† Corresponding author. E-mail:
The inelastic electron tunneling spectroscopy (IETS) of four edge-modified finite-size grapheme nanoribbon (GNR)-based molecular devices has been studied by using the density functional theory and Green‘s function method. The effects of atomic structures and connection types on inelastic transport properties of the junctions have been studied. The IETS is sensitive to the electrode connection types and modification types. Comparing with the pure hydrogen edge passivation systems, we conclude that the IETS for the lower energy region increases obviously when using donor–acceptor functional groups as the edge modification types of the central scattering area. When using donor–acceptor as the electrode connection groups, the intensity of IETS increases several orders of magnitude than that of the pure ones. The effects of temperature on the inelastic electron tunneling spectroscopy also have been discussed. The IETS curves show significant fine structures at lower temperatures. With the increasing of temperature, peak broadening covers many fine structures of the IETS curves. The changes of IETS in the low-frequency region are caused by the introduction of the donor–acceptor groups and the population distribution of thermal particles. The effect of Fermi distribution on the tunneling current is persistent.
With the miniaturization of materials and device size, carbon-based low-dimensional quantum structure has been more extensively studied both theoretically and experimentally.[1–6] The remarkable structural, electrical and chemical properties of graphene make it one of the important candidates of microelectronics and relative areas.[7–13] Nowadays, one can prepare, characterize and control extremely small scale systems, such as nano-scale molecular devices based on grapheme nanoribbon (GNR) slices.[14–18] The results of our previous work[19] show that the electronic and elastic transport properties of GNR slices are strongly dependent on edge modification types. When used as the molecular core of nano molecular devices, the edge type can tune the carrier distribution of the extended molecules. Furthermore, the connection types of the molecular core and electrodes will have a decisive impact on the contact conductance.[20–23] For small-sized materials and devices, it is difficult to measure and gain the configuration features of the device accurately in experiment. The inelastic electron tunneling spectroscopy (IETS) which is very sensitive to the configuration can give structural information about the molecular junction. Furthermore, the IETS curve can provide a direct access for molecular identification and chemical bonding investigations.[24–30] Thus, investigations of the effects of atomic structure and connection types on inelastic transport properties of edge-modified finite-size GNR-based molecular devices have significant technological implications. In this work, the IETS properties of several edge-modified GNR-based molecular devices have been studied and discussed systematically.
Here, four junctions based on finite-sized GNR slices with different edge functional types have been considered as the model systems. The construction process of model systems: based on the two-dimensional graphene structures, we cut to obtain an 8 × 8 rectangular graphene fragment with 8 carbon atoms on the zigzag (ZZ) edges and 8 carbon atoms on the armchair (AC) edges. This 8 × 8 GNR slice is the prototype structure of the molecular core. Then passivation and modification for the dangling bonds of edge carbon atoms have been done. Optimization and relaxation have been down for the molecular core systems. The extended molecules can be established by connecting the molecular core systems with triangle gold clusters. The introductions of a large number of metal atoms in the system often bring magnitude growth of the quantum chemistry calculations. Furthermore, the main and most important IETS information comes from the vibrations of the molecular atoms. Therefore, we choose triangle gold clusters composed of three gold atoms connected with the edge-terminated atoms of the molecular core as the contact geometry of the extended molecules. Two types of modification have been chosen: one is that all of the dangling edge carbon atoms on both ZZ and AC edges have been passivated by hydrogen atoms. The other is that donor–accepter functional groups NH2 and NO2 have been used to substitute the hydrogen atoms of the opposite side of ZZ edge carbon atoms respectively, that is, donor–accepter modifications have been used for the ZZ edges and hydrogen passivation has been used for the AC edges. Then the models have been constructed. For simplicity, the system can be defined as H–H AC/ZZ or A–D AC/ZZ according to the connection type. For example, the notation A–D ZZ indicates a junction constructed by connecting the AC edges of the GNR slice (in which the ZZ edge carbon atoms are modified by NH2–NO2 functional pairs) with Au electrodes, etc. The schematic diagrams of the molecular devices and the atomic structure of the molecular core are shown in Fig. 1. The results of one of our previous works show that edge modification can tune the elastic transport properties of GNR slices-based molecular devices.[19] In this work, we focus on the inelastic scattering process of the junctions.
