†These authors contributed equally to this work.
‡Corresponding author. E-mail: yukisusu@aliyun.com
§Corresponding author. E-mail: huaai@scu.edu.cn
*Project supported by the National Key Basic Research Program of China (Grant No. 2013CB933903), the National Natural Science Foundation of China (Grant Nos. 20974065, 51173117, and 50830107), and the Scientific Research Start-up Fund of Kunming University of Science and Technology (Grant No. KKSY201305089).
Recent progress of the preparation and applications of superparamagnetic iron oxide (SPIO) clusters as magnetic resonance imaging (MRI) probes is reviewed with regard to their applications in labeling and tracking cells in vivo, in diagnosis of cardiovascular diseases and tumors, and in drug delivery systems. Magnetic nanoparticles (NPs), especially SPIO nanoparticles, have long been used as MRI contrast agents and as an advantageous nanoplatform for drug delivery, taking advantage of their unique magnetic properties and ability to function at the molecular and cellular levels. Due to advances in nanotechnology, various means to control SPIO NPs’ size, composition, magnetization and relaxivity have been developed, as well as ways to usefully modify their surface. Recently, self-assembly of SPIO NP clusters in particulate carriers—such as polymeric micelles, vesicles, liposomes, and layer-by-layer (LbL) capsules—have been widely studied for application as ultrasensitive MRI probes, owing to their remarkably high spin–spin ( T2) relaxivity and convenience for further functionalization.
Medical imaging has long served as an important clinical tool for the diagnosis and treatment of diseases. With the development of molecular and cellular biology, molecular imaging has emerged as a new biomedical imaging field for understanding pathological changes at the cellular and molecular levels by using modern technologies and instruments.[1– 3] Several imaging modalities are involved in molecular imaging, including magnetic resonance imaging (MRI), x-ray computed tomography (CT), positron emission tomography (PET), single photon emission computed tomography (SPECT), fluorescence imaging (FLI), and ultrasound imaging. Each imaging modality has its own advantages and disadvantages in terms of spatial resolution, targeting, and penetration depth.[1] Image contrast can be improved with the help of contrast media, and small pathological changes can be easily discovered; multimodality imaging, combining two or more imaging techniques, can compensate for the disadvantages of each mode and provide more information than either single modality used alone.[4, 5]
Magnetic resonance imaging (MRI) is among the best noninvasive clinical methodologies today for assessing the anatomy and function of tissues. MRI offers several advantages including excellent temporal and spatial resolution, lack of exposure to radiation, rapid in vivo acquisition of images, and a long effective imaging window. However, MRI is much less sensitive than nuclear medicine or fluorescence imaging, so more than 40% of all MRI examinations rely on a contrast agent.[6] Superparamagnetic iron oxide (SPIO) nanoparticles are strong enhancers of proton relaxation, with superior MR T2 (transverse relaxation) shortening effects, and can be used at low concentrations.[7– 9] With the help of SPIO contrast agents, great progress has been made with MRI in gene delivery, cell tracking, drug delivery, tumor diagnosis and many other fields.[10– 13] Commercial SPIO-based MRI contrast agents such as FeridexⓇ and ResovistⓇ are available and widely used in clinics.
An SPIO nanoparticle-based MRI contrast agent can shorten the transverse relaxation time (T2) of the surrounding water and bring negative contrast, resulting in a hypointense imaging signal.[7, 9] One prominent characteristic of SPIO nanoparticle-based MRI contrast agents is that they have very high T2 relaxivity that can reach up to 900 Fe mM− 1· s− 1, [14] which is essential both in imaging small pathological changes and in in vivo tracking of drug delivery systems. The T2 relaxivity of SPIO nanoparticles depends on several factors, including particle composition, particle size, magnetic field, and self-assembly structure.[15, 16] Generally, a single SPIO nanocrystal with a larger size has a higher T2 relaxivity, as long as its particle size is smaller than 20 nm in order to keep its superparamagnetic properties. One interesting discovery is that the T2 relaxivity of an SPIO cluster of multiple nanoparticles is typically greater than that of a single nanocrystal.[10, 11, 17– 21] Although the reason for this phenomenon is still under study, SPIO clusters are considered to be an effective basis for developing sensitive SPIObased MRI contrast agents. Similar research has been reported − for instance, polymer micelle-encapsulated SPIO clusters have been demonstrated as sensitive probes for tumor imaging, and cell labeling and tracking.[11, 13, 21] This clustering concept can be stretched to SPIOs contained in liposomes, polyelectrolyte capsules and other particulate carriers, and these nanoparticle/carrier structures with further surface functionalization show similar characteristics. In this review, we will discuss how these SPIO nanoparticle clusters function as MRI imaging probes, as well as their potential applications in cell tracking, cardiovascular disease diagnosis, drug delivery and tumor diagnosis.
