† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 51202250).
The structural, electronic, mechanical properties, and frequency-dependent refractive indexes of GaSe1–xSx (x = 0, 0.25, and 1) are studied by using the first-principles pseudopotential method within density functional theory. The calculated results demonstrate the relationships between intralayer structure and elastic modulus in GaSe1–xSx (x = 0, 0.25, and 1). Doping of ε-GaSe with S strengthens the Ga–X bonds and increases its elastic moduli of C11 and C66. Born effective charge analysis provides an explanation for the modification of cleavage properties about the doping of ε-GaSe with S. The calculated results of band gaps suggest that the distance between intralayer atom and substitution of SSe, rather than interlayer force, is a key factor influencing the electronic exciton energy of the layer semiconductor. The calculated refractive indexes indicate that the doping of ε-GaSe with S reduces its refractive index and increases its birefringence.
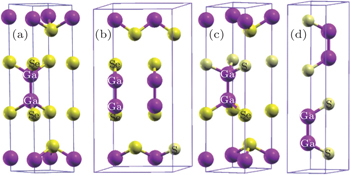
The layer ε-GaSe crystal, which belongs to space group 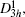
It was experimentally proved by many researchers that incorporation of different doping elements can significantly modify its mechanical and optical properties for frequency conversion. It is noted that In-doping[9,10] and Er-doping[11,12] ε-GaSe lead to 35% and 24% increase respectively in the intrinsic nonlinearity. Doping of ε-GaSe with Ag (0.04 mass%)[13] resulted in 10%–20% improvement in damage threshold and strengthened its hardness from 8 kg/mm2 to ≥ 10.7 kg/mm2, but 6% decrease in nonlinearity. Doping of ε-GaSe with Al (0.14 at%)[14,15] resulted in improved hardness up to 17 kg·mm–2 and increase of its ordinary refractive index. Te-doping[16,17] ε-GaSe resulted in 20% increase of nonlinearity and demonstrated the potential of frequency conversion into the THz range. In spite of the nonlinearity decreasing, S-doping[15,17,18] ε-GaSe resulted in 2.4 times and 1.5 times more efficient than pure and In-doping ε-GaSe crystal respectively due to its improvements of optical and mechanical properties. It was believed that the improvements of optical and mechanical properties in doping ε-GaSe were due to the modification of its cleavage property and decrease of stacking defects.
Despite the considerable amount of experimental work, there is relatively little research of the theoretical studies of doping ε-GaSe. In fact, the elastic and electronic properties of pure ε-GaSe have been investigated by using the full potential linear augmented plane-wave (FP-LAPW)[19,20] method, plane-wave pseudopotential (PW-PP)[21] method, and tight-binding (TB)[22] approach. It was suggested that the interlayer interactions were not solely of the van der Waals type in these layer compounds. Rybkovskiy and Arutyunyan[23] demonstrated the size-induced effects in ε-GaSe electronic structure by using pseudopotential method and GW approach. Further research was carried out by Rak et al.[24–27] to investigate the formation energies and electronic structures of In and Te defects in ε-GaSe, as well as the In-doping dependences of structural and mechanical properties. They pointed out that interstitial doping of cation-In can provide much higher energy barrier than those of the impurity of InGa and pure ε-GaSe as cleaving ε-GaSe crystal. However, there is a lack of theoretical approach to the electronic and mechanical properties of S-doping ε-GaSe. The present research focuses on the changes in electronic and mechanical properties caused by S-doping in ε-GaSe crystal.
Our calculations were performed by using plane-wave pseudopotential method of density functional theory (DFT) as implemented in the ABINIT code.[28] The exchange-correlation energy of electrons was evaluated in the local-density approximation (LDA) within the Perdew-Wang scheme.[29] The effective ionic potentials were approximated by Troullier–Martins[30] type of norm-conserving pseudopotentials (NCPPs) and the valence electrons included 4s and 4p states of Se and S as well as 4s, 4p, and 3d states of Ga. Since the spin–orbit effects are expected to be small for III–VI compounds,[21] we did not include spin–orbit interaction in our calculations.
