† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11475197, 11575205, 11404100, and 11304083) and the Key Scientific and Technological Project of Henan Province, China (Grant No. 102102210186).
The CS/PVA/Fe3O4 nanocomposite membranes with chainlike arrangement of Fe3O4 nanoparticles are prepared by a magnetic-field-assisted solution casting method. The aim of this work is to investigate the relationship between the microstructure of the magnetic anisotropic CS/PVA/Fe3O4 membrane and the evolved macroscopic physicochemical property. With the same doping content, the relative crystallinity of CS/PVA/Fe3O4-M is lower than that of CS/PVA/Fe3O4. The Fourier transform infrared spectroscopy (FT-TR) measurements indicate that there is no chemical bonding between polymer molecule and Fe3O4 nanoparticle. The Fe3O4 nanoparticles in CS/PVA/Fe3O4 and CS/PVA/Fe3O4-M are wrapped by the chains of CS/PVA, which is also confirmed by scanning electron microscopy (SEM) and x-ray diffraction (XRD) analysis. The saturation magnetization value of CS/PVA/Fe3O4-M obviously increases compared with that of non-magnetic aligned membrane, meanwhile the transmittance decreases in the UV-visible region. The o-Ps lifetime distribution provides information about the free-volume nanoholes present in the amorphous region. It is suggested that the microstructure of CS/PVA/Fe3O4 membrane can be modified in its curing process under a magnetic field, which could affect the magnetic properties and the transmittance of nanocomposite membrane. In brief, a full understanding of the relationship between the microstructure and the macroscopic property of CS/PVA/Fe3O4 nanocomposite plays a vital role in exploring and designing the novel multifunctional materials.
Polymer matrix nanocomposites, which exhibit distinct physicochemical characteristics by incorporating inorganic fillers into polymer networks, have received much attention due to their various industrial applications in drug delivery, water treatment, food industry, aeronautical and aerospace structures.[1–6] Recently, magnetic responsive polymer composite membranes have attracted tremendous interest in the fields of gas separation, ultrafiltration and biomedical, mainly because of their effective transport structures.[7–11] The Fe3O4 serving as effective magnetically sensitive filler has been widely investigated to fabricate polymer–inorganic hybrid materials due to its biocompatibility and chemical stability over physiological circumstances in addition to its natural magnetic response.[12–15] Chitosan (CS) is the most important derivative of chitin, and chitin is the second most important natural polymer in the world.[16] The CS, used in food, cosmetics, biomedical applications, has been widely investigated due to its outstanding biocompatibility, and appropriate biodegradability.[17] The CS-Fe3O4 membrane fabricated by combining infiltration and magnetic alignment was found to have a higher flux than the corresponding non-magnetized membrane in pervaporation for ethanol dehydration.[18] The addition of Fe3O4 nanoparticles changed the packing of CS molecular chains, increased the free-volume cavities in the interface region and enhanced the permeability of hybrid membranes further. Magnetic aligned Fe3O4 nanoparticles-based composite materials with other polymers matrix have been reported in many papers.[14,18–21] Poly vinyl alcohol (PVA) has excellent solvent resistance, membrane-forming ability, biodegradable, biocompatible and nontoxic properties, which makes it a possible candidate material for use in biomedicine.[22] As a kind of biomedical material, PVA/CS is more favorable for the cell culture than the pure PVA, and CS/PVA membrane is a potential candidate material in tissue engineering applications due to the biocompatibility and mechanical properties.[23] The magnetic biodegradable Fe3O4/CS/PVA membrane has been considered to be one of the promising biomaterials for bone regeneration.[24] However, most researchers have devoted their efforts to exploring new methods with tuning the distribution of functional fillers or developing new materials to modify properties such as ultrafiltration, gas separation, magnetic, electrical, optical and mechanical properties,[25,26] only a few research studies concentrate on the role of sub-nanoscopic structure in the evolved properties. Sharma et al. investigated the role of the microscopic structure at interphase in a PVA-metal organic framework (MOF) nanocomposite in the evolved thermo–mechanical property.[27] Due to attractive interfacial interaction between the surface of zeolitic imidazolate framework (ZIF) particles and PVA chains, the microscopic structure in the interfacial region is changed, indicating the enhancement in rigidity of nanocomposites. Wang et al. further studied the function of microscopic structure transition in the effective carbon capture and in grapheme oxide membrane.[28] These studies have shown that it is essential to understand the complex relation between the microstructure and the macroscopic properties of nanocomposites, as a fundamental step to design novel materials further. Nevertheless, a full understanding of the relation between the microstructure and the evolved magnetic and optical properties of the magnetic anisotropic membranes, is still rarely reported.
