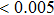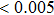† Corresponding author. E-mail:
In this study, we investigate the cross-frequency coupling and functional brain networks in the subjects with temporal lobe epilepsy (TLE) using interictal EEG signals. The phase to phase synchronization within and across frequency bands is calculated and a significant difference between the epilepsy and control groups is observed. Compared with the controls, the epilepsy patients exhibit a stronger within-frequency coupling (WFC) within theta and beta bands, and shows a stronger cross-frequency coupling (CFC) in the delta–alpha and theta–alpha band pairs, but a weakened CFC in alpha–beta band pairs. The weakened coupling between alpha and high frequency band reflects a suppression of phase modulation between the brain regions related to epilepsy. Moreover, WFC and CFC are positively correlated, which is higher in the patients relative to controls. We further reconstruct functional brain connectivity and find that both WFC and CFC networks show small-world properties. For the epilepsy, the small-world efficiency is enhanced in the CFC networks in delta–alpha and theta–alpha band pairs, whereas weakened between alpha and beta bands, which suggests a shift away from the optimal operating point in the epileptic brain with a new balance between WFC and CFC. Our results may help us to understand the important role of information communication across different frequency bands and shed new light on the study of pathology of epilepsy.
Epilepsy (EP) is a chronic neurological disorder that is characterized by recurrent episodes of abnormal neural dynamics in the brain. Globally, approximately 50 million patients suffer from this kind of disease.[1,2] The specific causes of most epilepsy cases remain unclear. However, neurophysiological research aiming to reveal the generation mechanism of the epilepsy have never stopped, especially for partial epilepsies. Temporal lope epilepsy (TLE), as a common partial epilepsy, was traditionally thought to originate in specific areas of the cortex known as the seizure onset zone (SOZ) and then gradually spread to other areas, known as epileptogenic zones (EZ).[3] This spreading process typically results in dramatic changes in neuronal synchrony in multiple temporal-spatial scales.[4] Severe epileptic seizures can cause excessive synchronized or abnormal activities in the brain cortex. These abnormal synchrony dynamics in the brain can impact function connectivity and information transmission within and cross different brain regions.[5,6]
Brain functions and dynamics have been widely studied through brain imaging techniques such as electroencephalogram (EEG), functional magnetic resonance imaging (fMRI), and positron emission tomography (PET).[7–9] EEG, as a non-invasive neuroimaging tool, has frequently been utilized to diagnose different neurological disorders and diseases.[10,11] Clinically, it has been proven that any pathology in the brain neurons leaves a signature on EEG signals.[12] Hence, scalp EEG is often the preferred choice and lots of advanced algorithms have been proposed based on EEG signals to analyse the abnormality induced by epileptic activity. In addition, the EEG based method for locating the epilepsy affected brain area is convenient and EEG signals provide good temporal resolution.[13] Many linear and nonlinear dynamics analyses based on EEG signals have revealed the characteristic affects of epilepsy on brain activity. Quraan et al. applied power spectrum density (PSD) on EEG signals recorded from left temporal lobe epilepsy (LTLE) patients and reported that patients exhibited higher power in theta band but lower power in high alpha bands.[14] Heijer et al. explored the functional brain connectivity using EEG signal and found global and focal functional connectivity were negatively correlated.[15]
The transmission of information in the brain is based on electrophysiological processes and refers to the type of connectivity between neurons.[16,17] The notion of synchronization was proposed to describe the functional connectivity between different neuronal oscillations.[18] Pfurtscheller et al. investigated event-related synchronization using EEG signals and found that the brain oscillations were negatively correlated with their amplitude.[19] Elul et al. demonstrated that the amplitude of oscillations was proportional to the number of synchronously active neural elements.[20] Discerning the synchronization of brain oscillations might offer the possibility of gathering deeper insight into the development of epileptic seizures. Thus, synchronization phenomena in chaotic system has attracted considerable interest in EEG studies of epilepsy. Mormann et al. applied mean phase coherence to investigate the phase synchronization of TLE and observed the decreased synchronization before the onset of seizure.[21] Ortega et al. reported that the TLE patients showed less synchronization degree in mesial areas than that in contra-lateral areas, which may be associated with seizures.[22] In addition, enhanced synchronization at the end of the seizure has also been observed in the focal epileptic patients and the increasing synchronization of neuronal activity can be considered as an emergent self-regulatory mechanism for seizure termination.[23]
The synchrony between EEG signals occurs not only within a given frequency range, which is known as within-frequency coupling (WFC), but also between frequency bands, which are referred as cross-frequency coupling (CFC). The CFC was recently considered in neuroscience to play a crucial role in supporting multiple brain functions, such as memory, learning and cognition.[24–26] Three different types of CFC are generally explored in recent studies, where each type represents a particular interaction pattern between frequency bands, they are: amplitude to amplitude coupling (AAC), phase to phase coupling (PPC) and phase to amplitude coupling (PAC). Li et al. reported that CFC is a feature of the brain’s neural networks and could effectively extract signals from brains.[27] Wang et al. found increased theta-to-gamma but decreased alpha-to-gamma band AAC in fragile X syndrome patients.[28] Canolty et al. observed robust PAC between high frequency gamma and low-frequency theta and found cortical activity was regulated by coupling patterns between low and high frequency bands.[29] In addition to the theta-gamma PAC, enhanced PPC among alpha, beta and gamma bands can be found in continuous mental arithmetic tasks.[30] Importantly, PPC is able to investigate the functional interaction between different brain regions across multiple frequency bands. Thus, in this study, we utilized PPC to explore the abnormality of cross-frequency synchronization between different regions.
