† Corresponding author. E-mail:
Project supported by the National Key Research and Development Program of China (Grant Nos. 2017YFA0303003 and 2016YFA0300300), the National Natural Science Foundation of China (Grant No. 11574370), and the Strategic Priority Research Program and Key Research Program of Frontier Sciences of the Chinese Academy of Sciences (Grant Nos. QYZDY-SSW-SLH001, QYZDY-SSW-SLH008, and XDB07020100).
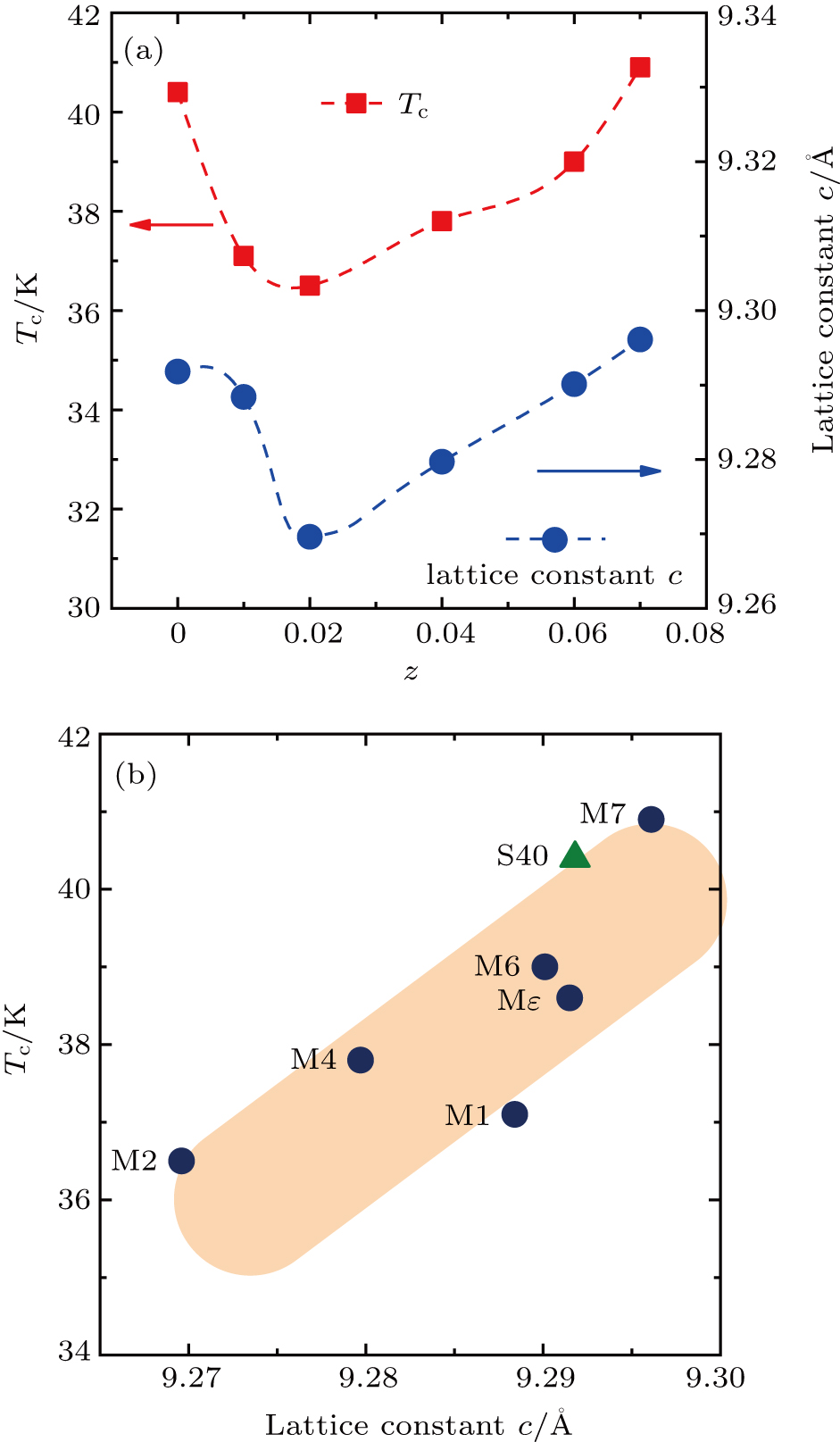
We synthesize a series of Mn substituted (Li, Fe)OHFeSe superconductor single crystals via a modified ion-exchange method, with the Mn concentration z (the atomic ratio of Mn:Se) ranging from 0 to 0.07. The distribution homogeneity of the Mn element incorporated into the lattice of (Li, Fe)OHFeSe is checked by combined measurements of high-angle-annular-dark-field (HAADF) imaging and electron energy-loss spectroscopy (EELS). Interestingly, we find that the superconducting transition temperature Tc and unit cell parameter c of the Mn-doped (Li, Fe)OHFeSe samples display similar V-shaped evolutions with the increasing dopant concentration z. We propose that, with increasing doping level, the Mn dopant first occupies the tetrahedral sites in the (Li, Fe)OH layers before starting to substitute the Fe element in the superconducting FeSe layers, which accounts for the V-shaped change in cell parameter c. The observed positive correlation between the Tc and lattice parameter c, regardless of the Mn doping level z, indicates that a larger interlayer separation, or a weaker interlayer coupling, is essential for the high-Tc superconductivity in (Li, Fe)OHFeSe. This agrees with our previous observations on powder, single crystal, and film samples of (Li, Fe)OHFeSe superconductors.
