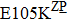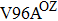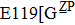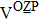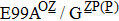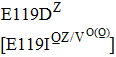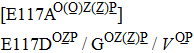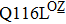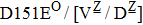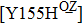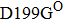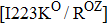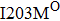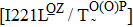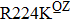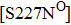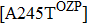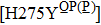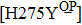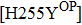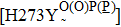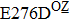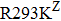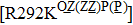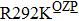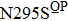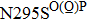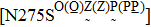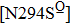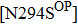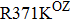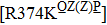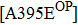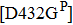† Corresponding author. E-mail:
Neuraminidase (NA), a major surface glycoprotein of influenza virus with well-defined active sites, is an ideal platform for the development of antiviral drugs. However, a growing number of NA mutations have drug resistance to today’s inhibitors. Numerous efforts are made to explore the resistance mechanisms through understanding the structural changes in mutated NA proteins and the associated different binding profiles of inhibitors, via x-ray, nuclear magnetic resonance, electron microscopy, and molecular dynamics methods. This review presents the architectural features of mutated NA proteins, as well as the respective inhibitor sensitivities arising from these spatial differences. Finally, we summarize the resistance mechanisms of today’s neuraminidase inhibitors and the outlook for the development of novel inhibitors.
Influenza is an acute respiratory infectious disease caused by influenza virus (A, B, and C types). A- and B-type influenza viruses give rise to human pandemics, while C type is less common.[1] Hemagglutinin (HA), neuraminidase (NA), and matrix protein 2 (M2) exist on the surface of virus particles and play an important role in virus proliferation.[1,2] Based on antigenic determinants, 18 HA subtypes and 11 NA subtypes have been identified from isolated influenza A strains.[3,4] The 11 NA subtypes could be further classified into three groups based on their sequences and structures.[5] As Fig.
 | Fig. 1. (color online) Comparison of the various NA subtype active sites. Structures of the active site in 09N1 (PDB ID 3NSS), VN04N1 (PDB ID 2HTY), N2 (PDB ID 1NN2), N3 (PDB ID 4HZV), N4 (PDB ID 2HTV), N5 (PDB ID 3SAL), A/chicken/Nanchang/7-010/2000 (H3N6) N6, A/mallard/ALB/196/1996 (H10N7) N7, N8 (PDB ID 2HT5), N9 (PDB ID 7NN9), N10 (PDB ID 4FVK), N11 (PDB ID 4K3Y), Flu B NA (B/Beijing/1/87; PDB ID 1NSB), and 09N1-Ile223Arg (PDB ID 4B7M) (N1 numbering).[3] |
Neuraminidase (NA) is generally considered to help virus release and replication through selectively breaking the a-glycosidic bond between sialic acid and glycoconjugate.[6,10] Therefore, inhibiting NA function by simply blocking its substrate-binding site can interfere with virus replication.[10–12] Currently, NA inhibitors (NAIs) are effective in therapy for influenza A and B virus infections, with the aid of their competing for the NA binding site with sialic acids (NA substrate). Among these, oseltamivir and zanamivir are the most widely authorized inhibitors around the world, while peramivir and laninamivir are approved in a few countries. However, the emergence of drug-resistant NA mutants (e.g., H274Y and N294S) has aroused great concern among developers of next-generation agents.[13,14] To our knowledge, these inhibitors were designed based on the highly conserved active sites of NA proteins.[14] Therefore, it is important for drug designers to consider mutations at the catalytic or framework residues that could alter the structures of NA active sites.[15] Besides, the resistance mechanisms of mutations have been studied by crystal structures and molecular dynamics (MD) simulations.[16–18] However, surveillance and cell-culture studies show that mutations away from the active site of B type NA (Flu B) also reduce susceptibility to current inhibitors. More specifically, these residues are located at the interface of monomers.[19] However, society needs to further explore the mechanisms of resistance.
