† Corresponding author. E-mail:
The electrical transport properties and structures of Y2O3/ZrO2 solid solution have been studied under high pressure up to 23.2 GPa by means of in situ impedance spectroscopy and x-ray diffraction (XRD) measurements. In the impedance spectra, it can be found that the pressure-dependent resistance of Y2O3/ZrO2 presents two different change trends before and after 13.3 GPa, but the crystal symmetry still remains stable in the cubic structure revealed by the XRD measurement and Rietveld refinement. The pressure dependence of the lattice constant and unit cell volume shows that the Y2O3/ZrO2 solid solution undergoes an isostructural phase transition at 13.1 GPa, which is responsible for the abnormal change in resistance. By fitting the volume data with the Birch–Murnaghan equation of state, we found that the bulk modulus B0 of the Y2O3/ZrO2 solid solution increases by 131.9% from 125.2 GPa to 290.3 GPa due to the pressure-induced isostructural phase transition.
Zirconia (ZrO2) is involved in many applications, such as foundry ceramics, refractories, paint pigments, fuel cells, and solid electrolytes in oxygen sensors, due to its outstanding chemical and physical properties including high hardness, high thermal conductivity, and high ionic conductivity.[1–3] ZrO2 has abundant phases at high temperatures: the monoclinic phase can be thermodynamically stable from room temperature to about 1100 °C, and then crystallize into a tetragonal phase at 1100 °C and a cubic phase at 1900 °C.[4–6] To quench these high-temperature tetragonal and cubic phases to ambient conditions, cations with a valence lower than 4+, such as Y3+, La3+, Mg2+, and Ca2+ are doped, by which CeO2/ZrO2, CaO/ZrO2, MgO/ZrO2, and Y2O3/ZrO2 (cubic) solid solutions are formed.
Important technical applications can be found in dopants. For example, transient optical measurements of materials under impact compression usually require different types of optical windows.[7–9] Dolan et al.[8] reported a new optical window material based on dopant-stabilized cubic ZrO2). At present, doping Y2O3 to obtain cubic ZrO2) is a common method.[10] A low impact pressure experimental study showed that cubic ZrO2) can be used as an optical window material and is a preferred substitute for sapphire window materials.[8]
Recently, there has been growing interest in the connection between the electrical properties and microstructures of dopant-stabilized ZrO2.[11–14] It is found that Sc2O3-stabilized ZrO2 solid solution with the cubic fluorite structure shows the highest conductivity among all ZrO2)-based electrolytes.[15,16] This provides potential to lower the operating temperature of the ZrO2)-based electrochemical devices, and makes the synthesis of the cubic structure ZrO2 an attractive goal. With the co-precipitate method, Y2O3-stabilized ZrO2 powders in the cubic structure have been directly synthesized, and the particle size is uniform.[17,18]
As another state parameter that is independent of temperature and chemical composition, pressure tunes the physical properties of materials by compressing the distance between atoms and modifying the electronic structures.[19,20] The phase transitions of ZrO2 under high pressure have been studied systematically with various techniques. It undergoes the phase transition sequence of baddeleyite (MI, monoclinic, space group: 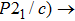
ZrO2/Y2O3 samples were prepared by the co-precipitate method. First, a weight of 2.58 g of zirconyl chloride (ZrOCl2


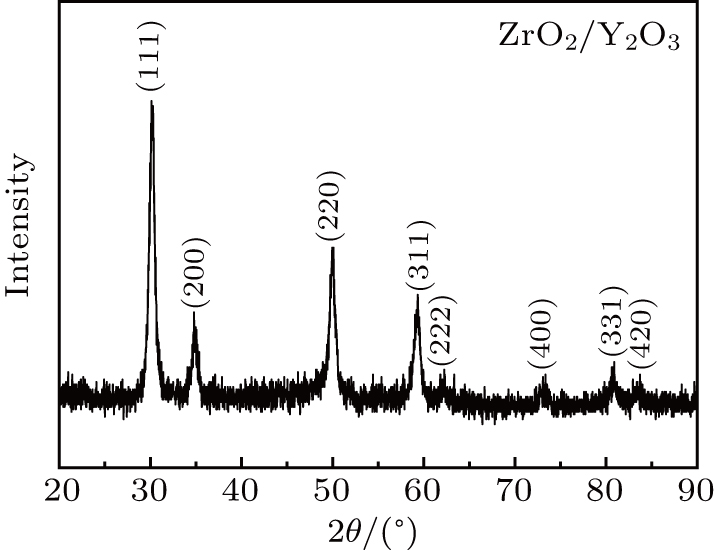
In order to identify the constitution and chemical valences, the samples were further analyzed by x-ray photoelectron spectroscopy (XPS). XPS was done on a PerkinElmer PHI 5000CESCA system to analyze the electronic states. All the binding energies were calibrated via the contaminant carbon (C 1s = 284.6 eV) as a reference. The initial structure of the sample was confirmed by x-ray diffraction (XRD) measurements.