The recently discovered iron selenide (Li, Fe)OHFeSe (FeSe-11111) superconductor[1] has the layered structure formed by a stacking of one superconducting (SC) FeSe layer alternating with one (Li, Fe)OH layer along the c axis. In contrast to the prototypal bulk FeSe (Tc ∼ 9 K) and intercalated FeSe-122 superconductors like KyFe2−xSe2 (Tc ∼ 30 K), the (Li, Fe)OHFeSe intercalate is free from the complications of chemical phase separation and structural transition. Moreover, it shows a high Tc over 40 K (SC phase I), even above 50 K (SC phase II) under a 12.5 GPa pressure,[2] and a Fermi topology similar to the high-Tc (> 65 K) monolayer film of FeSe.[3,4] Therefore, the (Li, Fe)OHFeSe system turns out to be an ideal platform for studying the intrinsic electronic property and high-Tc superconductivity in the iron-based family. Most recently, we uncovered the occurrence of intrinsic electronic phase separation in (Li, Fe)OHFeSe,[5] similar to that in high-Tc cuprate and iron arsenide superconductors, and established a complete electronic phase diagram for (Li, Fe)OHFeSe. In an earlier work,[6] we successfully synthesized Mn substituted (Li, Fe)OHFeSe single crystals, with the doping level z (the atomic ratio of Mn:Se) ∼ 0.08 and 0.16. We found that, at this doping range, the superconducting transition temperature remains almost unchanged, with a Tc (∼ 40 K) close to the optimal one for the SC phase I. However, how the Mn substitution at lower concentrations would affect the superconductivity of (Li, Fe)OHFeSe is unclear so far.