The primitive cells of ε-GaSe and β-GaS are schematically shown in Figs. 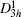
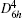
Structural optimizations of GaSe1–xSx (x = 0, 0.125, 0.25, and 1) were carried out by two steps. The first step is to optimize the ionic positions without cell shape and size optimization, and the second step is to optimize the cell shape and size from the cell with relaxed ionic positions. The theoretical structures of GaSe1–xSx (x = 0, 0.25, and 1) were obtained by using small unit cells (8 atoms/cell) where the Brillouin zone was sampled by 10 × 10 × 2 mesh. For GaSe0.875S0.125 crystal, the calculations were performed on 2 × 1 × 1 supercell with the BZ sampled by a 6 × 12 × 3 grid of k points. Convergence tests indicated that these sets of k-points with energy-cutoff equal to 54 Ha (1 Ha = 2 Ry = 27.2114 eV) give an accuracy of about 0.001 Å for lattice constants.
Linear-response properties such as the elastic module and Born effective charge (BEC) are obtained as second-order derivatives of the total energy with respect to external electric field or atomic displacements. Calculations of elastic modulus, dielectric functions and BEC were performed via the density functional perturbation theory (DFPT),[32–34] with a 60-Ha plane wave cutoff energy and Monkhorst–Pack grid equivalent to 12 × 12 × 4 grid on a primitive 8-atom GaSe1–xSx (x = 0, 0.25, and 1) cell. These sets of k-point energy cutoff can guarantee the adequate convergence of the calculations.
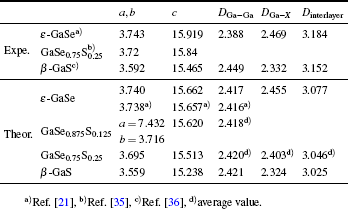
The calculated lattice parameters of GaSe1–xSx are listed in Table
| Table 1. The experimental and theoretical lattice parameters of GaSe1–xSx (x = 0, 0.125, 0.25, and 1). DGa–Ga, DGa–X, and Dinterlayer refer to the distances of Ga–Ga, Ga–X (X = Se, S) bond and interlayer. Unit is Å. . |
Since the electronic and elastic properties are sensitive to the inter-atomic distance of the semiconductor, we compare the distances of Ga–Ga (DGa–Ga) and Ga–X (DGa–X) of GaSe1–xSx (x = 0, 0.125, 0.25, and 1), as well as their interlayer distances (Dinterlayer). From ε-GaSe to β-GaS, we observe a decrease in the DGa–X and an increase in the DGa–Ga. This is because the S-atom is smaller than Se-atom and the ability for S atom to attract electrons is stronger than that for Se-atom, which could shorten the distance between S and Ga and lead to the decrease of DGa–X and the increase of DGa–Ga. With S incorporating into ε-GaSe, for GaSe0.875S0.125 and GaSe0.75S0.25, the above structural parameters are different between the multi-layer with SSe and multi-layer without SSe. As seen in Table
The electronic properties of ε-GaSe are calculated by using TB approach, PW-PP method and FP-LAPW method, as well as GW approximation. As pointed out in Ref. [26], the top of the valance band is derived from Se 4p electronic states. The effect of Ga 4s and Se 4p hybridization splits into two Se 4p derived bands near the top, giving rise to an energy gap in the band structure of ε-GaSe. Therefore, the electronic structure and band gap are sensitive to inter-atomic distance in this layer structure. Since different approaches give different results of lattice parameters and band gaps, we calculate the band structure of GaSe1–xSx crystals at optimized structural parameters obtained in LDA. In addition, we investigate the influence of interlayer distances on band gap of ε-GaSe.
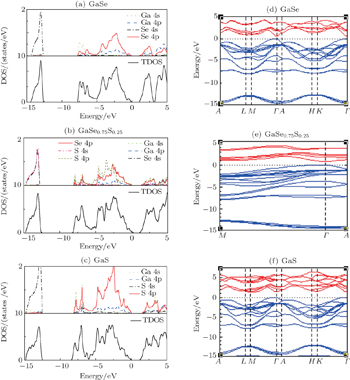
In order to characterize the substitutional impurity states, the densities of state (DOSs) of GaSe1–xSx (x = 0, 0.25, and 1) are calculated. The DOSs of these three materials look very similar. Their valence bands could be divided into three regions. It is seen in Fig.
