† Corresponding author. E-mail:
‡ Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 11464007), the Natural Science Foundation of Guangxi, China (Grant Nos. 2012GXNSFGA060002 and 2014GXNSFBA118241), the Guangxi Key Laboratory of Information Material Foundation, China (Grant No. 131021-Z), and the Guangxi Department of Education Foundation, China (Grant Nos. YB2014120 and KY2015YB104).
Glassy magnetic behavior and exchange bias phenomena are observed in single phase Mn3O4 nanoparticles. Dynamics scaling analysis of the ac susceptibility and the Henkel plot indicate that the observed glassy behavior at low temperature can be understood by taking into account the intrinsic behavior of the individual particles consisting of a ferrimagnetic (FIM) core and a spin-glass surface layer. Field-cooled magnetization hysteresis loops display both horizontal and vertical shifts. Dependence of the exchange bias field (HE) on the cooling field shows an almost undamped feature up to 70 kOe, indicating the stable exchange bias state in Mn3O4. HE increases as the particle size decreases due to the higher surface/volume ratio. The occurrence of the exchange bias can be attributed to the pinning effect of the frozen spin-glass surface layer upon the FIM core.
Magnetism of nanoparticles is interesting and is currently under extensive investigation.[1] The exchange bias (EB) effect, referring to a shift of the ferromagnetic hysteresis loop along the field axis by an amount of HE, which was first discovered in 1956 by Meiklejohn and Bean in fine particles of cobalt with a cobaltous oxide shell,[2] is gaining increased interest regarding its fundamental interest as well as potential applications, such as domain stabilizers in magnetoresistive heads and spin-valves.[3–5] In particular, both the core–shell systems with a ferromagnet (FM) core and an antiferromagnet (AFM) or a ferrimagnet (FIM) shell which could be obtained from an oxidation of transition metal nanoparticles, and the so-called “inverted” core–shell systems with an AFM core and a FIM shell, e.g., MnO/Mn3O4,[6,7] MnO/Mn2O3,[8] and Mn/Mn3O4,[9] have come into focus for the study of the EB effect resulting from the exchange coupling between the core and the shell.[10] Usually, the core and the shell correspond to two different magnetic materials in the core–shell nanoparticles systems. In addition to the FM/AFM and AFM/FIM interfaces, EB has also been observed in other types of interfaces involving a spin glass (SG) phase (e.g., FM/SG).[11–14] It is a common understanding that the uncompensated spins on the surface of the magnetic nanoparticles may result in a low-temperature SG phase.[15] However, particle systems may also exhibit the SG-like behavior due to the dipolar interactions between the particles according to the Stoner–Wohlfarth model.[16,17] Karmakar et al. argued that the low temperature SG state is very sensitive to the field and the degeneracy of the SG state can be reduced depending on the strength of the cooling field (HFC).[14] In some cases, the SG behavior can exist under high HFC.[13]
In this paper, we report an existence of a SG surface layer and a strong EB effect in single phase Mn3O4 nanoparticles with an average size of about 30 nm, 60 nm, and 90 nm. A detailed investigation of the frequency dependence of the ac susceptibility and Henkel plot for the Mn3O4 nanoparticles has been made, and the origin and nature of the observed EB have been elucidated. Our study suggests that the occurrence of the EB can be attributed to the pinning effect of the frozen SG surface layer upon the FIM core.
Single phase Mn3O4 nanoparticles with an average size of about 30 nm, 60 nm, and 90 nm were prepared by an auto-combustion method using analytical grade metal nitrate (Mn(NO3)2·6H2O) as the metal ion source, ethylene glycol (C2H6O2), citric acid (C6H8O7), and urea (CO(NH2)2) as the fuels, respectively.[18,19] Mn(NO3)2·6H2O and the fuel were dissolved in de-ionized water and mixed in an appropriate ratio to form the precursor solution. The mixed precursor solution was concentrated by heating until the excess free water was evaporated and spontaneous ignition occurred. The combustion finished within a few seconds and the resultant ash was collected. The ignition temperature has a significant influence on the particle size of the product, i.e., the particle size increases with the increase of the ignition temperature.[19] X-ray diffraction showed that the synthesized sample is single phase with a tetragonal space group I41/amd (No. 141).[19] Magnetic properties were measured on a physical properties measurement system (PPMS 9 T, Quantum Design).
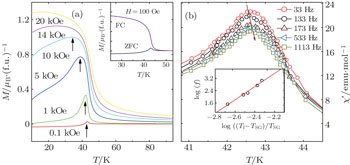
The temperature dependences of magnetization after zero-field cooled (ZFC) and field cooled (FC) processes are shown in the inset of Fig.