As Table
Mutations in NA may alter the structure of the active site and then reduce the binding abilities of NA inhibitors. The effect of NA mutations will be reviewed in this article for the fact of clinical relevance.[25] According to the substitution table made by the WHO GISRS antiviral susceptibility expert working group (http://www.who.int/influenza/gisrs_laboratory/collaboratin), we summarized the resistant mutations in Table
| Table 2.
Neuraminidase amino acid substitutions associated with reduced inhibition by neuraminidase inhibitors (WHO). . |
Interestingly, Flu B viruses include some specific resistant mutations that were not observed in Flu A. This may be explained by the remarkable structural differences between Flu A and Flu B. As shown in Fig.
Oseltamivir-resistant viruses were rarely detected during the application of NAIs. During the first five years of NAI use (1999–2004), only 0.33% (8/2287) of isolates in adults (13 years) collected worldwide had a reduced inhibition in susceptibility to oseltamivir, while the rate of that in children (12 years) was 4.0%.[27–29] During the 2007–2008 influenza season, an unexpectedly high rate of virus resistance to oseltamivir was reported worldwide. During the subsequent season (2008–2009), a higher rate of resistance was reported from North America and Europe due to the H274Y mutation.[30] After the first wave of the 2009 pandemic, the level of oseltamivir resistance in H1N1pdm09 remained relatively low:[31] 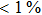
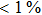
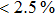
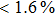
H275Y is a widely detected oseltamivir-resistant mutation in N1 (including H1N1pdm09, H1N1 and H5N1). H275Y variants in N1 subtype all showed highly reduced inhibition with the fold of 221–1637 in H1N1pdm09, 321–2597 in H1N1, 911–2502 in H5N1.[34–48] However, H275Y in group-2 viruses (N2, N9) and Flu B was rarely detected in oseltamivir-resistant isolates. Mutations at position 119 always conferred reduced susceptibility to NAIs. Among these, E119A (E99A in H5N1; E117A in B), E119V (E119V in N1 and H3N2; E117V in B), and E119I (E119I in H3N2) were detected to be oseltamivir-resistant. However, unlike H275Y, the level of reduced inhibition varies obviously among the subtypes of NA, and even different isolates of the same subtype show difference in inhibition level. For instance, E119V in H1N1 detected by Woo Young Choi[35] showed reduced inhibition with a fold of 7.4–23.4, while E119V in H1N1 detected by Yacine Abed[49] exhibited highly reduced inhibition (1727–2144 folds). This phenomenon may be explained by synergetic effects with other mutations. Although mutations at 119 confer highly reduced inhibition in subtype N1, all the isolates harboring these mutations are generated by reverse genetics or cell-culture studies.[35,39,49–51] These mutations may impair the fitness of the viruses to the degree that they have never been observed in the clinic.
Besides the mutations at position 275 and 119, the N294S is another common mutation with observably reduced susceptibility to oseltamivir. The oseltamivir-resistant variants conferred by N294S were shown to occur with or without NAI pressure in H5N1, H3N2, and Flu B. Among these, H3N2 isolates harboring N294S exhibited the most highly reduced sensitivity with a fold of 300–1879.[39,40,49,52–55] Note that the isolates of H1N1 and H1N1pdm09 with N294S substitution have not been found in the clinic.[35,49,50] The R292K substitution in catalytic site is another markedly resistant mutation in subtype N2. Notably, R292K in H3N2 increases inhibition more than 10000 fold.[47,49,55–60] Moreover, this substitution is also virulent in Flu B.[61] However, R292K in Flu B has never been detected in natural viruses.