To investigate the influences of S-doping on the band gaps of GaSe1–xSx, we calculate the electronic band structures of GaSe1–xSx (x = 0, 0.125, 0.25, and 1). As mentioned above, ε-GaSe and β-GaS belong to the hexagonal system, while GaSe0.875S0.125 and GaSe0.75S0.25 belong to the monoclinic system. The orbital character of β-GaS is similar to that of ε-GaSe which has been described by Rak et al.[26] Due to the hybridization between the Ga 4s states and X (X = S, Se) 4p states, some of the X 4p bands are pushed up in energy, giving rise to the semiconducting gaps. However, the bottom of the conduction band in β-GaS is shifted to the M point compared with that in ε-GaSe: it is converted from a direct gap semiconductor into an indirect gap semiconductor. For GaSe0.75S0.25, the isovalent S-doping does not change the numbers of atoms and electrons per unit cell, so the number of valence bands is the same as those of ε-GaSe and β-GaS. Its band structure along Γ–A–M path is also similar to that of ε-GaSe. The calculated result shows that both the top of the valence band and the bottom of the conduction band lie at the Γ point, indicating that GaSe0.75S0.25 is a direct band gap semiconductor. In the calculation of GaSe0.875S0.125, we use the 2 × 1 × 1 supercell, and the results show that the size of BZ is reduced by half, while the number of valence bands is increased by double. GaSe0.875S0.125 in LDA calculation is also a direct band gap semiconductor. The calculated band gaps of GaSe1–xSx (x = 0, 0.125, 0.25, and 1) in LDA correspond respectively to 0.67 eV, 0.72 eV, 0.78 eV, and 1.40 eV (Table
| Table 2. Band gaps (in units of eV) of GaSe1–xSx (x = 0, 0.125, 0.25, and 1) from experiments and LDA calculations. The band gaps of GaSe1–xSx increase with the composition x increasing. . |
As pointed out in the analysis of DOS, the upper valence band edge is formed by Se 4p states, whereas the conduction band bottom is composed of Ga 4s states. Moreover, the optimized structures indicate that the lattice constants decrease with the composition x increasing. The decrease of lattice constant is accompanied by the decreasing of Ga–X (X = S, Se) bond length, which leads to the increasing of overlapping between Ga 4s states and Se 4p states. In order to investigate the influence of interlayer force on band gap, the calculations have been done by altering the interlayer distance without changing the relative position of intralayer atoms. Based on the optimized result of ε-GaSe (named structure 0), the interlayer distances are assumed to increase 0.02 Å (named structure 1) and reduce 0.02 Å (named structure 2) respectively. The calculated band gaps of both structure 1 and structure 2, located at the Γ-symmetry point, are direct with the same value of 0.67 eV which is equal to that of structure 0. As the structure parameter is based on the optimized result of GaSe0.75S0.25, the band gap is also direct with a value of 0.76 eV which is between that of ε-GaSe (0.67 eV) and that of GaSe0.75S0.25 (0.78 eV). However, with using the structure optimized result of ε-GaSe without including Ga 3d electrons, the calculated band gap is shifted to be indirect with a value of 1.013 eV. Apparently, the positions of intralayer atoms and impurity of SSe are the key factor for influencing the electronic exciton energy. Note that S-doping could change van der Waals force between layers and then influence the equilibrium energy of GaSe. Therefore, the changes of van der Waals force could influence the distance of intralayer atoms and then alter its band gap.
Elastic constants represent the second derivatives of the energy density with respect to strain:
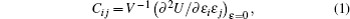
| Table 3. Calculated values of elastic constants (Cij), bulk modulus (B), shear modulus (G), Young’s modulus (E), and Poisson’s ratio (σ) for GaSe1–xSx (x = 0, 0.25 and 1) (unit is GPa for elastic constants and modulus). . |
The elastic constants C11 and C33 represent the resistances to longitudinal compression along the x and z crystallographic axes respectively, while C44 and C66 represent the resistances to transverse deformation in [100] and [001] crystal planes respectively. Therefore, the elastic constants of C11 and C66 are determined by the chemical bonds along [100] or [010] direction, while C33 and C44 are determined by the chemical bonds along [001] direction. For the GaSe1–xSx (x = 0, 0.25, 1) crystals, C11 is much higher than C33 and C66 is much higher than C44, suggesting that the bonding strength along the [100] or [010] direction is stronger than that along the [001] direction. These can be easily understood by the high anisotropy induced by the layer structure. From ε-GaSe to GaSe0.75Se0.25 to β-GaS, we observe monotonic increases in C11 and C66, but there is no obvious increasing nor decreasing trend in any of C33, C44, C12, and C13. These can be explained after a careful analysis of the relationships between the chemical bonds and elastic modulus of these layer compounds. The Ga–Ga bonds are along the z crystallographic axes and at about 120-degree angle with respect to Ga–X bonds (X = S, Se). The chemical bonds along [100] or [010] direction are composed of Ga–X bonds which determine the elastic constants of C11 and C66. The chemical bonds along the [001] direction are composed of Ga–Ga bonds and Ga–X bonds, and influenced by van der Waals force between layers. Therefore, the C33 and C44 are determined by the Ga–Ga bonds and Ga–X bonds, as well as interlayer force. As mentioned above, the Ga–S bond is shorter and stronger than Ga–Se bond, leading to the weakening of Ga–Ga bond from ε-GaSe to GaSe0.75Se0.25 to β-GaS. Thus, the doping of GaSe with S can increase its elastic constants of C11 and C66 and can change its C33 and C44.