High-pressure experiments were conducted in a diamond anvil cell with the anvil culet with a diameter of 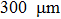
The impedance spectra were measured by a Solartron 1260 impedance analyzer equipped with a Solartron 1296 dielectric interface. An alternate-current sine signal with an amplitude of 1 V was sent to the sample, and its frequency ranged from 0.1 Hz to 107 Hz. For electrical measurements, the pressure was loaded up to 23.2 GPa.
The in situ high-pressure angle-dispersive XRD measurements were conducted at beamline 4W2 of the Beijing Synchrotron Radiation Facility.[28] The x-ray wavelength is 0.6199 Å. For better comparison with the measurements of the electrical transport properties, no pressure-transmitting medium was loaded. The distance between the sample and the detector, and the parameters of the detector, were calibrated with the CeO2 standard. Bragg’s diffraction images were integrated using Fit2d software, yielding one-dimensional intensity versus diffraction angle 2θ patterns. The Rietveld refinements of crystal structures were performed using the program Materials Studio.
The XPS spectra (Figs.
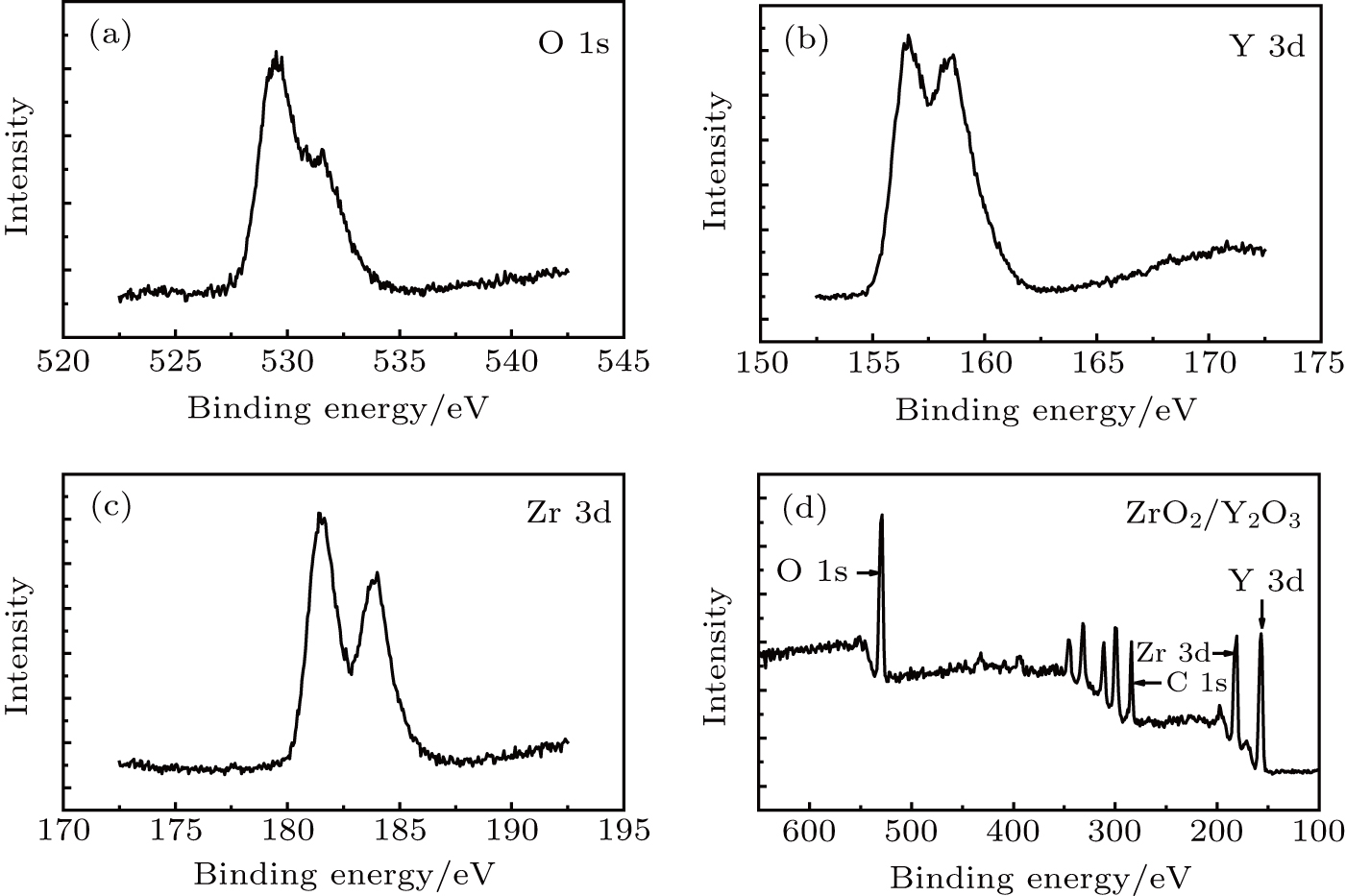 | Fig. 1. XPS spectra of the ZrO2/Y2O3: (a) O region; (b) Y region; (c) Zr region; and (d) a survey spectrum. |
| Table 1.
Parameters of Zr 3d, Y 3d, and O 1s in XPS spectra. BE stands for binding energy. . |
Figure 
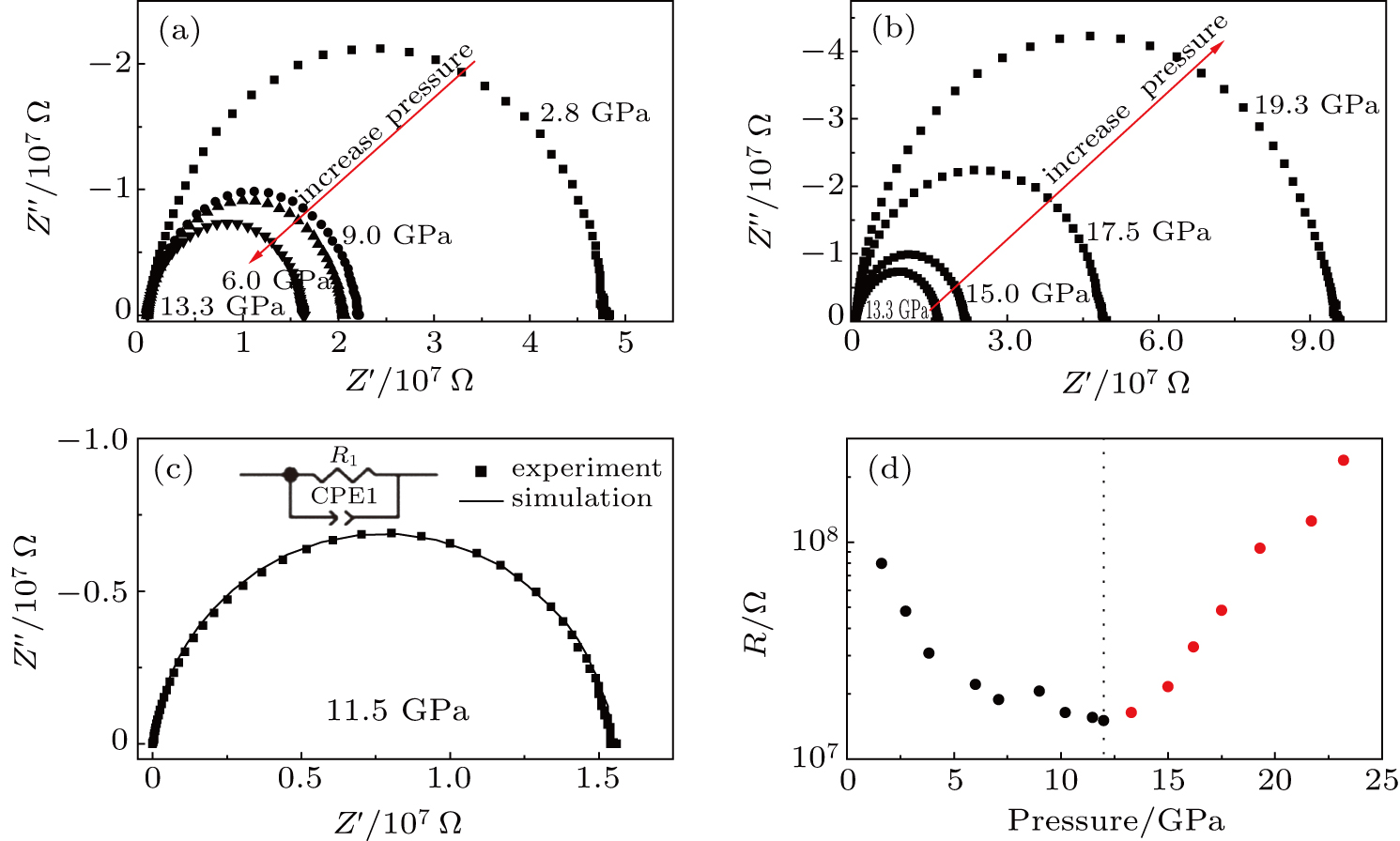
The impedance spectra of ZrO2/Y2O3 solid solutions under high pressures are shown in Figs.