Unlike conventional measurement techniques such as x-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR) and field emission scanning electron microscopy (SEM) which can provide nearly almost direct information about the microstructure, the position annihilation spectroscopy (PAS) has been used as a sensitive probe to investigate the microstructures of polymeric matrix nanocomposites, providing the information of molecular level packing about the complex bulk material structure.[29] Position annihilation lifetime spectroscopy (PALS) is one of the most common characterization techniques of PAS, which can provide the information about the free volume microvoid properties of the hybrid materials through their lifetime measurements. Recently, the PALS technique has been widely used as a probe to study the microstructures and the correlated positron annihilation characteristics in the fillers cross-linked polymer systems.[29–32] The induction of filler in the polymer matrix can change the original molecular chain packing and free volume nanohole properties. Generally, after positrons implanting into polymeric materials from a radioactive source, a large number of positrons are converted into positronium (Ps) rather than annihilating with electron directly. When positrons are located in the large holes of polymer matrix or in the interfacial region between polymer and functional fillers, the intrinsic lifetime of o-Ps is 142 ns. When located in the free volume of the polymer material, the positrons will annihilate through double gamma photons by picking up an opposite spin electron from the surrounding medium, which is called pick-off annihilation, shortening its lifetime to a few nanoseconds. In other words, o-Ps are located, formed and annihilated finally in the free volume. The lifetime and intensity of o-Ps represent the size and concentration of free volume in polymer, respectively.
In this paper, the magnetic and non-magnetic aligned CS/PVA/Fe3O4 nanocomposite membranes are prepared by a magnetic-field-assisted solution casting method. Through the controlled experiments, we mainly study the microstructure and the macroscopic properties of nanocomposites. The morphology of the membrane is characterized by using SEM. The crystalline structure and the chemical bonding changes of membranes are confirmed by XRD and FT-IR, respectively. To investigate the complex relationship between the microstructure and the optical and magnetic property of nanocomposites, PALS, UV-vis transmittance spectra and vibrating sample magnetometer (VSM) testing are carried out. The PALS offers the information about free volume properties, which is based on sub-nano level molecular packing of hybrid membranes. In addition, the effects of an externally applied magnetic field on optical and magnetic behaviors are also discussed.
Chitosan (CS) (degree of deacetylation = 98% − 99%, Mw = 1.2 × 105 g/mol, purity 98%) was supplied by Zhejiang Shaoxing Bio-technology. Poly (vinyl alcohol) (PVA) (purity 98%), ethylene glycol (purity 98%) and poly ethylene glycol, 2000 (PEG2000) (average Mn 2000) were purchased from Sinopharm Chemical Reagent Co., Ltd, China. Iron chloride hexahydrate (FeCl36H2O), sodium acetate anhydrous (NaAc) and glycerol (purity 99%) were purchased from Aladdin Co., Ltd, China. Reagents used in this study were all of analytical grade and directly used without any further purification.
Fe3O4 nanoparticles were synthesized in advance by a solvothermal reduction method according to Ref. [33]. The Fe3O4 nanoparticles were modified by polyethylene glycol 2000. For preparing CS/PVA/Fe3O4 membranes, 2-g CS powders were dissolved in 100-mL acetic acid (2 vol%), and PVA solution (5 g⋅d⋅L−1) was obtained by oil bath heating in deionized water at 90 °C for 2 h. Then, these two solutions were mixed at a volume ratio of CS : PVA = 40 : 60 to form homogeneous precursor solution. The Fe3O4 nanoparticles were mixed at 0, 0.5, and 1 wt% in the CS/PVA precursor solution (70 mL) by continuously stirring. Then 1-mL glutaraldehyde solution (0.25 wt%) and 1 mL glycerol were added into the resulting CS/PVA precursor solution in sequence. Finally, the mixture was poured into a glass petri dish and dried at 40 °C for 3–4 days. During the fabrication of magnetic membrane, two cuboid permanent magnets N35 (50 mm× 25 mm× 25 mm) were applied for 2 days. The samples were named CS/PVA/Fe3O4(x)−M(y) (where x represents the doped fraction of Fe3O4 nanoparticles; M denotes the magnetic field, and y refers to the magnetic induction intensity, y = 0.4 T in this paper).
The morphologies of CS/PVA/Fe3O4(x)−M(y) membranes were observed by FESEM (Hitachi S-4800). The phase compositions of membranes were determined by XRD (D8 Advance). The FTIR spectra were recorded by using a Thermo Scientific Nicolet IN10MX instrument with a liquid–nitrogen cooled detector in a range from 4000 cm−1 to 700 cm−1. The UV-vis transmittance spectra were obtained using a UV-vis spectrophotometer (Cary 5000 UV-vis-NIR, Australia). Magnetic properties were measured using a vibrating sample magnetometer (VSM) with a physical property measurement system (PPMS, Quantum Design).
The as-obtained samples were heated at 40 °C for 5 h–6 h prior to the measurements to ensure the absorbed water from the samples was removed entirely. The positron annihilation measurements were carried out at the room temperature in the air atmosphere by using an EG & G ORTEC fast–slow coincidence system with a time resolution of 210 ps. The positron source (22Na, 13 μCi) was fabricated with carrier-free NaCl deposited between two kapton foils each with a thickness of ~ 7 micrometer. The positron source was sandwiched between two stacks of samples with a size of 1 cm×1 cm to guarantee that all the positrons annihilated within the sample completely. Each positron annihilation lifetime spectroscopy (PALS) contained ~ 2.0 × 106 counts. A standard Si crystal was used as reference material to evaluate source correction (fraction of positrons annihilating from source material and kapton foils). All the lifetime spectra were analyzed by using LT-9 routine with three exponential components. The routine MELT was used to fit the PALS spectra to evaluate the continuous distribution function of annihilation rate from lifetime spectra. A semi-empirical equation
Figures
The XRD patterns of pure CS/PVA, CS/PVA/Fe3O4, and CS/PVA/Fe3O4-M are shown in Fig.