A major limitation for MRI is its lower sensitivity compared with nuclear radiation techniques or fluorescence imaging, especially when used in monitoring small tissue lesions or molecular activity. MRI contrast agents can shorten either longitudinal or transverse proton relaxation time and thus provide higher image contrast. Two kinds of MRI contrast agents are now available for both basic and clinical applications: paramagnetic small molecule agents and superparamagnetic iron oxide nanoparticles (SPIO NPs).[22, 23] Relaxivity (r) is an important parameter used to characterize the ability of MRI contrast agents to shorten the proton longitudinal relaxation time (T1) or transverse relaxation time (T2). Due to the relatively high transverse relaxivity (r2) values, SPIO-based MRI probes are strong enhancers of proton relaxation, with superior MR T2 (transverse relaxation) shortening effects at a much lower concentration, [8] and they have been used in clinical MRI practice for diagnostic applications (e.g., FeridexⓇ and ResovistⓇ ). For instance, the active particles of ResovistⓇ are carboxydextrane-coated SPIO with a hydrodynamic diameter ranging from 45 nm to 60 nm, and the r2 and r1 are 151.0 mM− 1· s− 1 and 25.4 mM− 1· s− 1 respectively. Aggregates of multiple SPIO nanocrystals were found to have much greater effects on T2, and the T2 relaxation rate is enhanced dramatically with increased particle size.
High quality SPIO nanocrystals are usually synthesized in organic phase at high temperatures for better control of particle size and morphology.[24] Due to the hydrophobic surface with a large surface-area-to-volume ratio, SPIO nanocrystals without any surface coating will aggregate into large clusters within which strong magnetic dipole– dipole attractions exist and ferromagnetic behavior is observed. Moreover, naked SPIO nanoparticles are chemically active and are easily oxidized in air, leading to a loss of both magnetism and dispersibility. It is thus crucial to develop protection strategies to chemically stabilize the naked magnetic nanoparticles against aggregation and oxidation before any applications.[25, 26] The commonly used strategies include grafting or coating with organic species, such as surfactants or polymers, or coating with an inorganic layer, such as silica or carbon. Note that surface encapsulation not only stabilizes the magnetic nanoparticles, but can also functionalize them, e.g., with targeting ligands or imaging probes, depending on the desired application. Particulate type carriers, also known as colloidal carrier systems − including polymeric micelles, capsules, and vesicular microand nano-systems like liposomes − have been used widely for SPIO encapsulations. As mentioned before, the resulting SPIO clusters have remarkably high T2 relaxivity and sensitivity, phenomena that have attracted many researchers’ attention, considering the clusters as potential ultrasensitive MRI probes. In the following part of this review, we will focus on the preparation and applications of these nanoparticles/carrier structures as MRI contrast agents.
Polymeric micelles are nano-sized core-shell structures self-assembled by amphiphilic block copolymers in aqueous solution, with the hydrophobic chains of the copolymer forming the core of the micelle, and the hydrophilic chains forming the corona. Hydrophobic drugs or nanoparticles are soluble in the hydrophobic micelle core and can be transferred into aqueous phase by protection of the hydrophilic corona. After the pioneering work of Kabanov et al., [27– 29] polymeric micelles have been used widely to encapsulate therapeutic agents. As nanocarriers, polymeric micelles have shown various advantages such as good colloidal stability owing to their low critical micelle concentration (CMC), reduced toxic side effects for the conveyed drugs, long circulation time in the blood, etc., and they have long been used to encapsulate hydrophobic anticancer drugs, genes and contrast agents.[10, 30– 32] To prepare polymeric micelles with good bio-safety for in vivo applications, polymers with excellent biocompatibility and biodegradation, such as PEG, dextran and PEI, are widely used. Commonly used biocompatible polymers for micelle preparation are listed in Table 1.