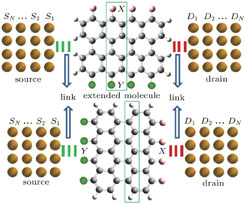 | Fig. 1. (color online) The Schematic diagrams of the molecular devices and the atomic structure of the molecular core. |
The geometric optimization and electronic properties are performed by using the density functional theory in the B3LYP level.[31, 32] In order to obtain more accurate results, we use the 6-31G basis set for light atoms such as carbon, oxygen, nitrogen, and hydrogen and the LanL2DZ basis set for Au atoms. The transport properties are calculated by the quantum chemistry for the molecular electronics (QCME) code.[33–35] The basic theories of the QCME code are the scattering theory and Green’s functional theory. The total current (I) in the junctions include the contributions of elastic (Iel) and inelastic (Iinel) electron scattering from the source to drain electrode. The elastic scattering current is mainly generated from electrons scattering from the molecular orbitals of the extended molecular. When energetic constraints are satisfied, the electron crossing the junction may exchange a definite amount of energy with the molecular nuclear motion, resulting in an inelastic component in the transmission current.[36] It is known that the electron-vibronic coupling can be mainly considered for the extended molecule part. In the adiabatic Born–Oppenheimer approximation, the purely electronic Hamiltonian of the molecular systems can be considered parametrically as dependent on the vibrational normal modes Q. By using a Taylor expansion, the nuclear motion depended wavefunction can be expanded along each vibrational normal mode. The small conductance change induced by the electron-vibronic coupling is commonly measured by the second harmonics of a phase-sensitive detector for the second derivative of the tunneling current d2I/dV2. Since most IETSs are measured at the electronic off-resonant region (where conducting levels are far from the Fermi energy), the adiabatic harmonic approximation can be applied. We can then use the first derivative to represent the vibrational motion part in the wavefunctions. In this case, the total quantum conductance is described as
 | (1) |
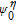
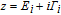
Comparing with the results of elastic scattering, the inelastic scattering current is very small. The energy exchange between the scattering electrons and nuclear motion will result in an inelastic component in the transmission current. The nuclear motion will introduce vibrational excited states which are the ones that contribute to the inelastic component in the case of off-resonant excitation. If the transmission is far below unity, the inelastic component can lead to a second tunneling path, which gives an additional current contribution to the tunneling current. While in case of a close to unity transmission the inelastic current can reduce the current (have a negative sign) by producing a “dip” in the inelastic tunneling spectrum. According to the characteristics of structure, the four molecular junctions studied in this paper can be seen as two major categories. The first system is that by connecting the hydrogen atom passivated AC edges with Au electrodes to form molecular junctions, and the ZZ edges modified by pure hydrogen or donor–acceptor pairs as the main scattering molecular core region. The other system is that by connecting the pure hydrogen atom or donor–acceptor pairs modified ZZ edges with Au electrodes to form molecular junctions, and the AC edges passivated by pure hydrogen as the central scattering molecular core region. We know that the electrode connections mainly affect the contact conductance. As the central scattering region, the actual composition and structure of the central portion of the extended molecule is the main electron transport channel of the devices. The electronic structure and molecular orbital distribution characteristics directly determine the tunneling conductance through the devices. Thus, functional modification on the open edges or body atoms of finite-sized GNR slices has become an important means to control the overall distribution of the carrier and the transport properties of the devices. The calculated inelastic tunneling spectrums of the four junctions as a function of applied bias voltage at room temperature have been shown in Fig.
 | Fig. 2. The inelastic electron tunneling spectroscopy as the function of source–drain bias voltage of the H–H AC, H–H ZZ, A–D AC, and A–D ZZ systems at room temperature a.u.. |
The main research point of this work is illustrating the effects of electrode connection types and central molecular modification on the inelastic tunneling current of the systems. So we have adopted arbitrary units in Fig.
It is known that the IETS is a powerful spectroscopic tool for molecular identification and chemical bonding investigations. In order to see the influence of the donor–acceptor groups on the IETS curves more clearly, in this part, we focused on two systems H–H ZZ and A–D ZZ and calculated the IETS at low temperature 4.2 K. The calculated results are shown in Fig.
 .
'> .
'> | Fig. 3. The inelastic electron tunneling spectroscopy as the function of source–drain bias voltage of the H–H ZZ and A–D ZZ system under lower temperature  |
It is known that the electron scattering process is sensitive to temperature. Furthermore, temperature also has a certain impact on the molecular motion, thus we discuss the temperature dependence of the IETS in this part. All of the transport properties have been calculated by the QCME code. The theoretical calculation methods of QCME show that there are two main factors which have exponential dependence on temperature closely related to temperature: the population distribution of thermal particles, which can be calculated using the Boltzmann distribution function Pn and the electron Fermi distribution at the end of source and drain electrodes f(E), where
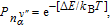 | (2) |
 |
 |
By using the A–D ZZ system as an example, we calculate the IETS under different working temperatures (from 4.2 K to 298 K). The results are shown in Figs.
In this paper, the inelastic tunneling spectroscopy of four junctions based on 8 × 8 finite-sized GNR slices have been calculated. Two edge functional modification types with hydrogen and donor–acceptor pairs have been considered. The IETS curves are sensitive to the atomic structures and electrode connection types of the systems. Comparing with the pure carbon-hydrogen systems, the donor–acceptor functional groups have effects on the rehybridization of edge carbon-groups and the carrier concentration and distribution of the molecular core. The nuclear motion and coupling between the electrodes and molecular core will introduce vibrational excited states which contribute to the inelastic electron scattering. New IETS peaks appear at multiple energy regions and the intensity distributions as a function of bias voltage of the IETS have been changed after the introduction of donor–acceptor groups. These changes mainly come from the vibration mode of atoms for the donor–acceptor groups and coupling between edge groups. Therefore, the IETS curves can be used as one of the qualitative detection methods of the composition of micro systems by comparing with the various vibrational modes obtained in experiments. For the temperature dependence of IETS, results show that the changes of IETS in the low-frequency region are caused by the introduction of the donor–acceptor groups and the population distribution of thermal particles, and that the effect of Fermi distribution on the IETS is persistent.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] |