The ac susceptibility versus temperature for the 60 nm Mn3O4 particles, i.e., χ′(T), is shown in Fig.
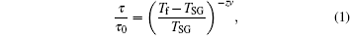
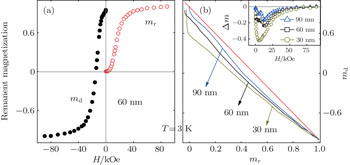
For an assembly of magnetic nanoparticles, the above SG behavior may also be associated with the dipolar interactions between the particles according to the Stoner–Wohlfarth model.[16,17] The interparticle interaction could be illustrated by the Henkel plot according to the Wohlfarth relation.[26] The DC demagnetization (DCD) remanence curve Md(H) is traced by taking initially the system to saturation, after that, the field is taken to a negative value of –H. The full curve is traced by repeating this procedure with negative fields of increasing amplitude up to the field of the same modulus as the initial saturation field. The isothermal remanent magnetization (IRM) curve Mr(H), on the other hand, is obtained by starting from a demagnetized sample (to be subjected to an ac demagnetization) and measuring the remanence for increasing fields up to Hsat (85 kOe). The measured IRM and DCD remanence curves for the 60 nm particles are shown in Fig.
As mentioned above, EB has been observed in some systems containing FM/SG interfaces.[11–13] Therefore, it would be interesting to explore if EB exists in the Mn3O4 nanoparticles containing FIM/SG interface. Figure
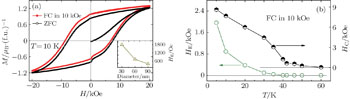 | Fig. 3. (a) Magnetic hysteresis loops of Mn3O4 at 10 K measured after ZFC and FC in 10 kOe field. Inset: particle-size dependence of HE at 10 K. (b) Temperature dependence of HE and HC. |
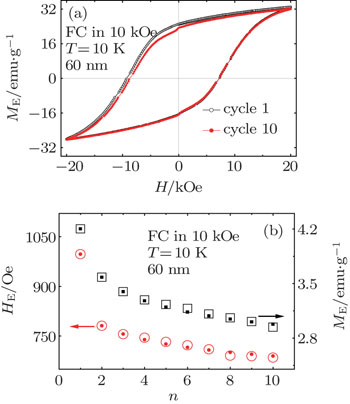
One of the important properties in the EB systems is the training effect, which describes a gradual decrease of HE when the system is continuously cycled through several consecutive hysteresis loop measurements.[16] The consecutive hysteresis loops were measured at T = 10 K after field cooling in 10 kOe. The first and the tenth loops for the 60 nm Mn3O4 particles are shown in Fig.

| Table 1. Parameters obtained from the best-fitting to the experimental data with Eq. ( |
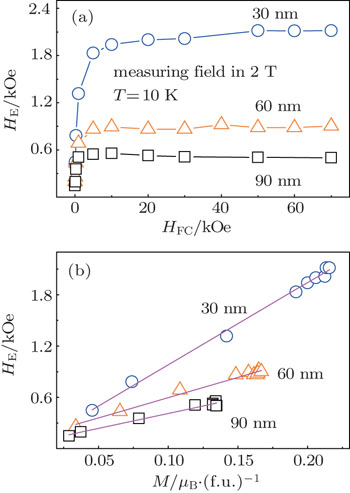
For further revealing the origin of the EB effect in Mn3O4, we studied the HFC dependence of the EB at 10 K. The M–H loops were measured between ±20 kOe after the sample was cooled under different fields from 300 K to 10 K. As shown in Fig.
In summary, we have observed spin glass behavior and EB phenomena in Mn3O4 nanoparticles. The dynamics scaling analysis of the ac susceptibility and the Henkel plot suggest that the observed SG behavior can be readily understood by taking into account the formation of surface spin glass due to the existence of broken bonds and the break of the translational symmetry of the lattice. The exchange bias field of the Mn3O4 nanoparticles shows a strong dependence on the temperature, strength of cooling field, as well as particle size. Below the SG freezing temperature Tp, HE increases with temperature decreasing. HE increases sharply with the cooling field HFC for HFC < 10 kOe and approaches to a saturation for larger HFC (> 10 kOe). HE increases as the particle size decreases due to the increased suface/volume ratio and the enhanced surface spin disorder. In addition, the observed training effect of the EB behavior has been explained well in terms of the existing relaxation model. These results imply that the existence of EB Mn3O4 nanparticles can be attributed to the pinning effect of the frozen SG surface layer upon the FIM core.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 |