Functional brain connectivity is an important feature in the study of the alteration of brain functions based on EEG.[31–33] Given that the electrophysiological dynamical system comprises numerous neurons, which makes it structurally and functionally connected, network theory is naturally applied to study the brain dynamics. Network theory, which is also referred to as graph theory, has shown great promise in exploring the brain impaired network function resulting from neurological diseases.[34,35] Many network metrics have been proposed to study network functional dynamics associated with various neurologic disorders.[36–38] The main outcome of these studies is that interactions in the human brain possess small world network (SWN) properties, which are in fact highly clustered like regular lattices and can be well distinguished from random networks.[39–41] Detailed network analyses have recently investigated the alteration of SWN property of brain activities affected by neurological diseases. Pedersen et al. reported that the clustering coefficient and local efficiency of focal epilepsy patients were significantly higher than healthy controls, whereas global efficiency were not significantly different between two groups and the increased clustering and local efficiency contributes to the improved SWN property.[42] Ponten investigated how the SWN property changed during ictal period based on EEG signals and found the increased SWN feature in the alpha, theta and delta bands.[43]
The main purpose of this study is to explore cross-frequency coupling and functional brain networks in epilepsy patients. Scalp EEG data were collected from patients with temporal lobe epilepsy during the interictal period and normal controls. Then, the phase synchronization index (PSI) was applied to measure the phase synchronization within and between frequency bands. The synchronization strength in WFC and CFC cases was first investigated. Then, the regional PSI was computed to explore how functional connectivity is distributed throughout the brain’s regions. Finally, we constructed WFC and CFC networks and extracted graph theory metrics to investigate the coupling difference within and across frequency bands.
This study’s subjects were recruited via an announcement at Tangshan Gongren Hospital and consist of 20 right-handed patients diagnosed with right temporal lobe epilepsy (8 females and 12 males, aged from 20 to 36) and 18 right-handed normal controls (10 females and 8 males, aged from 21 to 37). The determination of seizure lateralization was based on the presence of unilateral epileptiform activity evidenced by continuous video-EEG and magnetic resonance imaging (MRI) assessment. All of the participates were native speakers and had no reported history of other neurological disorders, neurological active medication, or any other factor that may affect EEG activity. All subjects gave their written informed consent and the experiment was approved by the ethics committee of Tangshan Gongren Hospital.