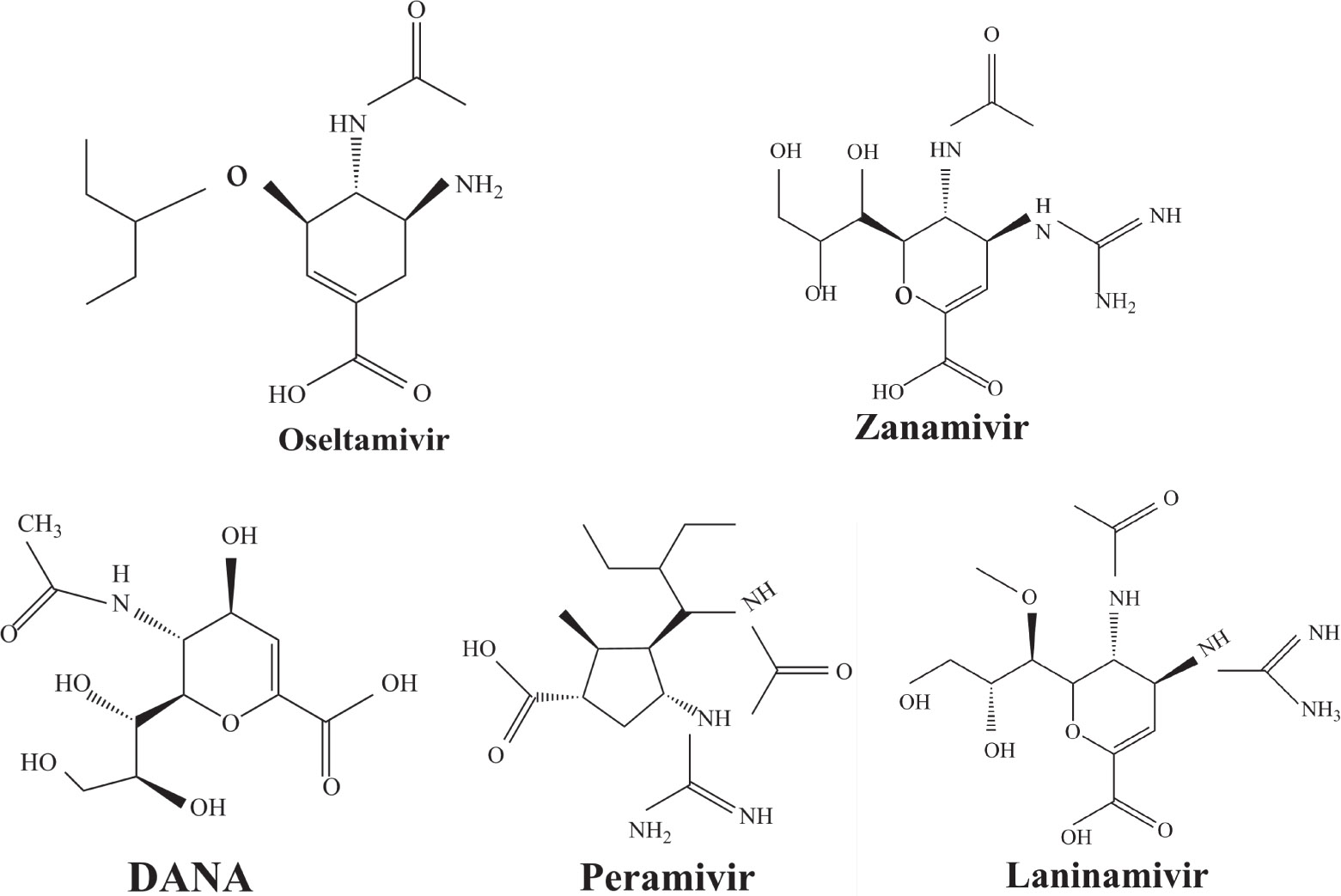
Some other mutations also show reduced sensitivity to oseltamivir. More oseltamivir-resistant mutations are detected in H5N1, H3N2, and B than in H1N1 (including H1N1pdm09). D199G and I223K/R in H1N1pdm09 all confer relatively mildly reduced inhibition,[50,62–65] while Y155H in H1N1 increases the inhibition to oseltamivir 123 fold.[28] None of the other oseltamivir-resistant mutations in H5N1 confers increased inhibition more than 100 fold, including V96A, Q116L, D179G, I203M, and S227N.[51,66–68] Among these mutations, V96A and S227N were detected by surveillance studies. The other oseltamivir-resistant mutations in H3N2 have not been observed in the clinic, including D151E, R224K, E276D, and R371K (H3N2).[60] Furthermore, only R224K confers an inhibition of more than 100 fold. Of note, mutation S227N occurs not only in H5N1, but in H1N1 (including H1N1pdm09) albeit with slightly reduced inhibition (4–8 fold). However, this mutation of H3N2 was not observed in the clinic, which suggests this mutation may impair N2 fitness. Flu B contains the most detected oseltamivir-resistant mutations. The observed mutations with highly reduced inhibition (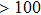
Zanamivir resistance has remained infrequent in contrast to oseltamivir. This may be explained by the high structural similarity of zanamivir with NA natural substrates. For example, mutation H275Y, which raised significant resistance to oseltamivir, was observed to retain normal inhibition to zanamivir in all subtypes. Similar situations also occur with N294S, except that one H5N1 variant with N275S mutation reduces sensitivity 27 fold.[35,39,40,49,50,52–55,64]
Substitutions at residue 119 confer remarkable resistance to zanamivir, and most of them are found by reverse-genetics or cell-culture studies. Notably, most of them increase inhibition more than 100 fold, like E119G/V (H1N1pdm09), E119V (H1N1), E99A/G (H5N1), and E117A/D/G (Flu B).[35,49,52] However, mutations at residue 119 in H3N2 were not notably resistant to zanamivir. More specifically, E119I increases inhibition by 17 fold, E119D by 32 fold, and E119V only by 7 fold, which cannot even be defined as reduced inhibition.[49,57,61,76] E117V in Flu B also confers normal inhibition.[61]