| Table 1. Typical polymers used for amphiphilic polymer micelles. |
A number of magnetic nanoparticles can be encapsulated inside the hydrophobic core of a micelle, forming a closepacked cluster, which results in much stronger T2 effects than those of micelles containing a single magnetic particle, at the same iron concentration.[17– 19] This unique structure provides opportunities for the design and development of ultrasensitive MR probes. Previous studies by our group found that a cluster of SPIOs incorporated in a micelle structure shows high T2 relaxivities, reaching up to about 300 Fe mM− 1· s− 1. For instance, we have developed an amphiphilic star-like polymer (β – CD-Dex-g-SA) that can assemble into nano-sized micelle structures with high stability in aqueous solution. As shown in Fig. 1, multiple SPIO NPs were encapsulated in the hydrophobic cores of these micelles, resulting in closepacked SPIO clusters with relatively high T2 relaxivity of 436.8 mM− 1· s− 1.[20]
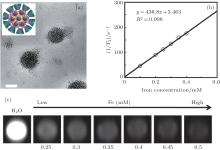 | Fig. 1. (a) TEM bright field image of the β – CD-Dex-g-SA/SPIO nanocomposite dried on a formvar coated copper grid (scale bar = 100 nm) and a cross-sectional schematic; (b) T2 relaxation rate (1/T2, s− 1) as a function of Fe concentration (mM) for SPIO nanocrystal loaded β – CD-Dex-g-SA micelles at 1.5 T; (c) T2-weighted MRI images (1.5 T, spin-echo sequence: TR = 5000 ms, TE = 12 ms) of β – CD-Dex-g-SA/SPIO micelles in water. Reprinted with permission from Ref. [20]. Copyright 2013, Elsevier Ltd. |
Liposomes, also known as artificially constructed vesicles, are formed in water by the self-assembly of lipid molecules. They have an aqueous core surrounded by a 4∼ 5- nm thick lipid bilayer membrane shell, which provides regions where hydrophilic and hydrophobic materials can be encapsulated simultaneously. This structure has been widely employed as a drug carrier for decades, resulting in several approved liposomal nanomedicines used clinically.[45, 46] The potential for use of liposomes as carriers of diagnostics has also been extensively investigated in the past decade. Liposomes containing SPIO nanoparticles, or paramagnetic chelates, have been evaluated mainly as MRI contrast agents for hepatosplenic imaging. Recently, lots of research has focused on the preparation and applications of these hybrid liposomenanoparticle assemblies as MRI contrast agents.[47– 49] For instance, Bé alle et al. developed an ultra magnetic liposome (UML) with a high amount of iron oxide nanoparticles (up to 30% by volume) encapsulated in the aqueous core.[49] In their study, a magnetic nanoparticle (γ -Fe2O3) aqueous suspension was first synthesized by alkaline co-precipitation of Fe2+ and Fe3+ , and then UMLs and UMLs-PEG of 217 nm and 238 nm in diameter were prepared by reverse phase evaporation. In vitro MRI study shows that the T2 relaxivities are higher for these hybrid liposome-MNPs assemblies than for single γ -Fe2O3 nanoparticles.
Similar to liposomes, polymer vesicles (or polymersomes), are also spherical shell structures that have an aqueous compartment enclosed by a bilayer membrane. Polymersomes are commonly formed by amphiphilic block copolymers in dilute aqueous solutions, making them easier to be further functionalized by adjusting the chemical composition of the block copolymers. Polymersomes are more stable and tougher than liposomes, their low molecular weight analogues, and polymersomes have tailorable membrane properties.[50– 52] These unique properties make them attractive candidates for multifunctional nanomedicine.[53] For instance, Yang et al. developed a stable and pH-responsive polymer vesicle nanocarrier for combining targeted delivery of an anticancer drug (DOX) and ultrasensitive MR imaging.[54] In the study, polymer vesicles grafted with folate (FA) targeting ligands were formed in an aqueous solution by self-assembly of heterofunctional amphiphilic tri-block copolymers (PEG114-P(Glu-Hyd-DOX)- PEG46-acrylate), and then hydrophilic SPIOs were encapsulated in the aqueous core as an active tumor-targeting MRI contrast agent (Fig. 2). The resulting SPIO/DOX-loaded vesicles demonstrated much greater T2 relaxivity than FeridexⓇ , a commercially available SPIO-based T2 contrast agent; the improvement was attributed to the high loading of SPIO NPs and the SPIO clustering effect in the aqueous core of the vesicles.
 | Fig. 2. (a) Schematic diagram of the multifunctional SPIO/DOX-loaded vesicles; (b) TEM image of the SPIO/DOX-loaded polymer vesicles; (c) T2 relaxation rates (1/T2, s− 1) as functions of iron concentration (mM) for both FA-conjugated SPIO/DOX loaded vesicles and Feridex. Reprinted with permission from Ref. [54]. Copyright 2010, American Chemical Society. |
After the pioneering work of Iler in 1966, [55] a layerby- layer (LbL) technique based on electrostatic attraction between alternating polyelectrolyte layers of opposite charge was employed by Decher to coat charged surfaces.[56, 57] Polymeric multilayer capsules, developed by LbL coating of a sacrificial template followed by dissolution of the template, have demonstrated their unique advantages and capabilities in storage, protection, release, and delivery of different functional agents.[58] These ordered micro/nano-structures are also promising candidates as imaging contrast agents for diagnostic and theranostic applications.