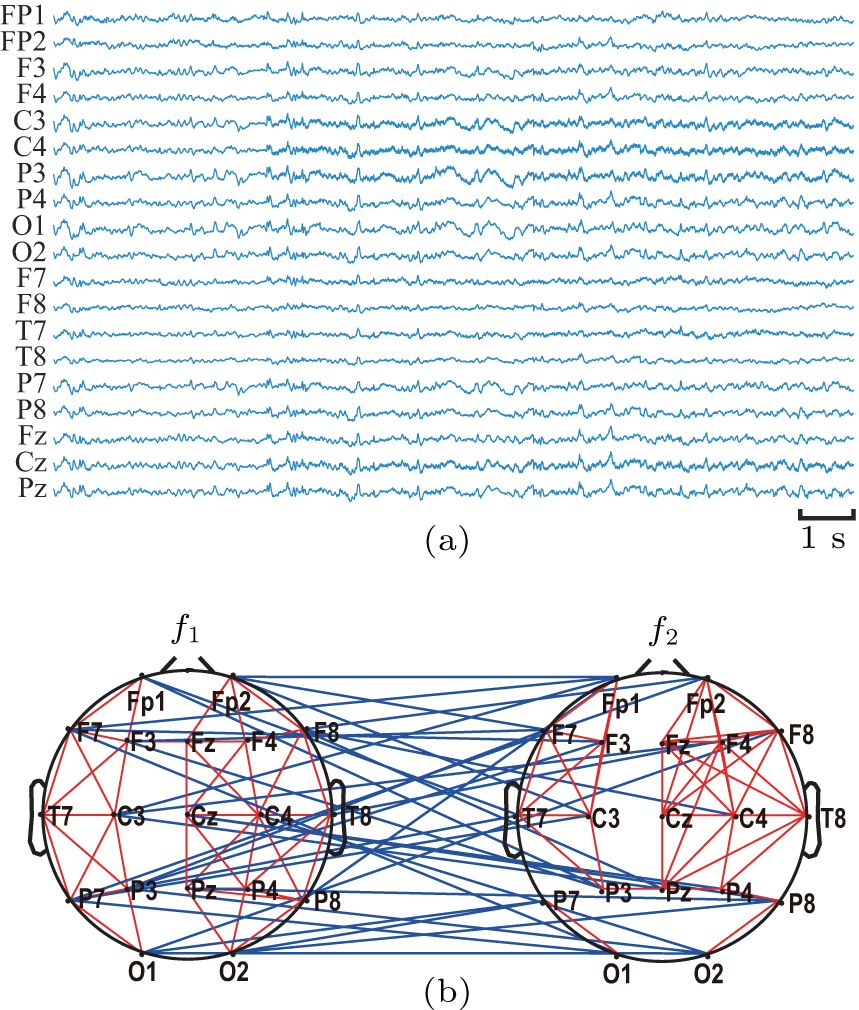
About 15 minutes EEG signals were collected after a 3-min relaxation phase for each subject. During the acquisition, the subject was required to sit upright on a comfortable chair in a semi-dark quiet room and to stay awake with their eyes closed. In addition, they were told in advance that any movements should be avoided during the experiment, such as body movements, eye movements and blinks. All EEG signals were collected by NIHON KOHDEN system. The Ag–AgCl scalp electrodes were placed according to the international standard 10-20 placement system, EEG data were recorded from the 19 scalp loci (Fp1, Fp2, F7, F3, Fz, F4, F8, T7, C3, Cz, C4, T8, P7, P3, Pz, P4, P8, O1, and O2). The linked earlobe A1 and A2 were used as a reference. The EEG data were filtered using a band-pass filter of 0.5 Hz–70 Hz and the sampling frequency was 200 Hz.
In the offline data preprocessing, the noise in the EEG data was first filtered out and useful information was extracted by a band-passed finite impulse digital filter of 0.5 Hz–30 Hz. Then, the EEG data of each channel was re-referenced to the average of all channels to remove systematic effects which might be caused by referencing to a particular channel, especially in the context of synchronization analysis. Next, we applied complex Morlet wavelet transform to compute the instantaneous phase in four frequency bands: delta (0.5 Hz–4 Hz), theta (4 Hz–8 Hz), alpha (8 Hz–15 Hz), and beta (15 Hz–30 Hz). The complex mother Morlet wavelet has a Gaussian shape around the central frequency f:
 |
To investigate the phase coupling within and between different frequency bands, the instantaneous phase was computed from the wavelet coefficients for all electrodes, which is defined as
 |

 |
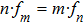
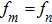
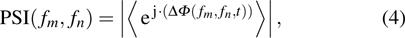 |

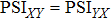
The quantified coupling within and across four frequency bands were used to construct the functional network including WFC network and CFC network, as shown in Fig.
