) coupling effect.[1,2] The SPR stimulated by metal nanostructures can effectively couple the light radiation energy to highly restricted surface mode, and thus form a highly enhanced local field in the nanoscaled metal surface.[3–5] It is a common research method to select the material structure for obtaining the required surface-enhanced optical properties, which makes surface plasmons possess broad application prospects in nano-optics, super-resolution imaging, photocatalysis, biosensor, environmental monitoring, and other fields.[6–14] So far, a great many of endeavors have been devoted to the design and synthesis of various nanostructures as SERS substrates, including two-dimensional (2D),[15,16] three-dimensional (3D),[17–20] and other various semiconductors nanostructures, such as Ag@SiO2@Ag,[15] Ag–Silica–Au hybrid nanostructure, ZnO@Ag 3D nanostructure,[21–24] etc. Tian et al. designed a shell-isolated nanoparticle (Au/SiO2 and Au/Al2O3 nanoparticles (NPs)) and obtained the higher detection sensitivity and higher enhanced Raman signal.[11] Tang et al. designed a highly sensitive and uniform 3D cone-shaped ZnO@Ag nanorods as SERS substrate and showed a superior SERS performance.[12] Zhu et al. provided an Au NP/graphene/Au NP sandwiched structure and investigated sub-nanometer gaps and demonstrated high Raman enhancement factors.[13]
Moreover, 3D SERS substrate has attracted much attention in recent years since 3D SERS substrates can provide more adsorptions of target molecules and “hot spots” in all three dimensions. Therefore, there is still room for further development of 3D multilayer SERS structure. It has been found that various 3D nanostructures, such as silicon-based nanowires,[25] silicon-based nanocones,[26] and ZnO nanorods,[27] effectively improve the electromagnetic enhancement. For further optimizing SERS performance, we choose the 3D ZnO@Ag/SiO2@Ag nanorod structure to build a surface plasmon coupling system because multilayer structure can significantly improve its optical coupling performance. On the one hand, in plenty of materials, the ZnO nanorod arrays’ structure can provide a huge specific surface area for the deposition of the inner-layer Ag nanoparticles and the target molecules. Moreover, as a semiconductor material, ZnO supports the chemical enhancement effect, which can be used to make SERS detection concentration limit reduced. On the other hand, the dielectric layer is another important factor for enhancing the SERS signal. Lots of researches about dielectric layer control for SERS have been reported, including SiO2,[17] Al2O3,[26] etc. Liu et al. demonstrated the tunable SiO2 interior insulator and obtained the optimized SERS substrate.[15] Compared with the traditional uncontrolled aggregation of the metal nanoparticles in the aqueous phase, the metal nanoparticles wrapped with an ultra-thin silica or alumina dielectric layer can greatly enhance the SERS signal.[15] Moreover, the dielectric layer (SiO2) can be precisely controlled by plasma enhanced chemical vapor deposition(PECVD) or atomic layer deposition (ALD) method.
Compared with the Ag@SiO2@Ag structure design, the ZnO@Ag/SiO2@Ag design highlights the 3D structure substrate, which increases the spatial density of hot spots due to large specific surface area. Meanwhile, compared with the ZnO@Ag structure design, the ZnO@Ag/SiO2@Ag design can improve the SERS effect by changing the thickness of the interior dielectric layer SiO2. Therefore we put forward a simple method of preparing a 3D SERS substrate to implement the SERS 3D hot spots and achieve high field enhancement factors simultaneously. For the 3D ZnO@Ag/SiO2@Ag nanorod arrays, the ZnO nanorods can be prepared by the simple hydrothermal method, and the ultrathin SiO2, as an interior insulator between the Ag inner layer and Ag outer layer, can be precisely tuned by changing the PECVD growth time, and the multilayer structure can be prepared on different substrates, such as silicon, ITO and PET. Through a study on theoretical simulation and SERS activities, the 3D structures can remarkably enhance the SERS signals. The method will provide new ideas in the development of flexible substrates and the expansion of 3D SERS substrates.