Zanamivir-resistant Q136K was identified by surveillance in Australia and Southeast Asia.[77,78] However, this mutation could not be observed when matching clinical specimens, which raised a debate of its clinical relevance.[78] Zanamivir-resistant mutation Q116L in H5N1 has not been observed in the clinic.[67] In contrast to H1N1 and H5N1, the Q136K mutation of H3N2 was detected in two clinical samples, both resistant to zanamivir.[45,79] Y155H is another zanamivir-resistant mutation of influenza H1N1 observed by surveillance.[28] Additionally, R293K in H1N1 generated by reverse genetics exhibited zanamivir resistance.[49,50] For H1N1pdm09, reduced sensitivity to zanamivir was identified in virus isolates with mutation I223R.[50,63] Furthermore, mutation V96A, which is specific to H5N1, shows reduced inhibition to zanamivir.[65,68] Though Q116L and D179G also show reduced susceptibility to zanamivir, unlike V96A they have never been observed in the clinic.[51,68]
Substitutions at 151 (D151V/D) of H3N2, which significantly increase IC 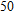
Observation of strains resistant to peramivir has usually followed an oseltamivir treatment. Notably, those resistant strains always show a higher level of resistance to oseltamivir but a lower level of resistance to peramivir, in accord with the structural homology between oseltamivir and peramivir.[33] Situations of resistance to peramivir in substitutions at residues 274 and 294 are quite the same as those of oseltamivir, except for the lower resistance level to peramivir. In fact, variants harboring mutation N294S show lower resistance to peramivir than oseltamivir in all subtypes. Viruses with N294S mutation in H3N2 are even not identified to be resistant to peramivir.[49,55]
However, some extraordinary situations still exist. For example, mutations at residue 119 seem to express a random inhibition compared to other mutations. While H5N1 and E99G reduce inhibition to peramivir and retain normal inhibition to oseltamivir, this is opposite to E99A.[39,51] Fewer mutations are resistant to peramivir in N1 and N2 in contrast to Flu B. Besides the above-mentioned mutations, mutation Q136K in H1N1 and R292K in H1N1, H3N2 and Flu B reduce sensitivity to peramivir instead of oseltamivir.[35,61,77] Like other NAIs, subtype B contains several specific resistant mutations, such as E105K, Q138R, P139S, G140R, R150K, D197E/Y, I221T, A245T, R374K, A395E, and D432G. Notably, all of these mutations were observed in the clinic or by surveillance.