In graph theory, the network was represented as a geometric model with a set of nodes (representing processing centers) and edges (represents the information transition between nodes). In this context, a number of local and global graph theory metrics were calculated. Local efficiency was used to show how efficient the communication is between the node k and its neighbor nodes. Local efficiency of node k is defined as
 |
 |
 |
 |
 |
 |


The Wilcoxon rank sum test was applied to evaluate the group difference in the functional connectivity and graph theory metrics between epilepsy patients and normal controls. A p value smaller than 0.05 indicates significant difference. We further used Bonferroni correction for multiple comparisons as the tests are performed across different frequency bands. In this paper, the statistical analyses were performed using SPSS (version 19.0) software package and Brain Connectivity Toolbox (BCT) was used to calculate the graph metrics (http://www.brain-connectivity-toolbox.net).
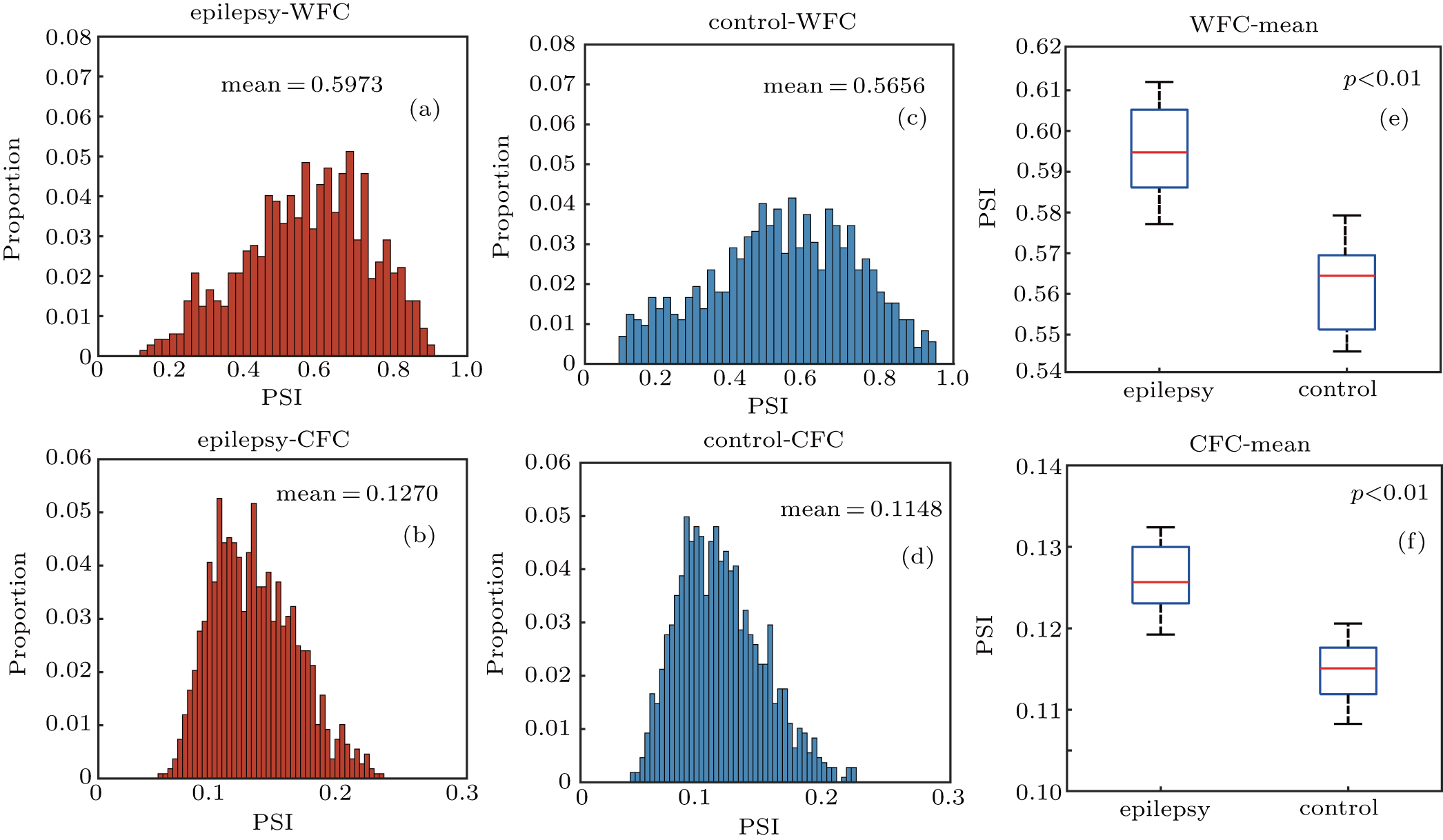
Using the measure of PSI, we explored the phase synchronization dynamics within and between different frequency bands. Figure
| Table 1.