In this work, we synthesize a series of lightly Mn-substituted (Li, Fe)OHFeSe single crystals via a modified ion-exchange method, with the doping level z ranging from 0 to 0.07. The homogeneity of Mn distribution in the lattice of (Li, Fe)OHFeSe is checked by combined experiments of high-angle-annular-dark-field (HAADF) atomic lattice imaging and electron energy-loss spectroscopy (EELS). Interestingly, we find that the SC transition temperature Tc and unit cell parameter c of the Mn-doped (Li, Fe)OHFeSe samples show parallel V-shaped evolutions with the increasing dopant concentration z. It appears that, with increasing doping level, the Mn dopant first occupies the tetrahedral sites in the (Li, Fe)OH layers before starting to substitute the Fe element in the FeSe layers, leading to the observed V-shaped change in the cell parameter c. A common positive correlation between the SC transition temperature Tc and lattice parameter c is also observed among the present Mn-doped (Li, Fe)OHFeSe samples, regardless of the Mn substitution concentration z. Such a positive correlation has been reported previously for powder,[7] single crystal,[5,8] and thin film[9,10] samples of Mn-free (Li, Fe)OHFeSe. Therefore, our results indicate that a larger interlayer separation, or a weaker interlayer coupling, is essential for the high-Tc superconductivity in (Li, Fe)OHFeSe. Actually, our previous study[8] has shown that high-Tc (Li, Fe)OHFeSe exhibits a strong electronic two-dimensionality.
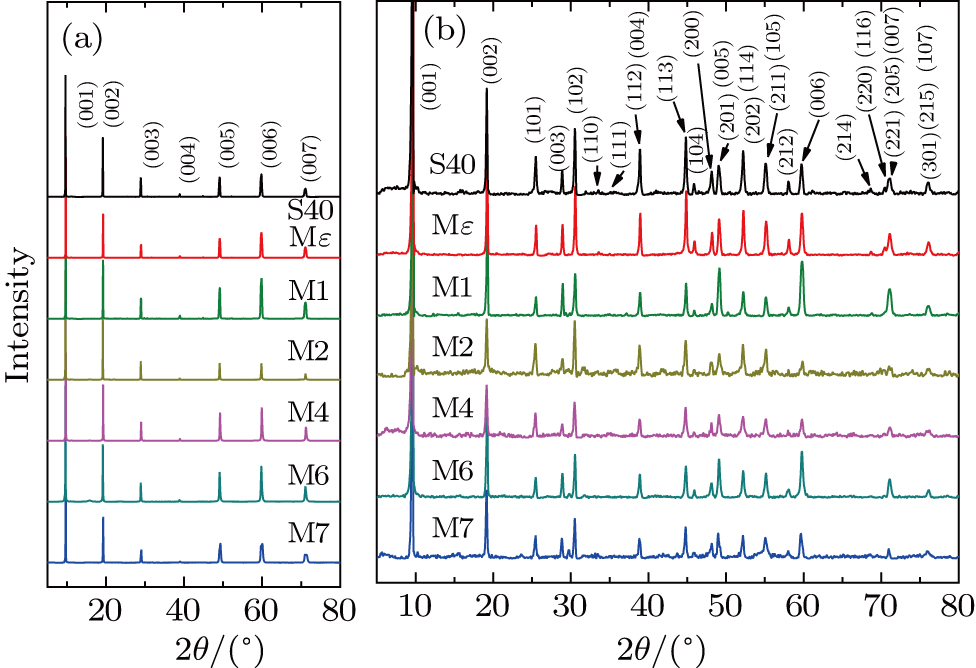
The x-ray diffraction (XRD) measurements were performed at room temperature on an 18 kW MXP18 A-HF diffractometer with Cu-Kα radiation, using a 2θ range from 5° to 80° and a 2θ scanning step of 0.01° (single crystal) or 0.02° (powder). The average chemical stoichiometry of Fe, Se, and Mn for the single crystal samples was determined by inductively coupled plasma atomic emission spectrometry (ICP-AES). The in-plane electrical resistivity was measured on a Quantum Design PPMS-9. The dc magnetic measurements were carried out on a SQUID magnetometer (Quantum Design MPMS XL-1). The EELS technique combined with a transmission electron microscope (TEM) was used for probing the local composition and spectroscopic information of the specimens. A JEM ARM200F 80–200 TEM (JEOL, Inc.) operated at 200 kV was used for EELS and atom-column-resolved scanning transmission electron microscopy (STEM) HAADF imaging. The microscope is equipped with both probe and image aberration correctors (CEOS GmbH). The electron energy-loss (EEL) spectra were acquired in STEM mode using a ∼ 1 nm electron probe with an energy resolution of ∼ 0.6 eV or better, as evaluated from the full-width at half-maximum of the zero-loss peak. The 1 nm probe size in STEM mode was intentionally broadened to minimize the electron beam damage on the sample, with parameters for high EELS signal and energy resolution rather than maximum spatial resolution. The spectra were recorded using a dispersion of 0.3 eV/channel and acquisition time of 0.5 s for each spectrum image to minimize electron irradiation damage. The convergence and collection semi-angles were estimated to be 20 mrad and 26.5 mrad, respectively.