In order to prepare LbL polyelectrolyte capsule-carried SPIO particles, charged SPIO nanoparticles were directly self-assembled with polyelectrolyte as one component of the capsule membranes, resulting in capsules with wall structures of (nanoparticles/polyelectrolytes)n.[59] SPIO nanoparticles with uniform size distribution, good crystallinity, and charged surface must be prepared first in this process. As demonstrated by many reports, water soluble SPIO nanoparticles with either positive or negative charges can be prepared by modifications of charged molecules, such as N-alkyl- PEI, (3-carboxypropyl)trimethylammonium chloride, [60] 2- carboxyethyl phosphonate, [60] and tetramethylammonium 11- aminoundecanoate.[22] In fact, PEI is one of the most popular polyelectrolytes used in the LbL self-assembly process, and amphiphilic N-alkyl-PEI stabilized SPIO nanoparticles have shown good stability and magnetic properties.[13, 19] A recent study by our group found that amphiphilic N-alkyl- PEI2K wrapped SPIO nanocomposites are suitable candidates for further LbL self-assembly. These N-alkyl-PEI2K/SPIO nanoparticles show a strong positive charge with zeta-potential of more than 40 mV, and can be adsorbed onto negatively charged SiO2 nanotemplates, resulting in LbL particle/polymer structures.[17] These SPIO/polymer capsules show greater T2 relaxivity than single SPIO nanoparticles. In addition, SiO2 nanotemplates covered with a higher density of SPIO nanoparticles (Fig. 3(a)) displayed a 70% increase in T2 relaxivity compared with the lower density ones (Fig. 3(b)) and an increase of about 250% compared with single N-alkyl- PEI2k/SPIO nanocomposites.
 | Fig. 3. N-alkyl-PEI2k/SPIO nanocomposites adsorbed on polyelectrolyte covered SiO2 nanotemplates with (a) higher and (b) lower anchoring density. (Scale bar = 100 nm). Reprinted with permission from Ref. [17]. Copyright 2011, Elsevier Ltd. |
Among those medical imaging techniques mentioned before, no single modality is perfect and sufficient to provide physicians with all the necessary information. It is thus natural to develop multimodality imaging by combining two or more imaging techniques together in a platform.[5, 61– 63] For example, a combination of MRI with fluorescence imaging (FLI) allies the high sensitivity of fluorescence techniques to the high spatial resolution of MRI. Self-assembly of SPIO NPs in nanocarriers makes it convenient to prepare MRI/FLI hybrid nanocomposites by two processes:[64] 1) simultaneous encapsulation of fluorescent nanoparticles; [65, 66] 2) conjugation of fluorescent organic dye onto the nanocarriers.[67– 69] Multimodality imaging such as MRI/optical, MRI/PET has already demonstrated their promising prospects in tumor detection or therapy.
In addition to their functions as imaging contrast media, SPIO nanoparticles were designed to be used as a multifunctional platform of diagnostic probes or imaging visible drug delivery systems. Surface encapsulation, especially with polymeric nanostructure carriers, can be used for simultaneous loading of imaging probes and therapeutic agents, resulting in theranostic nanoplatforms.[10, 70] For diagnostic and drug delivery applications, in vivo bio-safety and circulation time are two important performance dimensions that we need to address. In order to improve biocompatibility, SPIO nanoparticles were coated with gold, silica or biocompatible polymers such as PEG or dextran. PEG is a widely accepted biocompatible polymer in the pharmaceutical industry for drug formulation, PEGylated nanocomposites have shown prolonged circulation time with improved pharmacokinetics.[71, 72] The capability to deliver the diagnostic and therapeutic nanoparticles to the site of interest with high accuracy is another important aspect of performance. Surface modification of superparamagnetic contrast agents with various targeting ligands, including monoclonal antibodies, peptides, and aptamers, has been reported.
In vivo cell tracking is a crucial tool in the developing and evaluating cell-based therapies such as cell-based repair, replacement and treatment. Also, it is important to develop methods that can monitor the location, fate and distribution of administered cells non-invasively. Among the available imaging-based cell tracking tools, magnetic resonance imaging (MRI) is advantageous due to its high spatial and temporal resolution, noninvasiveness, and deep penetration. Superparamagnetic iron oxide (SPIO) nanoparticles have been widely used as imaging probes for cell labeling to provide possibilities of future monitoring of cell migration and proliferation by use of MRI.[73– 75] Cell-based therapy involving stem cells and immunocytes (including macrophages, T-cells and dendritic cells) has recently attracted many researchers’ attention, and the success of such cellular therapy depends on accurate delivery of cells to the target organ.