Drug resistance occurs when mutations attenuate the binding to the inhibitors rather than to the substrate. In other words, resistance means shifting the balance toward substrate binding and away from inhibitor binding.[81] MD studies reveal that the weakening of Van der Waals (VDW) contacts to inhibitors compared to substrates may be a predominant factor that results in the resistance. Additionally, destroying or attenuating hydrogen bonds to inhibitors may be a factor contributing to drug resistance. This correlates to the fact that the overall NA substrates have fewer intermolecular hydrogen bonds than inhibitors in wild type NAs.[81]
As Fig.
MD studies show that active-site residues R118, D151, R152, E276, R292, and R371 directly interact with oseltamivir in the wild type and form a mixed polarity surface within the binding pocket (Fig.
 | Fig. 4. (color online) Interactions of inhibitors with individual active site residues (red: N1-oseltamivir complex; blue: N2-oseltamivir complex; pink: N1-zanamivir complex; yellow: N2-zanamivir complex).[81] |
Regarded as an N1 subtype, E119 has similar interaction with substrates and inhibitors. Hence, mutations at 119 generated by reverse genetics or cell cultures confer resistance to oseltamivir; they also inhibit the substrate binding within N1 subtype. In the case of wild type N2 strains, oseltamivir has more advantageous VDW contacts and hydrogen bonds than substrates. MD simulations reveal that the oseltamivir orientations in 
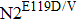
MD simulations reveal that the interaction of residue D198 with residue R152 is destroyed due to the R152K mutation. Furthermore, the mutation causes residue 152 to deviate from the active site center in both N2 and Flu B, and the H-bond interactions with the oseltamivir acetamide group also disappear.[12] According to the crystal structures (with and without inhibitors), E275 could rotate when binding to peramivir in both the D197 and E197 NAs. However, mutation D197E prevents the E275 from rotating upon binding to oseltamivir. Lack of the E275 rotation plays an important role in Flu B NAs resisting oseltamivir.[90]
I222 exhibits similar interactions with both the substrate and the oseltamivir in N2 subtype. Hence, mutation at I222 is likely to impact both the oseltamivir and substrate binding. However, in N1 subtype, I222 has more favorable interaction with the oseltamivir in contrast to substrates.[12] I222 is located near R224 and the pentyl ether group of oseltamivir (Fig.
