† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11365003 and 11165004).
The effect of physiological and pathological abnormal excitation of a myocyte on the spiral waves is investigated based on the cellular automaton model. When the excitability of the medium is high enough, the physiological abnormal excitation causes the spiral wave to meander irregularly and slowly. When the excitability of the medium is low enough, the physiological abnormal excitation leads to a new stable spiral wave. On the other hand, the pathological abnormal excitation destroys the spiral wave and results in the spatiotemporal chaos, which agrees with the clinical conclusion that the early after depolarization is the pro-arrhythmic mechanism of some anti-arrhythmic drugs. The mechanisms underlying these phenomena are analyzed.
Spiral waves exist widely in nature, such as the oxidation of CO on platinum, [ 1 ] the physical system, [ 2 ] and the neuronic system, [ 3 , 4 ] etc. Researchers studied the dynamics of spiral waves in different systems, [ 5 – 13 ] among which the spiral waves in myocardium is one of the hot topics. [ 11 – 13 ] The results showed that the spiral waves appearing in myocardium is one of the reasons for tachyarrhythmia, and the spatiotemporal chaos generated from spiral waves will lead to fibrillation, which is the main cause of sudden cardiac death. [ 14 ] Some factors affect the dynamics of spiral waves during tachyarrhythmia, such as the mechanical deformation, [ 11 ] electrophysiological heterogeneity, [ 12 ] and the early after depolarization (EAD). [ 13 ]
EAD is a secondary depolarization phenomenon that occurs occasionally before the completion of repolarization of an action potential. [ 15 ] The interest in EAD can be traced back to decades ago. In 1989, the clinical experimental results indicated that some anti-arrhythmic drugs had actually increased mortality, [ 16 ] and then EAD was considered to be one pro-arrhythmic mechanism of these drugs in 1992. [ 17 ] From then on, topics about EAD have been studied, such as the mechanisms underlying the EAD, [ 18 ] the predictors of the EAD-induced cardiomyopathy, [ 19 ] the relation between the EAD and the cardiac arrhythmias, [ 13 ] etc. The EAD includes the supernormal excitation and the abnormal excitation. [ 15 ] The effect of the supernormal excitation on the spiral waves has been discussed numerically, [ 20 ] but the numerical investigations about the abnormal excitation are lacking.
The abnormal excitations are considered as the result of the modification of the ion channel and/or electrogenic pump currents, and they can be induced in several experimental models. [ 17 ] In contrast to the supernormal excitation, the probability of the abnormal excitations is very low, because they appear occasionally. [ 15 ] When the abnormal excitation happens, a cell can be excited under a lower stimulation but cannot be excited as the stimulation is higher. [ 15 ] In this paper, the effect of abnormal excitation on spiral waves in the ventricle is studied using cellular automaton model.
The organization of the rest of the paper is as follows. First, the cellular automaton model for the ventricle considering the abnormal excitation is introduced. After that, the computer simulation results and the analysis are presented. The conclusion is provided finally.
For simplicity, the differences of the repolarization characteristics between cells in different layers of the ventricle are ignored here, and then the transmembrane potential is divided into 14 states according to the electric characteristics of the ventricular cell. [ 21 ] The relation between the states and the transmembrane potential is shown in Fig.
 | Fig. 1. Relation between the states and the transmembrane potential of a ventricular cell. Symbols ○, •, and ⋆ represent the rest, excited, and refractory states, respectively. |
The ventricle slice is put on the xoy coordinate surface and divided into 256 × 256 lattices, and the cells are distributed on these grid points. The no-flux boundary conditions and the Moore neighborhood are used throughout this paper. For the neighborhood radius R , there are (2 R + 1) 2 − 1 neighbors in a cell’s neighborhood. The state of the cell ( x , y ) at t is represented by u x , y ( t ) ∈ (0, 1, 2, …, 13), where u x , y ( t ) = 0, 1 denote the rest and excited state, respectively, while the states of u x , y ( t ) = 2, …,13 represent the refractory states. Under the normal conditions, the time evolution of a cell is given as follows:
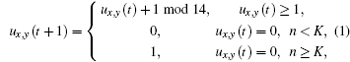
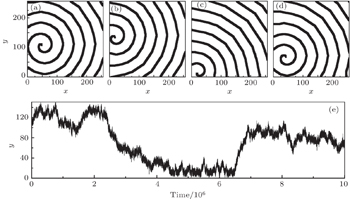
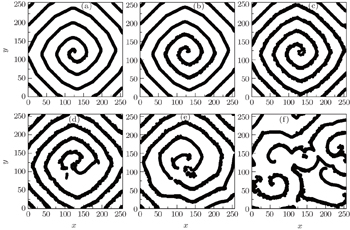
The electric impulse coming from the sinoatrial node can be impressed only when the excitability of the ventricular myocyte is high enough. In the above model, for R = 3, the spiral waves can be formed only when K ≤ 7: a plane wave produced initially ( t = 0) is cut at t = 40, and then the spiral wave comes into being (Fig.