Average PSI values of both groups for within and gross frequency bands. . |
The distributions of WFC and CFC values were investigated, respectively, for both epilepsy and normal control. The results are shown in Fig. 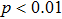
We further investigated the difference of coupling capability of individual channels using regional PSI by averaging PSI values of one channel with all other channels. Figure
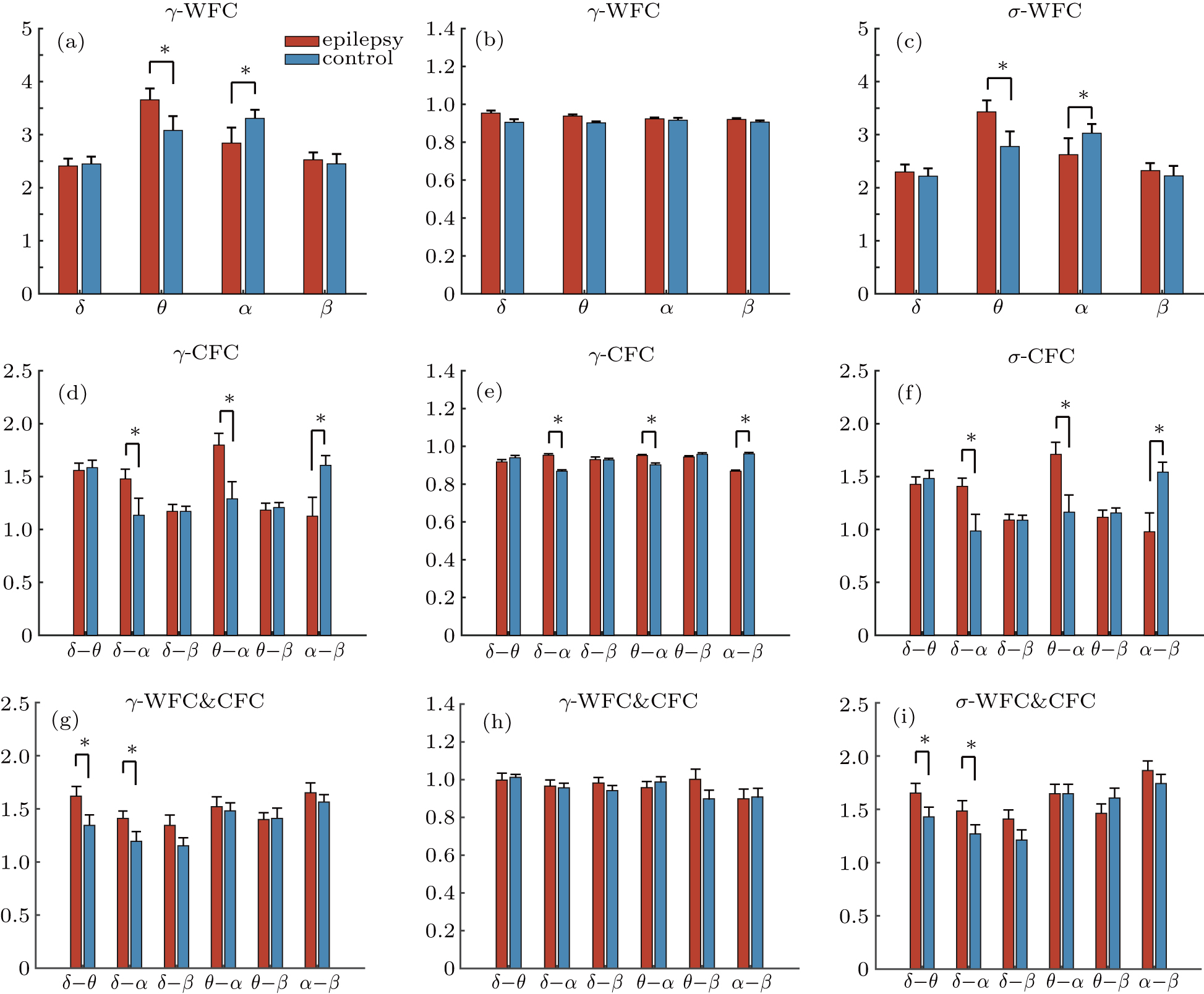
Finally, we constructed weighted functional networks within and cross frequency bands for both groups. The strongest 30% coupling strength was preserved to make sure that there was no isolated node in the network. Graph theory metrics was applied to gain more insight into the alteration of WFC and CFC networks in the epilepsy brains. Normalized local efficiency γ, normalized global efficiency λ, and small world network (SWN) index σ were calculated and shown in Fig.