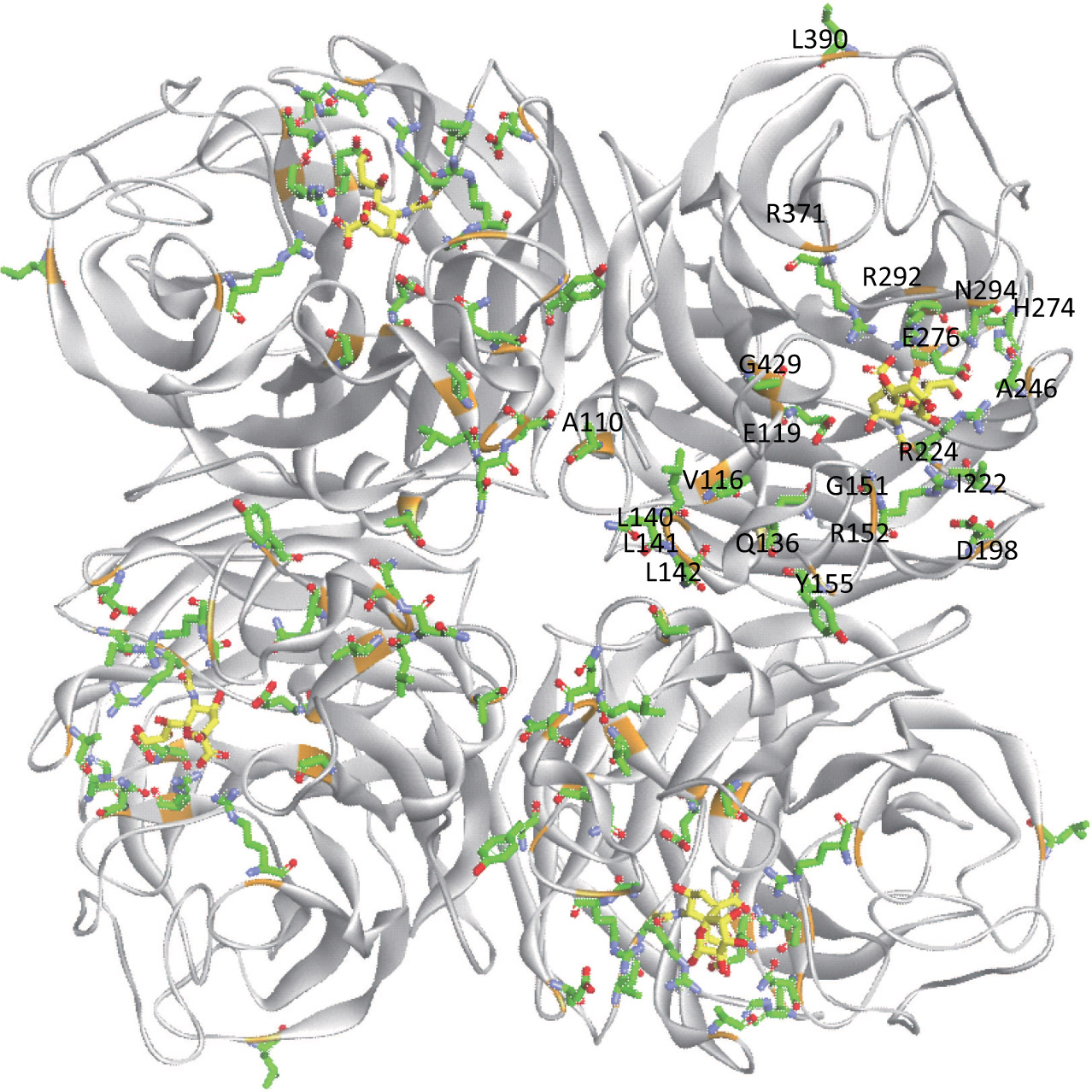 | Fig. 2. (color online) Mutation sites leading to drug resistance (WHO); yellow represents the substrate (PDB ID: 4GZX). |
According to the crystal structures of N1 subtype, residue H274 is not located at the active site. However, both mutations R292K and H274Y reduce inhibition activities. MD results show that these mutations can prevent the rotation of E276, leading to the E276 side chain moving further from the active site and losing interaction with oseltamivir.[14,81] Note that the NA structure of wild-type H5N1 does not show full rotation of E276, which further suggests that lack of the full E275 rotation may account for partial resistance to oseltamivir. Besides, MD studies show that the region around the mutated residue (H274Y) is more positively charged than the wild-type N1 structure, and residue E276 has more significant favorable VDW contacts with oseltamivir compared to substrates, which agrees with the mechanism revealed by wild-type N1 crystal structures.[81]
On the basis of N1N294S-oseltamivir complex, S294 forms a hydrogen bond with E276 and allows water molecules to penetrate deeply into the binding pocket. Additionally, the N294S binds to the oseltamivir side chain. Those water molecules destabilize and break the hydrogen bond between the E277 residue and the amide moiety of oseltamivir, while simultaneously helping to form a new hydrogen bond between E277 and R151.[86]
MD studies reveal that the dominant interactions between zanamivir and the wild-type NA are the bindings of the side chains (E119, D151, R152, W178, I222, E227, E276, and Y406), see Fig.