Because the abnormal excitations occur before the completion of repolarization of an action potential, [ 15 ] the state 13 can be excited with abnormal excitation probability P for every cell in this model. After considering abnormal excitation, the time evolution of states [0, 12] is still in the form of Eq. (

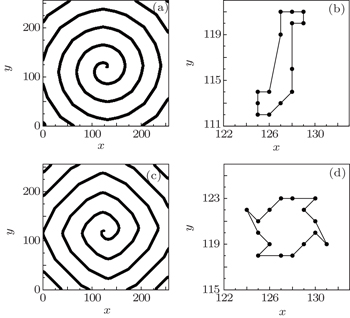
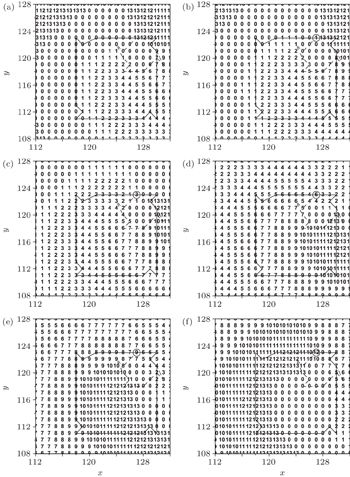
Because the plane wave can be formed and evolved only when K ≤ 7 for R = 3, so the lower and higher excitability of the medium may be represented by K = 7 and K = 6, respectively (the further results show that the conclusions obtained from K < 6 are the same as that from K = 6). The results have indicated that the excitability of the medium affects the dynamics of the spiral waves, [ 22 ] so the effect of the physiological abnormal excitation under different excitabilities will be discussed in the following. To investigate the effect of the physiological abnormal excitation on the spiral waves when the medium is highly excitable, the physiological abnormal excitation represented by K 1 = 3, K 2 = 5, and P = 0.01 is added in the system producing Fig.
Figure
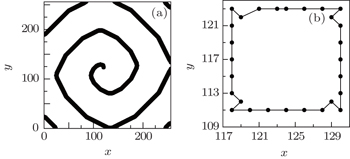
The excitability of the myocyte decreases with the cells aging. [ 23 ] To investigate the effect of the physiological abnormal excitation on the spiral waves in low excitable medium, the abnormal excitation represented by K 1 = 3, K 2 = 5, P = 0.01 is added in the system producing the Fig.
Figure
Comparing Fig.
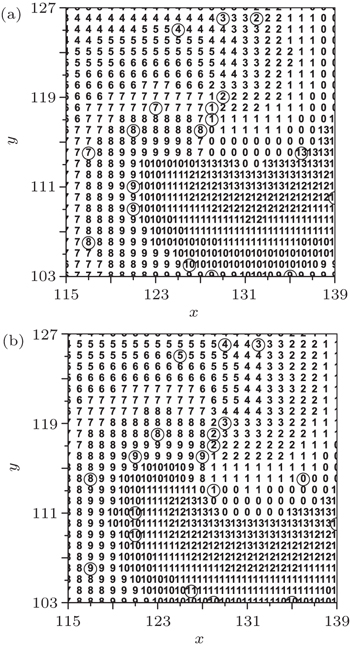
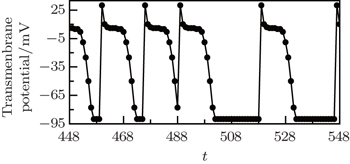
When the cell (127, 123), which is on the original tip trajectory, is selected randomly to be abnormally excited, it cannot return to 0 but to 1, so it undergoes the series of refractory states again, and the interval between its successive state 1 is twice of the states number, i.e., twice of 14. This means that the period of the new tip is 28. Because the tip is the controller of the spiral wave, [ 24 ] so the system falls into the new spiral wave with period of 28. To test the above analyses, the transmembrane potential of cell (127, 123) is followed, and the result is shown in Fig.
Data from the clinical experimental studies suggest that the abnormal excitation happens more frequently in hypertrophied ventricles than in normal ventricles, i.e., the abnormal excitation turns from physiological to pathological in hypertrophied ventricles, which will lead to fatal fibrillation. [ 25 ] To investigate the effect of the pathological abnormal excitation on the spiral waves, the abnormal excitation represented by appropriate P , K 1 , and K 2 are added into the system producing the Fig.
As an example, figure
The abnormal excitation is a modality of early after depolarization, and it appears occasionally in myocytes. Under the normal conditions, the electric impulse moves throughout the heart with plane waves or target waves, which make sufficient distance between the last refractory state and the excited state, so the abnormal excitation cannot happen for these wave patterns. But for the spiral pattern, the abnormal excitation can occur and play a role. In this paper, the effect of physiological and pathological abnormal excitation on the spiral waves is studied based on the cellular automaton model of a ventricle slice. The computer simulation results can be summarized as follows. (i) For the medium with a high excitability, the physiological abnormal excitation causes the stable spiral wave to meander irregularly with a very low velocity, but the form and the wavelength of the spiral wave hardly change. This phenomenon means that the physiological abnormal excitation hardly affects the tachyarrhythmia when the ventricle is highly excitable. (ii) For the medium with a low excitability, the physiological abnormal excitation happening on the tip trajectory blocks the movement of the excited state along the original trajectory, so the tip leaves the original trajectory and falls into a new trajectory finally, i.e., the system turns into a new spiral wave pattern. The period of this new spiral wave is longer than the original, which means that when the system is of low excitability, the physiological abnormal excitation can mitigate the tachyarrhythmia. (iii) For the system with a low excitability, the pathological abnormal excitation may cause the break up of the spiral waves and result in the spatiotemporal chaos, which agrees with the clinical conclusions that the early after depolarizations is the pro-arrhythmic mechanism of some anti-arrhythmic drugs. This paper uncovers the mechanisms of the effect of the abnormal excitation on spiral waves on cellular level, and the results give an insight into the mechanisms of some anti-arrhythmic drugs.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 |