The Mn-doped and Mn-free (Li, Fe)OHFeSe single crystals were synthesized via a modified ion-exchange method that we developed and first reported elsewhere.[6,8] The raw materials were 0.003 mol selenourea (Alfa Aesar, 99% purity), 4 g LiOH·H2O (Alfa Aesar, 98% purity), 0.00375 mol mixture of Fe (Alfa Aesar, 99.998% purity) and Mn (Alfa Aesar, 99.95% purity) powders. All the starting materials were mixed with 5 mL de-ionized water and a piece of K2Fe4Se5 single crystal, before being loaded into an autoclave. The autoclave was heated at 120 °C for 72 h and cooled to 50 °C at a rate of 0.5 °C/h. The as-grown Mn-doped and Mn-free (Li, Fe)OHFeSe single crystals were washed by de-ionized water for several times.
Figure
| Table 1. The superconducting transition temperature, unit cell parameters, and chemical composition for Mn-free and Mn-doped (Li, Fe)OHFeSe samples. . |
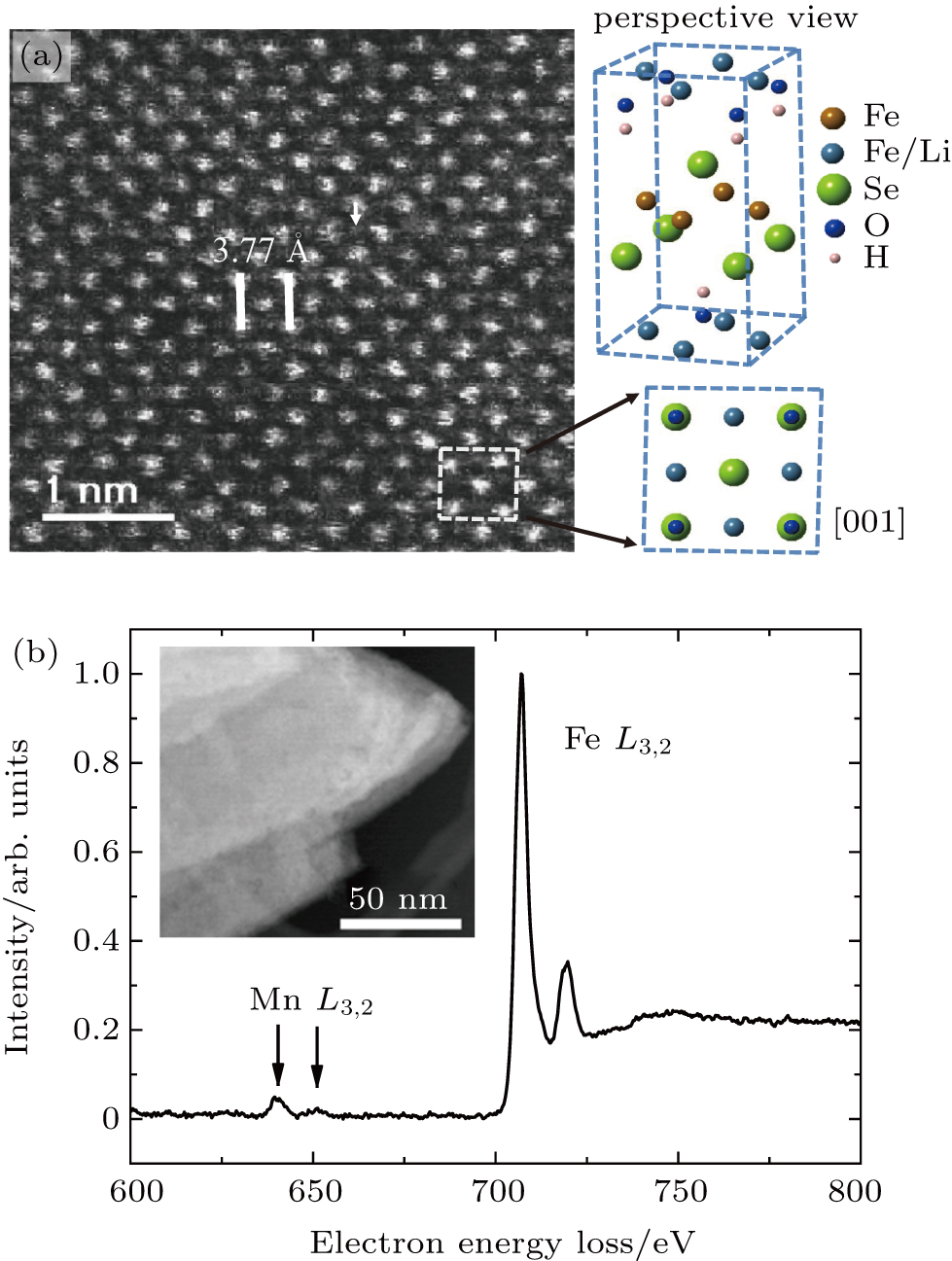
The homogeneity of Mn distribution in the structure of (Li, Fe)OHFeSe is checked by HAADF atomic lattice imaging. In the HAADF imaging, the recorded contrast arises primarily from Rutherford and thermal diffuse scattering. Under our experimental conditions, the HAADF intensity scales to a good approximation with the atomic number Z1.7 and the specimen thickness, as the selected area of interest is sufficiently thin. Figure
The EEL spectra were simultaneously recorded with relative low-magnification HAADF images in the STEM mode. The characteristic energy of core loss edges in EELS, resulting from inner-shell ionization, provides direct spectroscopic information about the samples. The usefulness of STEM–EELS techniques has been demonstrated in the previous work on iron arsenide superconductors. In the present study, we combine STEM and EELS to probe the local chemical information of the Mn/Fe sites, especially the substitution homogeneity. Figure
Both the HAADF atomic lattice imaging and STEM–EELS measurements of the Mn-doped (Li, Fe)OHFeSe samples indicate that the employed ion-exchange hydrothermal syntheses have successfully incorporated the Mn element into the (Li, Fe)OHFeSe lattice. However, quantification of the doped Mn and locally pinpointing the lattice sites at which the Mn dopant locates (in the FeSe or (Li, Fe)OH layers) remain unsolved in the present experiments, in part due to the difficulties in obtaining thin specimens oriented along the [100]-type zone-axis. Future studies may address the exact nature of Mn substitution and provide microstructural and compositional correlations with the superconductivity in Mn-doped (Li, Fe)OHFeSe.