Stem cells, with extensive renewal capacity and abilities to generate daughter cells that undergo further differentiation, have shown enormous potential for the treatment of a wide variety of degenerative, malignant, and genetic diseases.[76– 78] However, it is difficult for practitioners to establish whether stem cells have survived following transplantation in the body and whether they reach their target site or migrate elsewhere. In order to track stem cells in vivo using MRI, superparamagnetic iron oxide nanoparticles are used to label them before they are administered into the body. Stem cells labeled with SPIO nanoparticles can induce changes in the T2 relaxation rate of adjacent tissues, resulting in darkening of T2-weighted images compared to control images. Taking mesenchymal stem cells (MSCs) as an example, we labeled them with alkyl- PEI/SPIO nanocomposites and assessed their MRI visibility in vivo in a previous report (Fig. 4).[13] After labeling, MSCs are unaffected in their viability, proliferation and differentiation capacity. Labeled MSCs dispersed in a collagen type I hydrogel and injected subcutaneously showed strong image contrast against unlabeled cells under a clinical 3 T magnetic resonance imaging (MRI) scanner.
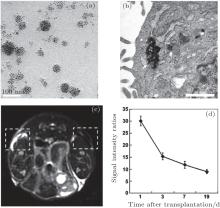 | Fig. 4. (a) TEM image of Alkyl-PEI/SPIO nanocomposites (polymer/SPIO mass ratio = 0.6); (b) electron microscopic image of cells labeled with SPIO probes (arrows), scale bar = 100 nm; (c) T2-weighted gradient echo image showing a prominent hypointense area of labeled injection in the right frame (19 d after transplantation); (d) ratios of signal intensities of the control and the labeled injection. Reprinted with permission from Ref. [13]. Copyright 2011, Elsevier Ltd. |
Dendritic cells (DCs) are highly specialized immune cells with antigen-presenting function that can induce primary immune responses and are important for the regulation of T cellmediated immune response.[79, 80] Although the understanding of DC immunobiology is still under study, DC-based immunotherapy protocols are now exploited to elicit immunity against cancer and infectious diseases. Similarly, monitoring DC traffic in tissues and then to lymph nodes is a crucial aspect of the success of DC-based immunotherapy. Radioactive labels of DCs with 111indium-oxinate have been reported, [81, 82] however, the short half-life of the radionuclide and lack of information on the exact location of the injected DCs limit their application in DC tracking. DC tracking based on MRI represents an attractive alternative because of these advantages of MRI. DCs labeled with SPIO and in vivo MR-based tracking are currently being explored.[12, 36, 83– 86]
Cardiovascular disease (CVD) is the leading cause of death worldwide, especially in many high-income countries. At the same time, cardiovascular deaths and disease have increased at a fast rate in low- and middle-income countries[87, 88] Pathophysiology studies show that atherosclerosis is the major precursor of CVD. Atherosclerosis is a chronic inflammatory response in the walls of arteries, caused largely by the accumulation of macrophage white blood cells and promoted by low-density lipoproteins (LDL)[89, 90] and involved with several highly inter-related processes including lipid disturbances, platelet activation, thrombosis, endothelial dysfunction, inflammation, oxidative stress, and so on.[91] In order to moderate the death rate of CVD and monitor the efficacy of atherosclerosis treatments, imaging strategies are used to detect the volume and constituents of plaques at early stages of atherosclerosis.[92]
Of all noninvasive imaging techniques, MRI shows the most promise for imaging the artery lumen and providing detailed information about the artery wall. An inflammation process developing concurrently with the accumulation of oxidized LDL in the arterial wall leads to accumulation of inflammatory macrophages within the plaque site.[89] Passive targeted uptake of SPIO nanoparticles by macrophages might be used to detect early atherosclerotic changes in MR images by means of an SPIO-related T2 shortening effect.[93, 94] Schmitz and Debatin did pioneering work on MR imaging of atherosclerotic plaque using ultrasmall SPIOs (USPIOs) in hyperlipidemic rabbits.[95, 96] Introduction of USPIOs phagocytized by macrophages in atherosclerotic plaques of the aortic wall was demonstrated to be sufficient to cause susceptibility effects detectable by MRI.
Thrombosis is another important component of atherosclerosis, often occurring at sites of plaque rupture or erosion.[91] Conjugation of platelets with fibrin forms a blood clot and leads to thrombosis; therefore, fibrin plays an important role in the progression of CVD and is likely to be a target for cardiovascular molecular MR imaging. Recently, the development and characterization of fibrin-targeted MRI contrast agents with high avidity raises the potential for sensitively detecting active vulnerable plaques.[97, 98] However, today’ s fibrin-targeted MRI contrast agents are based mainly on paramagnetic small-molecule agents; SPIO-based fibrintargeted MRI contrast agents have rarely been reported.
