† Corresponding author. E-mail:
‡ Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 11325421).
We have realized robust quantum anomalous Hall samples by protecting Cr-doped (Bi,Sb)2Te3 topological insulator films with a combination of LiF and AlOx capping layers. The AlOx/LiF composite capping layer well keeps the quantum anomalous Hall states of Cr-doped (Bi,Sb)2Te3 films and effectively prevent them from degradation induced by ambient conditions. The progress is a key step towards the realization of the quantum phenomena in heterostructures and devices based on quantum anomalous Hall system.
A topological insulator (TI) is an insulator with topologically protected gapless surface states in its bulk gap.[1–4] This class of materials has received extensive research interests in the past decade for a number of novel quantum phenomena predicted in them.[5–14] The quantum anomalous Hall effect (QAHE), a quantum Hall effect that can occur without external magnetic field, is among the few quantum phenomena that have been experimentally observed.[15–17] The effect is expected to appear in a TI film with its time-reversal symmetry broken by ferromagnetism, introduced via magnetic doping or proximity.[5–7,10,18] The QAHE was experimentally realized in magnetically doped (Bi,Sb)2Te3 three-dimensional (3D) TI films grown by molecular beam epitaxy (MBE), opening a path toward the observation of many other exotic quantum effects such as topological magnetoeletric effect.[9–11] and chiral topological superconductivity.[13,14]
The magnetically gapped topological surface states play a key role in QAHE in magnetic 3D TI films.[19] Hence QAHE is sensitive to the surface conditions of the films. QAHE was first observed in Cr-doped (Bi,Sb)2Te3 films with no capping layer.[15] Such bare magnetic TI films cannot hold the QAH property in atmosphere for a long period of time (usually less than one month even in inert atmosphere) and in some microfabrication processes such as electron beam lithography (EBL) and atomic layer deposition (ALD). This greatly impedes studies and applications of QAH-based structures and devices. It is highly desired to find a satisfactory capping layer material capable of preserving the native properties of QAH films and effectively protecting them from degradation under various conditions.
We have tried to protect Cr-doped (Bi,Sb)2Te3 QAH films by depositing a layer of tellurium on them. Tellurium atoms form a single-crystalline film covering the whole surface of the films,[20] but destroying the QAHE in them. Figures
Lithium fluoride is an insulator with rocksalt structure (a = 4.026 Å) and a gap of 12.6 eV.[21] The small lattice constant and the big difference between the electro-negativities of Li and F make the compound resistant to diffusion and reaction with atoms in the atmosphere and adjacent layers. So it is a good candidate material for a capping layer of QAH samples. In this study, we investigated the influence of LiF capping layer on the QAHE of Cr-doped (Bi,Sb)2Te3 films and found that LiF can make an ideal protection layer for the QAH films if covered with a layer of water-resistant AlOx.
The QAH samples used in this study are 5-QL thick Cr0.15(Bi0.1Sb0.9)1.85Te3 films prepared on SrTiO3 (111) substrates with MBE. The films were grown in an ultra-high vacuum chamber (base pressure < 1.0 × 10−10 mbar, 1 bar = 105 Pa) equipped with a reflective high energy electron diffraction (RHEED) facility. Lithium fluoride and Al were prepared by thermal evaporation of LiF powder and Al granules with home-made evaporators, respectively. The QAH films were kept at room temperature for LiF and Al deposition. The transport properties of the films were measured with standard ac lock-in methods. Strontium titanate substrates were used as the dielectric layer of the bottom gate to tune the chemical potential of the films.[22,23]
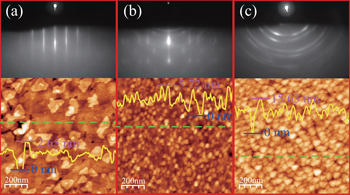
Figure
We can also observe diffraction circles indicating existence of islands with different crystalline orientations. With increasing thickness of LiF, the transmission spots gradually disappear, and eventually only the diffraction circles remain (Fig.
In Figs.
To check if an LiF capping layer can effectively protect the QAHE of Cr-doped (Bi,Sb)2Te3 films, we measured the LiF-capped Cr0.15(Bi0.1Sb0.9)1.85Te3 film after keeping it in an argon glove box for 60 days. The data are plotted with red lines in Figs.
It is not clear why LiF cannot effectively protect the QAH films with its good chemical stability and compact lattice structure. A fact probably relevant is that LiF is slightly soluble in water (0.132 wt% at 23.7 °C).[24] Thus an LiF layer could adsorb water molecules from the atmosphere and in turn influence the properties of the underlying QAH films. Such a deficiency of LiF can be removed by capping it with a water-resistant layer such as AlOx.
We deposited 3-nm thick Al on an LiF (5 nm)-covered Cr0.15(Bi0.1Sb0.9)1.85Te3 film and had it oxidized into a compact AlOx layer in air. As shown in Fig.
In order to further test the protection effect of the composite capping layer, we attempted using ALD to deposit AlOx on the AlOx/LiF-capped samples with ozone (O3) as the precursor at 70 °C for 2 hours. Such a process is a key step toward the realization of dual-gate structures and tunneling junctions based on quantum anomalous Hall films. The strong oxidation of ozone plus the elevated temperature used in ALD can easily destroy the fragile quantum anomalous Hall states of the magnetically doped (Bi,Sb)2Te3 films. The black lines in Figs.
In summary, we found that a LiF capping layer can very well preserve the QAHE of Cr-doped (Bi,Sb)2Te3 films, but is unable to effectively protect them from contamination of ambient conditions. By covering the LiF surface with an AlOx layer, we significantly improved the protection effect of LiF, which enabled us to obtain robust QAH samples that survive a longer period of time and endure the harsh conditions of some microfabrication processes. The excellent performance of AlOx/LiF composite capping layer makes it useful in various studies and applications of QAHE and Bi2Se3 family TIs.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 |