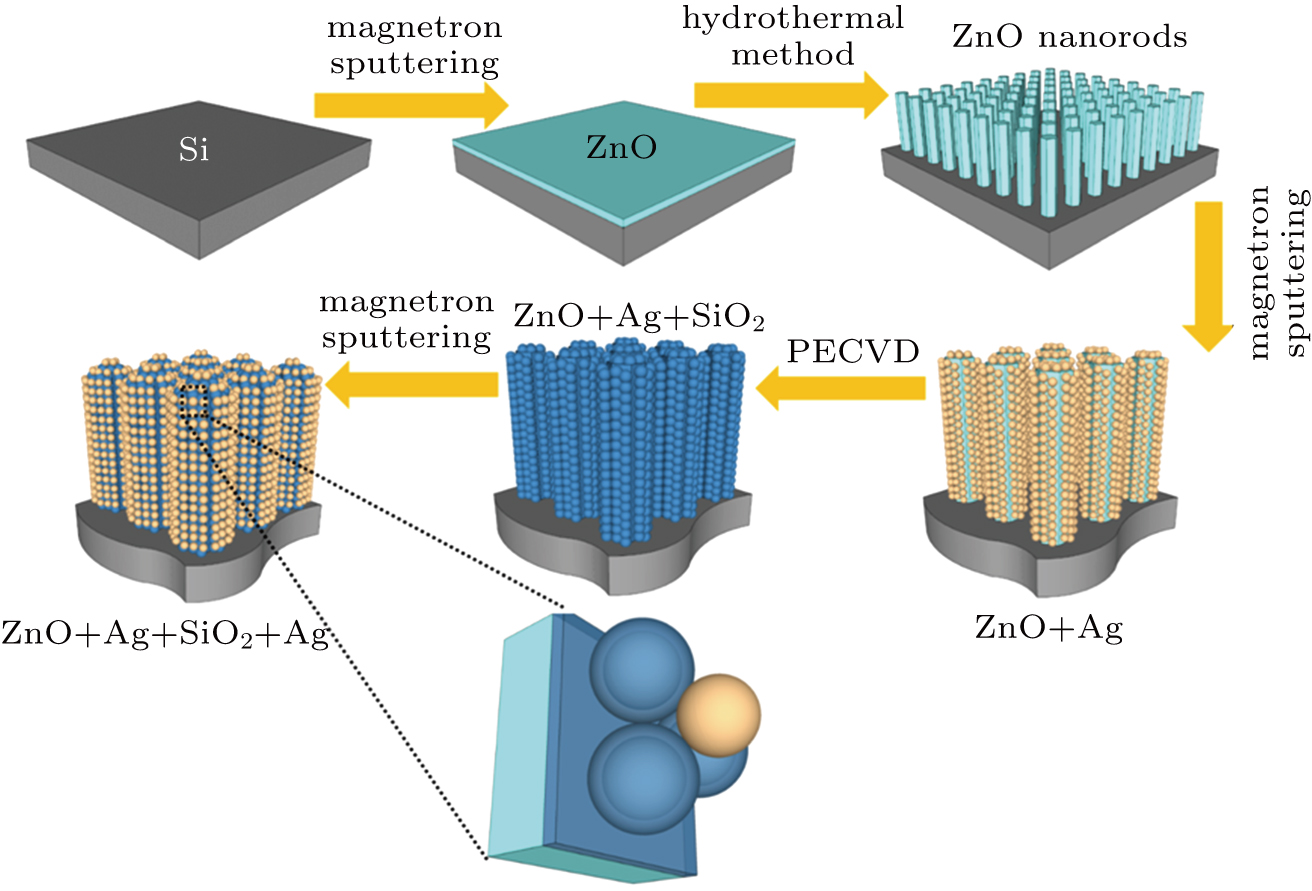
The fabrication process of multilayer SERS substrate is illustrated schematically in Fig.
The 3D SERS substrates were analyzed by scanning electron microscopy (SEM) (FEI/Nova Nano SEM 450). The SERS measurements were performed at room temperature with a Raman microscope (Horiba Jobin Yvon/LabRAM HR Evolution). The wavelength of the excitation laser of SERS experiments was 532 nm, the laser power was about 1 mW, the spectra acquisition time was 10 s, and an optical microscope with a 100× objective lens was used. The laser beam was normally incident on the substrate, which was parallel to the surface normal for SERS spectra collection. In this study, the plasma enhanced chemical vapor deposition system (SENTECH/SI 500D) was used to grow SiO2 dielectric layer, and the multi-cathode sputter deposition platform (Denton Vacuum/Discovery 635) was used to implement the metal deposition. Rhodamine 6G (R6G) was used as the probe molecule for the SERS test. The solution was diluted to a solution with a concentration of 1 × 10−6 M. The prepared ZnO@Ag/SiO2@Ag substrate was soaked for 12 h and then washed with ethanol to obtain the sample to be measured.
Figure
The ZnO@Ag/SiO2 nanorods are further investigated by using transmission electron microscope (TEM) and energy dispersive x-ray spectroscopy (EDXS) as shown in Fig.
As is well known, the SERS performance of multilayer structure is mainly determined by the density and morphologies of outer layer noble metal nanoparticles. In order to evaluate the importance of the outer layer noble metals, the SERS spectra of the as-prepared samples are systematically collected. Figure
In order to confirm the direct influence of SiO2 dielectric layer thickness on SERS performance, the SiO2 dielectric layer is varied with a certain thickness of Ag. Figure
To better understand the contribution of Ag nanoparticles and dielectric layer in ZnO@Ag/SiO2@Ag multilayer nanorods, a 3D finite difference time domain (FDTD) method is used. Our theoretical results show that there are stronger electromagnetic enhancements on the interface between Ag nanoparticles and dielectric layer, and that the resonant coupling between the inner dielectric layer core and outer Ag layer can propagate surface plasmons to induce SPR with enhanced electrical fields as shown in Fig.
In this work, ZnO@Ag/SiO2@Ag multilayer structure is successfully synthesized by the physico–chemical combination method. The geometries of the nanorods and the thickness of dielectric layer and Ag layer can be well controlled, which opens up the way to modulating the local electric field and therefore the SERS enhancement factor is controlled. The products are characterized by SEM, TEM, Raman spectrum, and theoretical simulation. By regulating the thickness of both the Ag outer-layer and the SiO2 dielectric layer, the ultimately optimized SERS substrate is implemented. The experimental results accord with FDTD simulation. Our results suggest that the ZnO@Ag/SiO2@Ag substrate is feasible to generate large-area hot spots and achieve high field enhancement factor simultaneously. Moreover, comparing with the ZnO@Ag or Ag@SiO2@Ag structures, the proposed method can achieve 3D multilayer structure electromagnetic field regulation, and the simplicity and generality of our method for further understanding the SERS mechanism induced by the SPR effect offer great advantages. In view of the green and sustainable application protocol, we expect that the 3D multilayer structure has potential applications in flexible substrate, SERS, photocatalysis and biological imaging.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] |