Since the difference between the grain and grain boundary is barely distinguishable, the equivalent circuit model used for further quantitatively analyzing the impedance spectra can be simplified as shown in Fig.
Generally, the most possible reason for the abrupt change in resistance under high pressure is the pressure-induced structural phase transition or metallization.[32,33] However, the pressure-induced metallization should be excluded from our case because the resistance value still remains at a high level (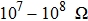
High-pressure XRD measurements are taken for ZrO2/Y2O3 solid solutions up to 25.7 GPa. Selected representative spectra are shown in Fig.
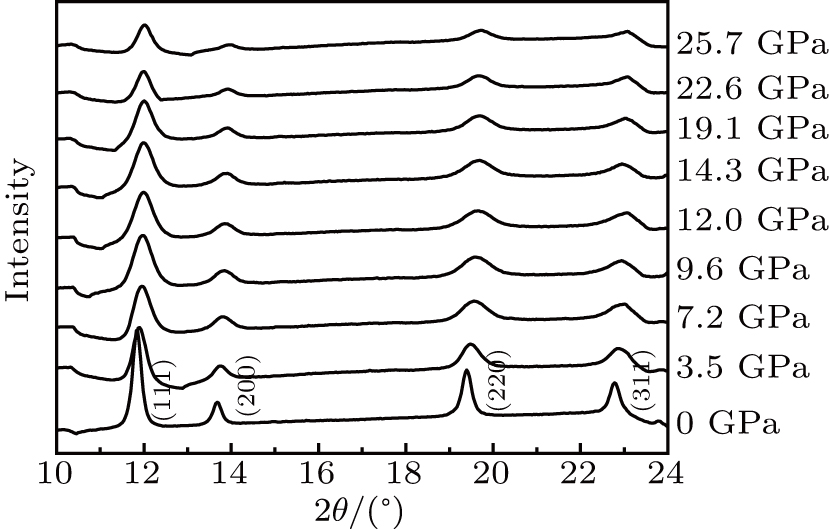 | Fig. 4. Representative angle dispersive XRD patterns of ZrO2/Y2O3 at various pressures, with an incident wavelength λ = 0.6199 Å. |
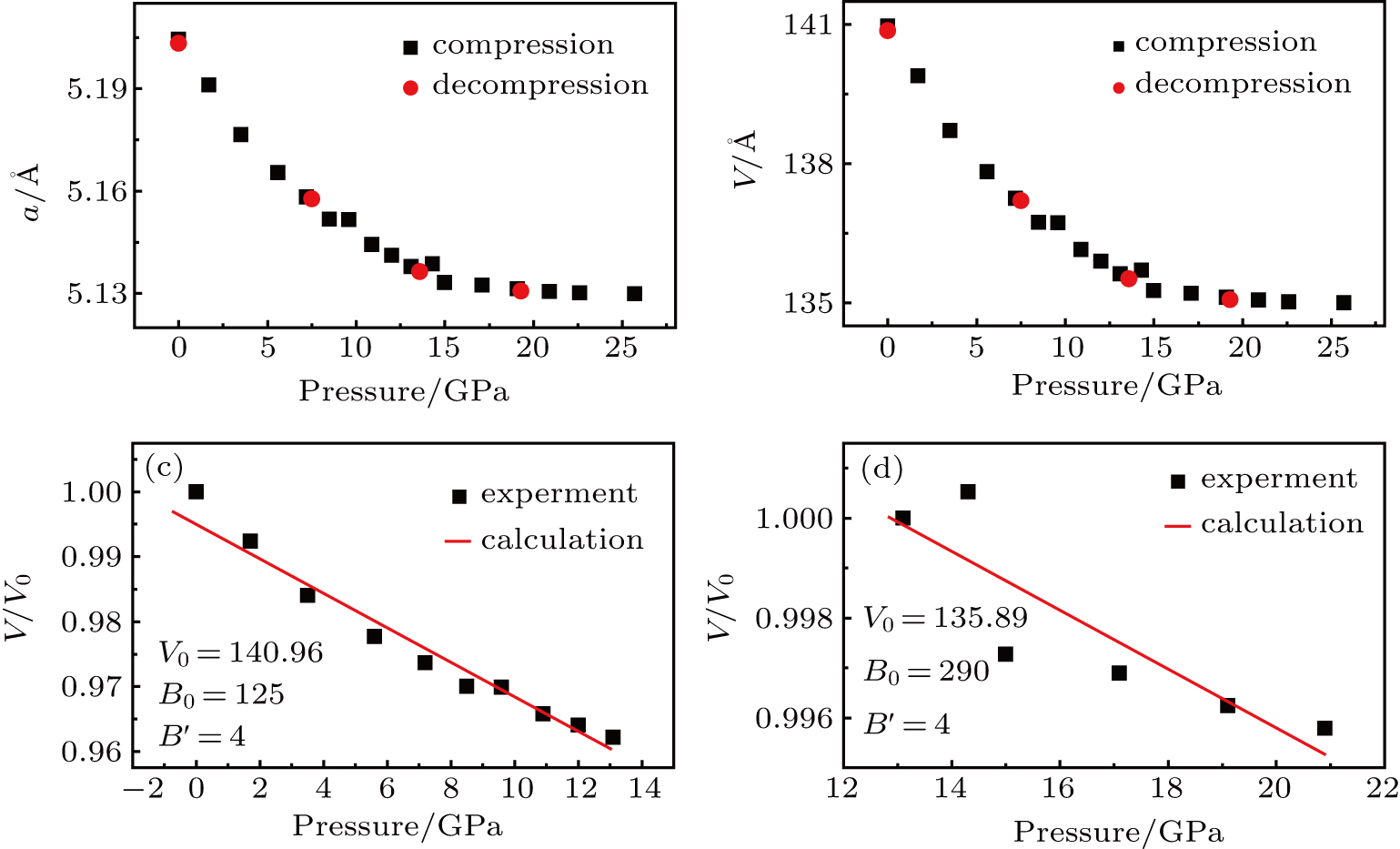
By using the Rietveld fitting method, we analyze the XRD spectra carefully, and obtain the pressure-dependent lattice parameters and the equation of state. Figures 
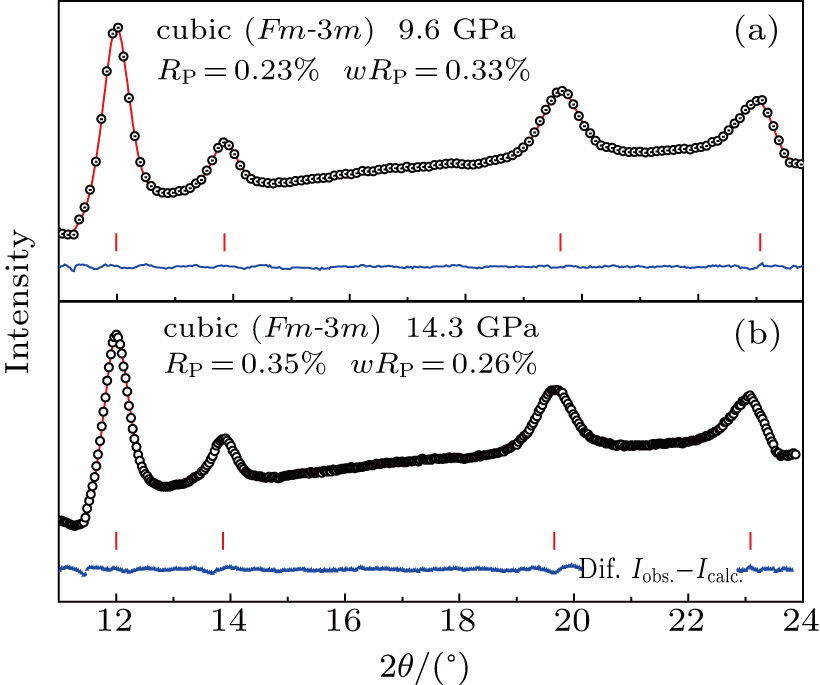 | Fig. 5. Representative Rietveld refinement results for XRD spectra of ZrO2/Y2O3 at (a) 9.6 GPa and (b) 14.3 GPa with an incident wavelength λ = 0.6199 Å. |
The pressure-dependent d values of crystal planes (111), (200), (220), and (311) are drawn in Fig.
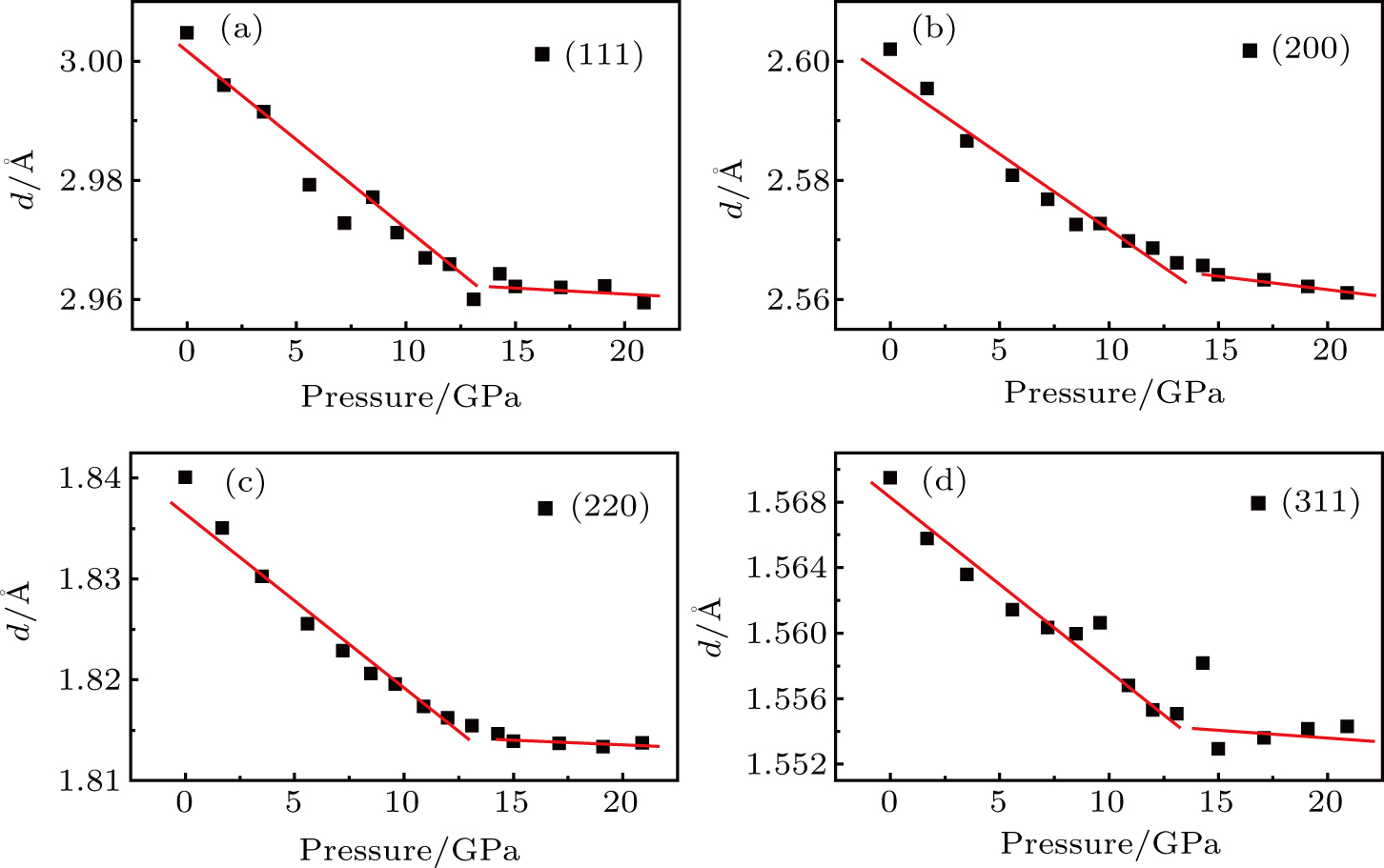 | Fig. 6. Pressure dependence of different interplanar spacings: (a) (111), (b) (200), (c) (220), and (d) (311). |