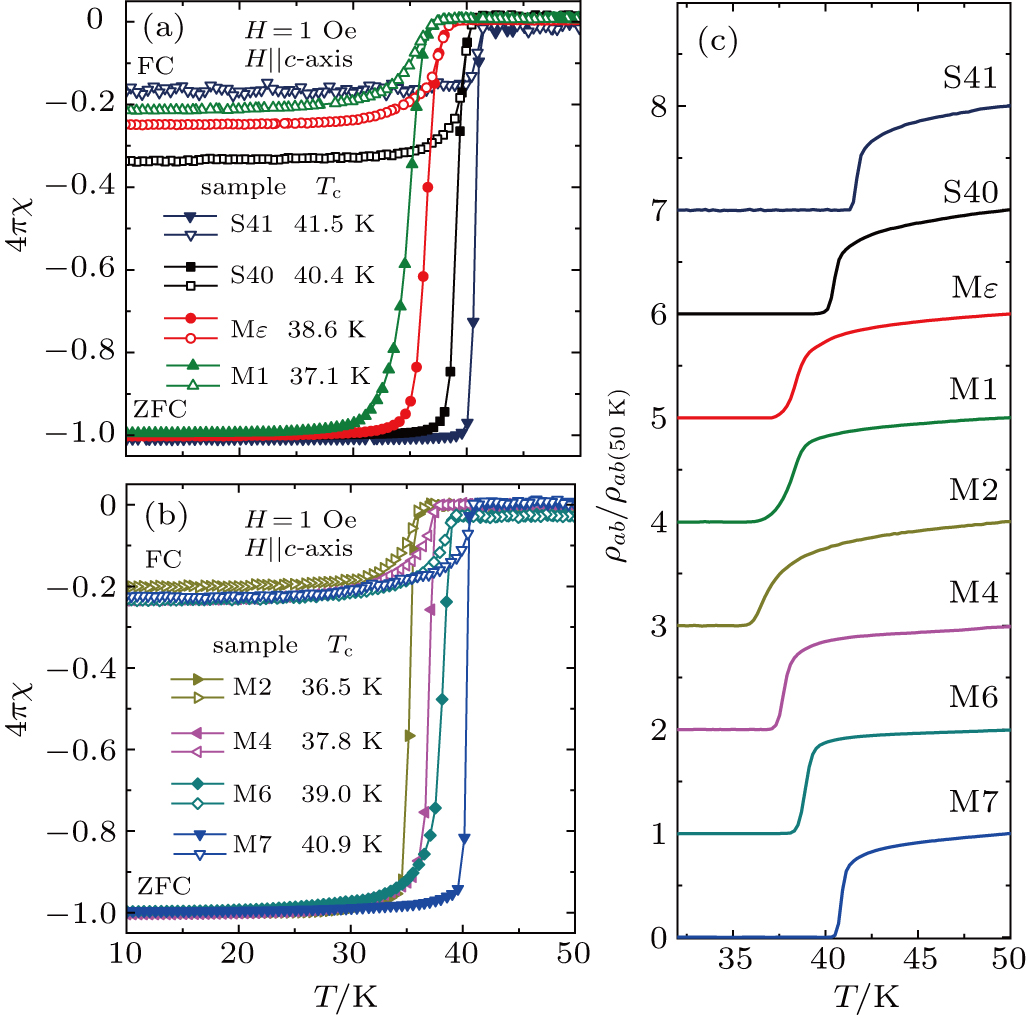
The superconductivity of the single crystal samples is characterized by magnetic susceptibility (Figs.
In Fig.
The modified hydrothermal ion-exchange method manifests itself as an effective technique to dope the Mn element into the layered structure of (Li, Fe)OHFeSe superconductors. The Mn dopant may be accommodated in the tetrahedral sites in the (Li, Fe)OH and FeSe layers depending on the doping level, reducing or enhancing the interlayer separation of (Li, Fe)OHFeSe. A common positive correlation between the superconducting transition temperature Tc and lattice parameter c is also observed among the present Mn-doped (Li, Fe)OHFeSe samples, regardless of the Mn doping level. It is indicated that a larger interlayer separation, or a weaker interlayer coupling, is essential for the high-Tc superconductivity in (Li, Fe)OHFeSe. This is in agreement with the strong electronic two-dimensionality reported previously for the high-Tc (Li, Fe)OHFeSe superconductor.[8]
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] |