Figure
In order to study the effects of external magnetic field inducement on the physical and chemical properties of the nanocomposites, the optical transmittance measurement, sub-nanoscopic structural measurement of nanocomposites and magnetic testing are carried out. Figure
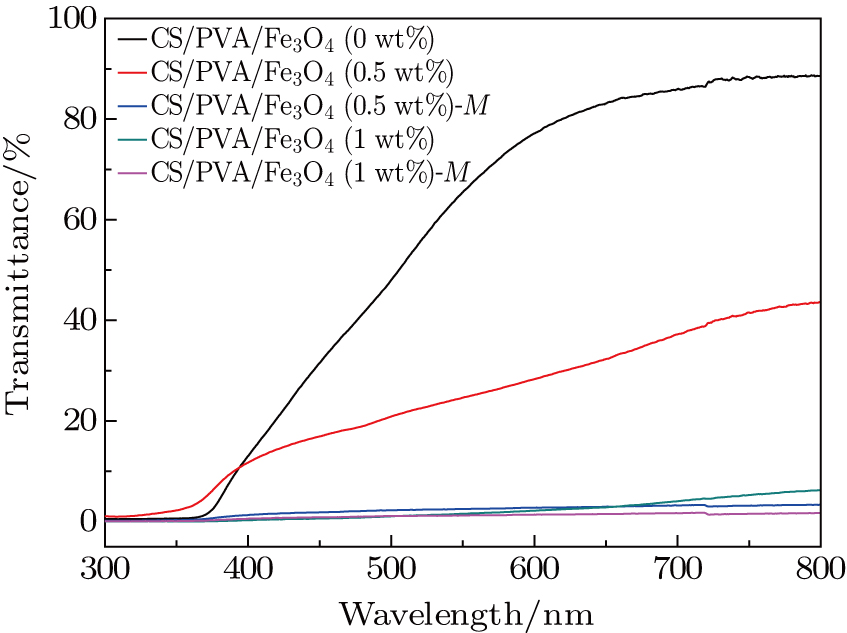 | Fig. 4. (color online) The UV-visible transmittance spectra of CS/PVA/Fe3O4(x) and CS/PVA/Fe3O4(x)-M (x = 0, 0.5, 1 wt%) nanocomposite membranes. |
To investigate the sub-nanoscopic molecular packing in CS/PVA/Fe3O4 hybrid membrane, the free volume properties of CS/PVA and CS/PVA/Fe3O4 nanocomposites are characterized using the positron annihilation technique. It is well known that the variations in free volume and hole properties are usually investigated by using an ortho-positronium (o-Ps) probe. Thus PALS spectra of CS/PVA and the nanocomposites are analyzed using LT-9 program and the results are reported in Table
| Table 1. The values of o-Ps lifetime τ3 and free volume fraction fvol of CS/PVA/Fe3O4(x) and CS/PVA/Fe3O4(x)-M (x = 0, 0.5, 1 wt%) nanocomposites’ membranes. . |
The continuous lifetime distributions of unfilled CS/PVA and CS/PVA-Fe3O4 nanocomposites are shown in Fig.
To further evaluate the magnetic properties of the CS/PVA-Fe3O4 nanocomposites, VSM measurements are carried out. Figure
In this paper, the magnetic ordered CS/PVA/Fe3O4 nanocomposite membranes are prepared by a magnetic-field-assisted solution casting process. Due to a co-operative effect between the hydrogen bonding interactions of the CS/PVA matrix and the external field induced magnetic moment interactions, the magnetization-induced loading Fe3O4 nanoparticles exhibit chain-like arrangement in the CS/PVA matrix as seen from the surface and cross section morphologies. On the incorporating of Fe3O4 nanoparticles, the original hydrogen bonded structure of semi-crystalline CS/PVA matrix is disrupted and no chemical bonding is formed between the polymer molecule and the filler, conferring these membranes with more compact chain packing and smaller fractional free volume, which are confirmed by PALS, XRD, and FTIR data. In addition, it is indicated that the introduction of external field results in forming an analogous optical barrier belt, which lowers the transmittance of nanocomposite membrane in the UV-visible region. Meanwhile, the VSM data indicate that the saturation magnetization value of CS/PVA/Fe3O4-M is obviously increased, which is mainly due to the enhanced magnetic vector ordering. It is suggested that the CS/PVA/Fe3O4 nanocomposite membranes of magnetic ordered chain-like structure promote the magnetic properties and optical properties. This study offers some valuable ideas to investigate the microstructure and macroscopic properties of magnetic responsive polymer nanocomposites, which have a great application prospect in the future.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] |