Residue E119, significantly resistant to zanamivir, has more favorable contacts with zanamivir, in contrast to oseltamivir and peramivir within the wild-type N1 subtype. Crystallographic analysis reveals that the E119G mutation breaks the interactions of the carboxylate side chain with the C4-guanidinium group of zanamivir.[14] MD studies show that the conformation of zanamivir shifts significantly away from the original state in the wild-type complex, which indirectly affects the binding of the surrounding residues. Moreover, zanamivir moves closer to the residues R224 and N294 along with an obvious rotation in the structure itself.[91] Mutation E119V can break the salt-bridge between residue 119 and the 4-guanidino group of zanamivir. However, the zanamivir 4-guanidino group still retains its typical interactions with E227, W178, and D151.
Similar to the case of oseltamivir, mutation H274Y may break zanamivir’s hydrogen bond to E276, forming a new hydrogen bond with R151. In addition, the electrostatic potential of the binding pocket is slightly more negative than in the wild-type complex. However, the region close to the hydroxyl groups of the zanamivir turns rather more positive. In N1 subtype, mutation N294S could break the interactions of zanamivir with R117, E276, and R371, while forming new hydrogen bonds between zanamivir and residues D150, R151, and S246. That is why mutation N294S is hardly detected as resistant to zanamivir.
In light of the current pandemic threat and the ongoing emergence of drug resistance, continual development of new-generation drugs must be a high priority. Apart from the above NA mutations of influenza virus, the accumulation of drug-resistant mutations within HIV-1 protease also shows that the most prominent mutations do not have extensive interactions with substrates, but instead are important for inhibitor binding, due to the differences in spatial structures of proteins.[92] These conformational alterations are involved with both active site residues (e.g. I50V, V82A, and I84V)[93,94] and framework residues (L90M).[95] These reports further support the assessment that mutation-inducing spatial differences seem to be one of the general principles for drug resistance and deserve more attention in structure-based design.[96]
The exploration of current NAIs benefits from sufficient knowledge of the NA active sites.[97–99] The presence of novel druggable hot spots in the 150- and 430-loop regions provides further support for developing high-affinity inhibitors capable of binding to these two areas.[100,101] Besides, current drugs are designed for one binding site only. However, multi-target treatment deserve more study, such as targeting an active site and a 150-loop region (and/430-loop region), simultaneously binding with two/three/four NA monomers, or even concurrently binding with NA and hemagglutinin. In addition, the study of NAIs via molecular mechanics might lead us to propose a mechanism for the enzymatic reaction and, ultimately, an effective reaction-analogue inhibitor that has the potential as a novel drug with a feasible cost of drug production, high absorption and high bioavailability.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [52] | |
| [53] | |
| [54] | |
| [55] | |
| [56] | |
| [57] | |
| [58] | |
| [59] | |
| [60] | |
| [61] | |
| [62] | |
| [63] | |
| [64] | |
| [65] | |
| [66] | |
| [67] | |
| [68] | |
| [69] | |
| [70] | |
| [71] | |
| [72] | |
| [73] | |
| [74] | |
| [75] | |
| [76] | |
| [77] | |
| [78] | |
| [79] | |
| [80] | |
| [81] | |
| [82] | |
| [83] | |
| [84] | |
| [85] | |
| [86] | |
| [87] | |
| [88] | |
| [89] | |
| [90] | |
| [91] | |
| [92] | |
| [93] | |
| [94] | |
| [95] | |
| [96] | |
| [97] | |
| [98] | |
| [99] | |
| [100] | |
| [101] |