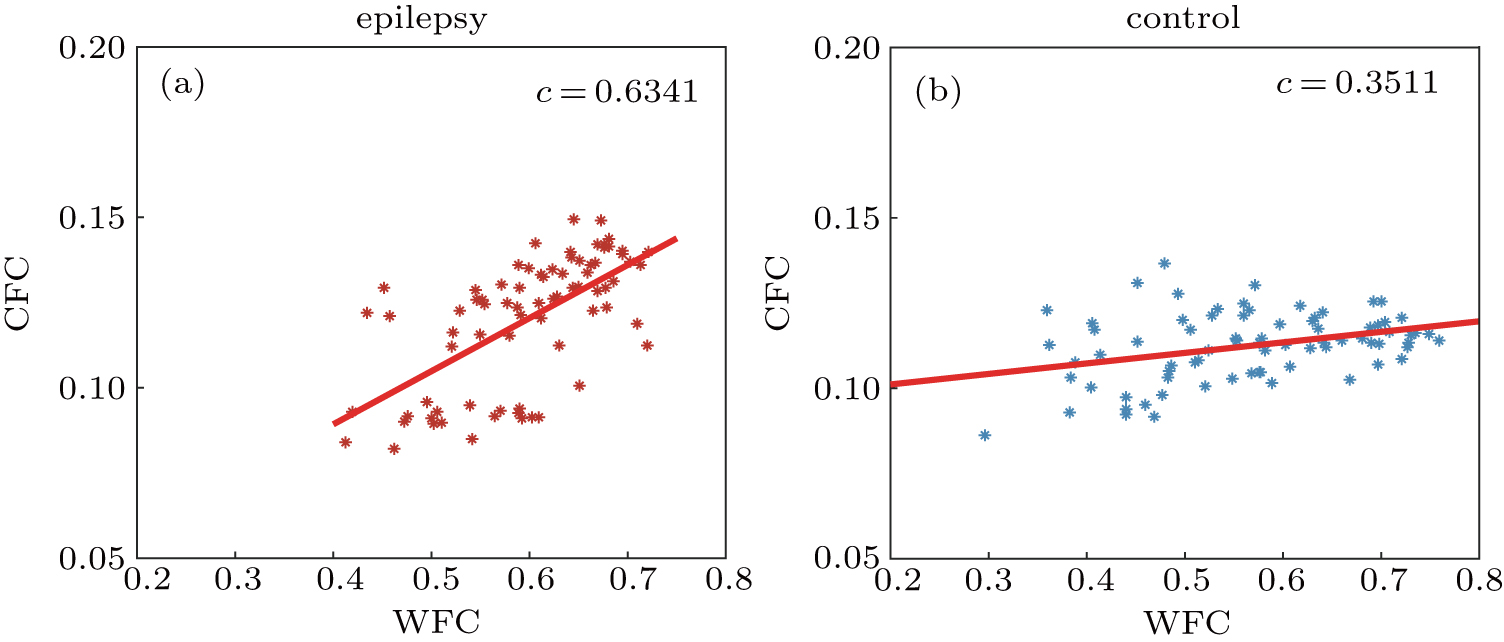
In this paper, we investigated the cross-frequency coupling and functional brain networks in epilepsy patients during the interictal period. The phase synchronization index (PSI) was utilized to quantify the coupling strength between each pair-wise channel within and across different frequency bands of EEG. Our results show that a significant difference of coupling strength between two groups can be observed in both WFC and CFC. Compared with the normal controls, the epilepsy patients exhibit stronger WFC coupling within theta and beta bands, and a stronger CFC coupling in the δ–α and θ–α band pairs. Importantly, a positive correlation between WFC and CFC is found, and the correlation is higher in the epilepsy. We further explored the properties of brain network based on the coupling within and between frequency bands. It is demonstrated that both WFC and CFC networks show small-world properties. We found that for the patients, the small-world efficiency is enhanced in the CFC networks between alpha and low frequency band but weakened in the CFC networks between alpha and high frequency bands, which indicates that oscillation in alpha band plays an essential role in information communication cross different frequency bands.