From these calculated elastic constants, some important characteristics, such as bulk modulus B, shear modulus E, Young’s modulus EH and Poisson’s ratio σ, can be evaluated, and their results are also listed in Table
Assuming the monotonic behavior for the mechanical properties of GaSe1–xSx to be a function of composition x value, doping of ε-GaSe with sulfur becomes stiff as the concentration of substitutional S increases. This can be explained by the changes of intralayer bonding induced by element-doping. However, it is experimentally observed that the doping of ε-GaSe with heavy atoms (like Te, In, and Er) can modify its cleavability and reduce its stacking defects. Although the C44 of S-doping crystals is larger than that of pure ε-GaSe, the increased effects from calculations are much smaller than experimental results. In the next subsection, we will try to search for different mechanisms and analyze the effects of Born effective charge on interlayer force.
The Born effective charge (BEC) tensor 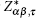
The BEC tensors of atoms in ε-GaSe and β-GaS each with hexagonal structure are diagonal and have only two independent components: along and perpendicular to the tetragonal axis, 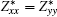
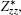
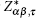
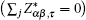
We observe that the 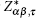
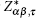
| Table 4. Calculated Born effective charge tensors along with their average values for Ga, Se and S atoms in GaSe1–xSx (x = 0, 0.25, and 1). . |
Rak et al.[25] have investigated the formation energies of In-induced and Te-induced defects by first principles. Their calculations indicated that Te and In prefer the substitutional Se and Ga sites, respectively. Nevertheless, cation-In can also occupy interstitial site between multi-layers and form a chemical bond with anion–Se of adjacent multi-layers. They pointed out that it is this new bonding that strongly modifies the cleavability of the crystal along the plane parallel to the atomic layer. However, it is experimentally observed that not only the cation (In, Al, and Er) but also the anion (S, Te) can significantly modify the cleavage property of ε-GaSe and lower its stacking defects. We will explain this modification by analyzing BEC, which is shown in Table
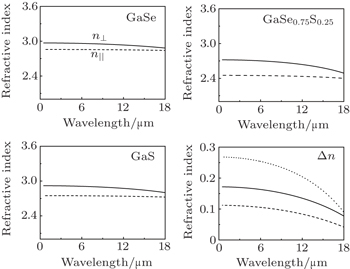
In order to investigate the effect of S-doping on refractive index of ε-GaSe, we calculate and compare the low-frequency dielectric permittivity tensors of infrared crystals: ε-GaSe, GaSe0.75S0.25, and β-GaS. The frequency dependent dielectric permittivity can be decomposed in the contributions of different modes as[32]
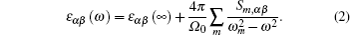
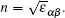
| Table 5. Calculated and experimental high-frequency dielectric tensor components ε‖ (parallel to the c axis) and ε⊥ (perpendicular to the c axis) of GaSe1–xSx (x = 0, 0.25, and 1). . |
In this paper, we present the results of first principles: lattice parameters, electronic structures, elastic moduli and birefringences of GaSe1–xSx (x = 0, 0.25, and 1). The relationships between intralayer structure and elastic modulus from ε-GaSe to GaSe0.75S0.25 to β-GaS are demonstrated. The calculated results indicate that the strengths of Ga–X (X = Se, S) bonds and elastic moduli of C11 and C66 increase with the increase of composition (x value) for GaSe1–xSx. The analysis of Born effective charge provides an explanation for the significant modification of cleavage properties for the doping of ε-GaSe.
We find that the distance between intralayer atoms and substitutions of SSe, rather than interlayer force, is a key factor of influencing the electronic exciton energy of the layer ε-GaSe semiconductor. However, the change of interlayer distance may influence the distance between intralayer atoms and further affect the band gap of ε-GaSe. From ε-GaSe to GaSe0.75S0.25 to β-GaS, the refractive index is decreasing and the birefringence is increasing, indicating that the doping of ε-GaSe with S may lower its absorption coefficient. However, the increase of birefringence may worsen the walk-off effect in laser frequency conversion.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 | |
| 34 | |
| 35 | |
| 36 | |
| 37 |