Cancer is now a major public health problem and one of the leading causes of death worldwide. Detection and diagnosis of early-stage tumors are the primary factors for a patient’ s survival of many cancers, but this work remains a major challenge. MRI is advantageous over other imaging models for early detection of tumors, due to the high spatial resolution and lack of radiation. MRI contrast agents, mostly SPIO nanoparticles, can be detected by MRI at very low concentration; they are nonspecifically taken up by the reticuloendothelial system (RES) and have been used for liver and spleen imaging. These non-targeting SPIO nanoparticles can also accumulate at the tumor site through an enhanced permeability and retention (EPR) effect and macrophage uptake.[99, 100] Because of the limitations of passive targeting, there is growing interest in exploring tumor-targeting SPIO nanoparticles with surfaces modified with targeting groups such as antibodies or aptamers. Both passive and active tumor-targeting have been widely studied with regard to early diagnosis and cancer therapy.
The EPR effect of solid tumors is one of the few tumorspecific characteristics that are useful for imaging probes and drug delivery for tumor imaging and therapy. The concept of the EPR effect was first reported by Matsumura and Maeda, [99] and EPR was further demonstrated to be capable of a key role in tumor-targeted drug delivery.[101– 103] Increased endothelial leakage and reductions in lymphatic drainage in the tumor site lead to accumulations of macromolecules or nanoparticles. Kataoka et al. recently reported a type of SPIO-loaded Cy5-cross-linked polyion complex vesicles (SPIO-Cy5-PICsomes) for tumor detection with high sensitivity.[104] They prepared an MRI contrast agent by loading FDA-approved SPIO nanoparticles into nano-scaled vesicles with diameters of about 100 nm. In vitro T2 relaxivity measurements indicate that these nano-PICsomes have a relatively high value of 663 mM− 1· s− 1. MRI signal alteration of the tumor region was observed in T2-weighted images of a small and early-stage tumor before injection and 24 h afterward, which indicates that SPIO-Cy5-PICsomes accumulated in the tumor site due to the EPR effect. These SPIO-loaded PICsomes were demonstrated to be valuable in detection of small metastatic tumors, and this may contribute to earlier identification for treatment in clinical practice.
It is well known that over-expressions, deletions or mutations of membrane proteins can be observed in practically all cancer cells. Identification of such targets can specifically cause therapeutics or imaging probes to persist in the vicinity of cancer cells or tissues, increasing the rate of internalization into cells by receptor-mediated endocytosis.[105, 106] For example, the integrin α vβ 3 highly expressed in many cancer cells plays a critical role in regulating tumor growth and metastasis as well as tumor angiogenesis.[107, 108] Arg-Gly-Asp (RGD)- containing peptides can strongly bind with the α vβ 3 receptor; thus, a variety of RGD peptide conjugated probes have been developed for tumor-targeted imaging. Zhang et al. developed c(RGDyK) peptide conjugated Fe3O4 nanoparticles and demonstrated their in vivo orthotopic glioblastoma targeting capability.[109] In this study, c(RGDyK)-Fe3O4 nanoparticles with a diameter around 45 nm were obtained by coupling RGD peptides to IONPs coated with a crosslinked PEGylated amphiphilic tri-block copolymer. IONP-RGD, IONPs and IONP-RGD+ block were intravenously administrated and 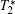
Molecular imaging now plays a key role in the clinical diagnosis and treatment of many diseases. However, among all the molecular imaging techniques, no single modality is perfect and sufficient to gain all the necessary information. The idea of multimodality imaging has already attracted many researchers’ interest.[5, 62, 63] MRI is among the best noninvasive methodologies today in clinical medicine for assessing the anatomy and function of tissues. But it is much less sensitive than nuclear techniques or optical imaging in monitoring small tissues lesions or molecular/cellular activities. Combining MRI with other imaging techniques can overcome this drawback and offer synergistic advantages over any single modality. Multimode imaging, such as MRI/optical and MRI/PET, has already demonstrated promise in tumor detection or therapy.
In near-infrared (NIR) fluorescence imaging, light penetrates deeper into living tissues because the absorbance spectra for all biomolecules have minima in the NIR region (700 nm– 900 nm).[110– 112] Therefore, NIR fluorescence imaging is very sensitive and offers a unique advantage for imaging pathophysiological changes at cellular and molecular levels, which is most suitable for rapid and cost-effective pre-clinical evaluation in in vivo imaging of small-animal models. Combining MRI and NIR imaging modalities can provide useful information with high spatial resolution and good sensitivity; realization requires dual modality probes. For instance, we conjugated NIR fluorescent dye Cy5.5 with an amphiphilic polyethylenimine (PEI), and used the product to encapsulate SPIO nanoparticle clusters.[67] The MRI/NIR dual-modality probe can label MCF-7/Adr cells with high efficiency. Accumulation of alkyl-PEI25-Cy5.5/SPIO nanocomposites in the cytoplasm results in a depressed MR signal and strong NIR fluorescence of these labeled cells in vitro. Similarly, Leary et al. prepared dual-modality imaging probes by loading SPIOs into self-assembled glycol chitosan nanoparticles labeled with Cy5.5.[113]In vitro and in vivo experiments have demonstrated the potential of these nanoparticles as NIRF and MR imaging agents for cancer diagnosis.