The pressure dependence of lattice constants and unit cell volume of ZrO2/Y2O3 can be obtained from the Rietveld fitting results, as shown in Figs.
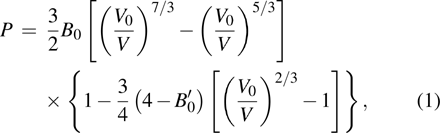 |
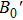
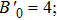
According to the method of least squares,[36] we calculate the standard error values of B0 in the two pressure stages. The results are shown in Table
| Table 2.
B0 in two different pressure ranges. . |
In summary, by combing in situ impedance and XRD measurements, we have studied the electrical transport and structural properties of the Y2O3-stabilized ZrO2 solid solution under a high pressure up to 23.2 GPa. The pressure dependence of resistance reveals one potential and unknown change in Y2O3/ZrO2 at 13.3 GPa, which had not been found before. The XRD measurement and Rietveld refinement further confirm that the abnormal change in resistance actually results from the pressure-induced isostructural phase transition in the Y2O3/ZrO2 solid solution. By fitting the volume data with the Birch–Murnaghan equation of state, it is also found that the bulk modulus B0 of the Y2O3/ZrO2 solid solution increases by 131.9% from 125.2 GPa to 290.3 GPa due to the isostructural phase transition. This study may help deepen our knowledge for future applications of ZrO2 solid solution-based devices under extreme conditions.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 | |
| 34 | |
| 35 | |
| 36 |