A growing body of evidence from the empirical data shows that altered synchronization from a balanced state can be associated with various neuropsychiatric disorders.[22,46,47] As a common neurological disease that results from intermittent abnormal neuronal firing, the abnormalities of synchronization in epilepsy have been investigated in multiple temporal-spatial scales, which can be also confirmed by the scalp EEG recorded from the whole brain.[5,12,13] By using EEG of TLE patients, Bettus and colleagues have reported increased synchronization within the epileptogenic zone, which was significant in most frequency bands (e.g., theta and alpha).[13] Horstmann et al. have further reported increased broad band synchronization in epilepsy patients.[48] In this work, we found that epilepsy patients exhibit higher synchronization in the theta and beta band but lower synchronization in the alpha band. These results are in accordance with the observations of Quraan’s work, in which epilepsy patients demonstrated significantly higher synchronization in the theta band but lower synchronization in alpha band.[14] Therefore, it is reasonable that abnormal synchronization of EEG can be an important character for epilepsy.
Apart from the investigation of synchronization within band, the cross-frequency coupling of EEG has been explored in previous epileptic studies.[13,14] Guirgis et al. have found that the phase-amplitude coupling (PAC) strength between delta and high frequency was higher in the epileptogenic zone.[49] Zhang et al. have studied the coupling strength of multi-channel EEG signals from temporal lobe epilepsy and observed significantly increased PAC strength in the delta, theta, and alpha oscillations.[50] However, the CFC analyses mentioned above were mainly performed on individual electrode or brain areas. In this study, we applied a phase to phase coupling measurement to calculate the synchrony between pair-wise channels and cross various frequency bands. It was shown that the CFC in the epilepsy was significantly decreased between alpha and high frequency bands. Similarly, Ramon also found the cross-frequency couplings between alpha and gamma bands were decreased in and near to the seizure area.[51] In fact, the alpha to higher frequency band interactions are related to the phase modulation of motor region by premotor region. For instance, finger movement can cause the suppression of phase-phase CFC to the α–β band.[52] According to the widely held view that CFC plays an important role in supporting advanced brain functions such as memory and cognition,[25,26] the abnormalities of between band synchronization may also be linked with the epileptiform activity or dysfunction in epileptic brain.
The within and between frequency couplings should not be treated separately because they may show some specific relationship.[53] A recent research integrating cross-frequency and within band functional interactions has shown that the strength of cross-frequency connectivity significantly correlates with the averaged magnitude of within band connections, and the strength of CFC strongly influence the spatial network similarity across different frequency bands.[54] In our previous work, the relationship between WFC and CFC has also been investigated for patients with Alzheimer’s disease (AD). A significant correlation was observed as a result of declined cross frequency coupling for AD patients.[55] In this study, we found that WFC and CFC are positively correlated and such correlation is higher for the patients. This correlation difference between epilepsy and the control group can be attributed to a significant enhancement in cross-frequency couplings compared with the within band interactions in epileptic brain. These findings also imply that it may be a good choice to analyze the functional interactions across the electrophysiological spectrum in a single framework for the investigation of brain networks.
Considering the group difference of within and between band synchronization strength, we further extracted the graph theory metrics to explore the properties of brain networks constructed by the coupling within and cross frequency bands. In this study, decreased small world properties can be observed in the alpha network, which is in accordance with Quraan’s study in which the patients showed deviations toward a random network in the high α band. While for the CFC network that contains connections within and between frequency bands, the patients showed elevated small-world properties in delta-alpha and theta-alpha band pairs. Besides, the epilepsy group also exhibit improved small-world properties in the delta-alpha network containing both within and between band connections. These alterations suggest an enhanced capacity of information transmission between alpha and low frequency bands, which may compensate for the suppression within alpha band for the epilepsy patients. According to the widely held view, the healthy human brain operates at a transition point that allows the brain to switch between separation and integration. The changes in the network properties (for both WFC and CFC cases) also imply a shift away from the optimal operating point in the epileptic brain with a new balance between WFC and CFC. Taken together, our results indicate that the investigation of cross-frequency functional connectivity can provide more information about the network abnormalities for the patients and may facilitate our further understanding of epilepsy and other neurological diseases.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [52] | |
| [53] | |
| [54] | |
| [55] |