Another widely studied dual-modality imaging technique is the combination of positron emission tomography (PET) and MRI. PET is another system that has opened new horizons for multimodality molecular imaging by offering simultaneous morphologic, functional, and molecular information about a living system.[114, 115] Combined PET/MRI systems were first reported by Shao et al. in the late 1990s.[116– 118] Shortly after that, PET/MRI dual-modality imaging was reported to have been used in oncology of small-animal tumors. Nowadays, integrated PET/MRI scanners are commercially available for clinical use, and the search for multimodality agents has drawn a lot of attention. Chen et al. reported a trimodality probe combining SPIOs, 64Cu-DOTA and Cy5.5 in a single nanoplatform coated by human serum albumin (HSA) matrices.[119]In vivo PET, NIR, MRI imaging and ex vivo histological examinations confirm that the particles have a good retention rate and a high extravasation rate at the tumor site.
As nanotechnology develops, nanoscopic platforms that combine therapeutic agents and capabilities for diagnostic imaging and molecular targeting are emerging as the next generation of multifunctional nanomedicine to improve the therapeutic outcome of drug therapy.[70, 120– 122] The idea of timely monitoring of the medical treatment process led to the concept of theranostics.[123, 124] In order to monitor drug delivery and drug efficacy in vivo, many different types of nanoplatforms co-loaded with drugs, imaging agents and targeting ligands have been developed. Because of their excellent biosafety, well developed surface chemistry and prominent T2 relaxation time, SPIO nanoparticles have been used widely as nanoplatforms for theranostics.[125– 127] Much recent research on the imaging of drug delivery has focused on the development of nanomedicine formulations that can prolong circulation time and improve target-site accumulation. Some time ago, we developed a multifunctional magnetic nanoparticle with cancer-targeting capability for controlled drug delivery and MRI contrast enhancement.[10] These nanoscale micelles self-assembled from PEG-PLA block copolymers were used to encapsulate doxorubicin (DOX) and SPIO nanoparticles simultaneously, and they were labeled with a cRGD ligand that can target α vβ 3 integrins (Fig. 5). Similar polymeric nanostructures based on amphiphilic dextran were also developed as carriers of chemotherapeutic agents and MR imaging probes.[20]
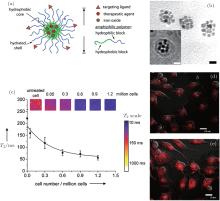 | Fig. 5. (a) Schematic diagram of Doxorubicin (Dox) and Fe3O4 nanoparticle-loaded PEG-PLA micelles, with cRGD peptides conjugated on the micelle surface. (b) TEM image of cRGD-DOXO-SPIO-loaded polymeric micelles (scale bars: 20 nm). (c) T2 values of SLK cells treated with 16% cRGD-DOXO-SPIO micelles as a function of cell number. (d) and (e) Confocal laser scanning microscopic images of SLK cells treated with 0% and 16% cRGD-DOXO-SPIO micelles. Reprinted with permission from Ref. [10]. Copyright 2006, American Chemical Society. |
Since most biomedical applications require delivery of a large number of MNPs into cells or the human body, much attention has been paid to MNPs’ toxicological properties and long-term impact on human health. So in vitro and in vivo behaviors and toxicology of iron oxide nanoparticles have been studied intensively. Many studies have demonstrated that at doses of 100 μ g/mL or higher, SPIONs with various physicochemical characteristics all exhibited low toxicity and cytotoxicity. Generally, oxidative stress induced by SPIO NPs is regarded as the primary cause of observed intracellular and in vivo toxicities.[128– 130] As is well known, excessive reactive oxygen species (ROSs), including free radicals— such as the superoxide anion, hydroxyl radicals, and the non-radical hydrogen peroxide — can be generated from the leaching of iron ions from the degraded surface of SPIO NPs with a large surface area. Subsequently, the ROSs are transferred to the interior of the cell, where they can produce oxidative stress by activating transcription factors for pro-inflammatory mediators— very high levels of oxidative stress are associated with activation of apoptotic pathways.[131]
The toxicity of magnetic iron oxide nanoparticles is reportedly influenced dramatically by their physicochemical parameters, such as particle size, shape, surface charge, coating materials.[130, 132] Since the interactions between SPIO NPs and biological organisms typically take place at the surface of the nanoparticles, proper surface functionalization is an effective method to manipulate their toxicity for bio-applications. With continuous efforts by multidisciplinary teams, there is great potential for further breakthrough developments in the design of iron oxide nanoparticles for biomedical and other applications.
Magnetic nanoparticles possess unique magnetic properties along with the ability to serve as MRI contrast agents, making them an attractive platform for cell tracking, tumor diagnosis and drug delivery at the molecular and cellular level. Enabled by recent advances in nanotechnology, magnetic nanoparticles with controllable size, morphology, composition, magnetization, relaxivity, and surface chemistry have been reported. In particular, nano-sized iron oxide particles show an excellent superparamagnetic property and have been used as strong enhancers of proton relaxation with superior MR T2 (transverse relaxation) shortening effects.
As a widely used MRI contrast agent, SPIO nanoparticles have the advantages of good biocompatibility, high T2 relaxivity, and low working concentration. Use of an SPIO-based contrast agent in an MR scan can improve the image contrast and delineate small lesions that may be difficult to discover in regular scans. In order to explore SPIOs with improved MRI sensitivity and better biocompatibility, doped iron oxide nanocrystals (e.g. MnFe2O4, CoFe2O4, NiFe2O4) were developed and conjugated with a number of biocompatible polymers (e.g. PEG, dextran) and clusters of SPIOs encapsulated in nanoparticles assembled from polymers by different means, such as hydrophobic encapsulation and electrostatic adsorption. Along with the advances of surface engineering chemistry for SPIO nanoparticles, much research about MRI contrast media recently focuses on specific targeting capability, which is commonly achieved by conjugating small biomolecules, peptides or aptamers that have high affinity to the target cells. Conjugation of these targeting ligands onto the surface of SPIO nanoparticles emerges as a promising means to increase the accumulation and retention of SPIOs in the tumor site. Another field now attracting many researchers’ attention is multifunctional SPIO nanoparticles combining two or more imaging modalities with a capability of drug delivery or cancer therapy in a single platform.
Although the possible physiological metabolism of iron released from SPIO degradation is still under investigation, SPIO-based MRI contrast agents have already been used widely in clinical MR scans for diagnosis and treatment of many diseases. The search for multifunctional ultrasensitive SPIO-based MRI contrast agents has drawn a lot of attention during the last decade.
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
| 10 |
|
| 11 |
|
| 12 |
|
| 13 |
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
|
| 18 |
|
| 19 |
|
| 20 |
|
| 21 |
|
| 22 |
|
| 23 |
|
| 24 |
|
| 25 |
|
| 26 |
|
| 27 |
|
| 28 |
|
| 29 |
|
| 30 |
|
| 31 |
|
| 32 |
|
| 33 |
|
| 34 |
|
| 35 |
|
| 36 |
|
| 37 |
|
| 38 |
|
| 39 |
|
| 40 |
|
| 41 |
|
| 42 |
|
| 43 |
|
| 44 |
|
| 45 |
|
| 46 |
|
| 47 |
|
| 48 |
|
| 49 |
|
| 50 |
|
| 51 |
|
| 52 |
|
| 53 |
|
| 54 |
|
| 55 |
|
| 56 |
|
| 57 |
|
| 58 |
|
| 59 |
|
| 60 |
|
| 61 |
|
| 62 |
|
| 63 |
|
| 64 |
|
| 65 |
|
| 66 |
|
| 67 |
|
| 68 |
|
| 69 |
|
| 70 |
|
| 71 |
|
| 72 |
|
| 73 |
|
| 74 |
|
| 75 |
|
| 76 |
|
| 77 |
|
| 78 |
|
| 79 |
|
| 80 |
|
| 81 |
|
| 82 |
|
| 83 |
|
| 84 |
|
| 85 |
|
| 86 |
|
| 87 |
|
| 88 |
|
| 89 |
|
| 90 |
|
| 91 |
|
| 92 |
|
| 93 |
|
| 94 |
|
| 95 |
|
| 96 |
|
| 97 |
|
| 98 |
|
| 99 |
|
| 100 |
|
| 101 |
|
| 102 |
|
| 103 |
|
| 104 |
|
| 105 |
|
| 106 |
|
| 107 |
|
| 108 |
|
| 109 |
|
| 110 |
|
| 111 |
|
| 112 |
|
| 113 |
|
| 114 |
|
| 115 |
|
| 116 |
|
| 117 |
|
| 118 |
|
| 119 |
|
| 120 |
|
| 121 |
|
| 122 |
|
| 123 |
|
| 124 |
|
| 125 |
|
| 126 |
|
| 127 |
|
| 128 |
|
| 129 |
|
| 130 |
|
| 131 |
|
| 132 |
|


