† Corresponding author. E-mail:
Project supported by the Scientific and Innovative Action Plan of Shanghai, China (Grant No. 18511110502) and Equipment Pre-research Fund Key Project, China (Grant No. 6140922010601).
High quality 0.02 mol%, 0.05 mol%, and 0.08 mol% Fe: β-Ga2O3 single crystals were grown by the floating zone method. The crystal structure, optical, electrical, and thermal properties were measured and discussed. Fe: β-Ga2O3 single crystals showed transmittance of higher than 80% in the near infrared region. With the increase of the Fe doping concentration, the optical bandgaps reduced and room temperature resistivity increased. The resistivity of 0.08 mol% Fe: β-Ga2O3 crystal reached to 3.63 × 1011 Ω ⋅cm. The high resistivity Fe: β-Ga2O3 single crystals could be applied as the substrate for the high-power field effect transistors (FETs).
The β-gallium oxide (β-Ga2O3) crystal has become an increasingly attractive semiconductor for the potential applications in the fields of high-power devices, solar blind ultraviolet photodetectors, and Schottky x-ray detectors[1–3] due to its outstanding material properties. Unintentionally-doped β-Ga2O3 has a bandgap of 4.5–4.9 eV[4–6] with a theoretical breakdown electric field of 8 MV/cm, which is two times larger than those of SiC and GaN.[7,8] Baliga’s figure of merit (BFOM) is 3214 which is only lower than that of diamond. Gallium oxide commonly has five polymorphs named as α, β, γ, δ, and ε. Among them, β-Ga2O3 is the most stable crystal structure.[9] High crystalline quality β-Ga2O3 bulk crystal can be grown by the melt-growth techniques such as Czochralski (CZ), edge-defined film-fed growth (EFG), and floating zone (FZ) method.[10–12] With the development of β-Ga2O3 devices, the high resistance β-Ga2O3 substrate is needed to fabricate high-power field effect transistors to ensure lower leakage current.[13–15] However, unintentionally-doped β-Ga2O3 usually exhibits n-type due to the residual impurities in raw materials such as Si, Sn, and Ge.[16] The electron concentration and resistivity of unintentionally-doped β-Ga2O3 are typically about 1017 cm−3 and 10−1 Ω ⋅cm, respectively.[17–20] Doping deep acceptors is needed to compensate the free carriers to achieve semi-insulating (SI) β-Ga2O3.
Currently, β-Ga2O3 can be made SI by doping Fe.[21–23] The Fe ion acts as deep acceptors in β-Ga2O3 and pins the Fermi level away from the conduction band (CB). By thermoluminescence (TL) spectroscopy, an additional defect center with an activation energy of 0.62 eV was introduced in Fe: β-Ga2O3 with the resistivity of 5.10× 106 Ω ⋅cm.[24] An acceptor energy of 0.86 eV was tested by the high temperature Hall effect measurement in the conduction band with Fe doping concentration of 8× 1017 cm−3.[25] Based on first principles study, the bandgap of β-Ga2O3 reduced to 3.30 eV by doping Fe.[26] These results show that the defect energy may have relationship with the Fe doping concentration and influence the physical characterization of high resistivity Fe: β-Ga2O3 crystal.
In this paper, high resistivity Fe: β-Ga2O3 single crystals were grown by the FZ technique. The optical, electrical, and thermal properties of the as-grown crystals have been studied, which would provide basic performance parameters for the device fabrications.
The 0.02 mol%, 0.05 mol%, and 0.08 mol% Fe: β-Ga2O3 single crystals were grown by the FZ method. Feed rods were prepared with gallium oxide powder (purity 99.9999%) and Fe2O3 powder (purity 99.99%). The rods were shaped by a cold press of 50 MPa for 20 min and then sintered at 1500 °C for 24 h in the air atmosphere. Crystal growth was carried out by advanced four halogen lamp floating zone furnace (Quantum De-sign-IRF01-001-00). The crystal growth rate was 5 mm/h with a rotation speed of 8 rpm in the air flow atmosphere along the [010] direction.
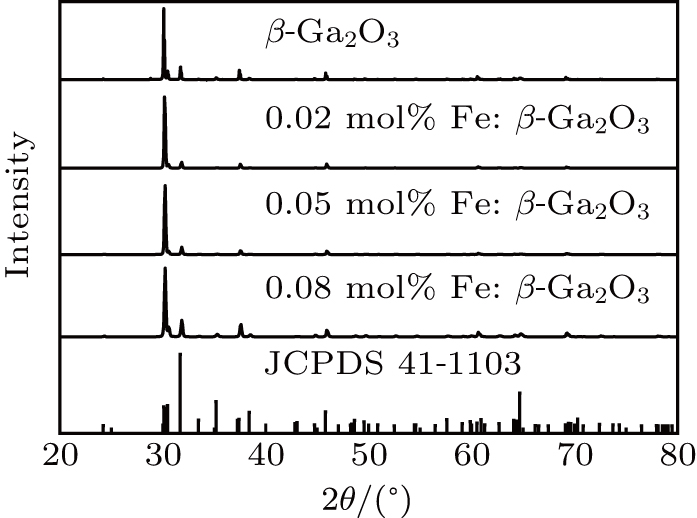
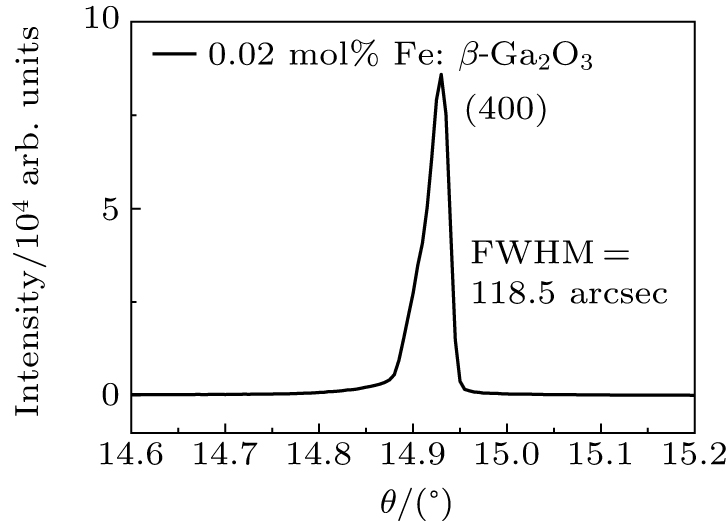
To analyze the single crystal structure and quality, the x-ray powder diffraction and x-ray diffraction rocking curve were tested by a Bruker D8 ADVANCE diffractometer. The optical transmission spectra were measured by an UV–VIS–NIR spectrophotometer (Varian Cary 5000). The room temperature current–voltage (I–V) curves and the temperature dependence resistivity were obtained by a Keithley 4200 semiconductor characterization system and an Agilent 6517B megger. The specific heat and thermal diffusivity were tested by a DSC 8000 differential scanning calorimeter and a LFA467 Hyper Flash conductometer. The samples were cut along the (100) plane and double-sided polished by chemical mechanical polishing.
The as-grown Fe: β-Ga2O3 crystals are brown, transparent without cracks, as shown in Fig.
Figure
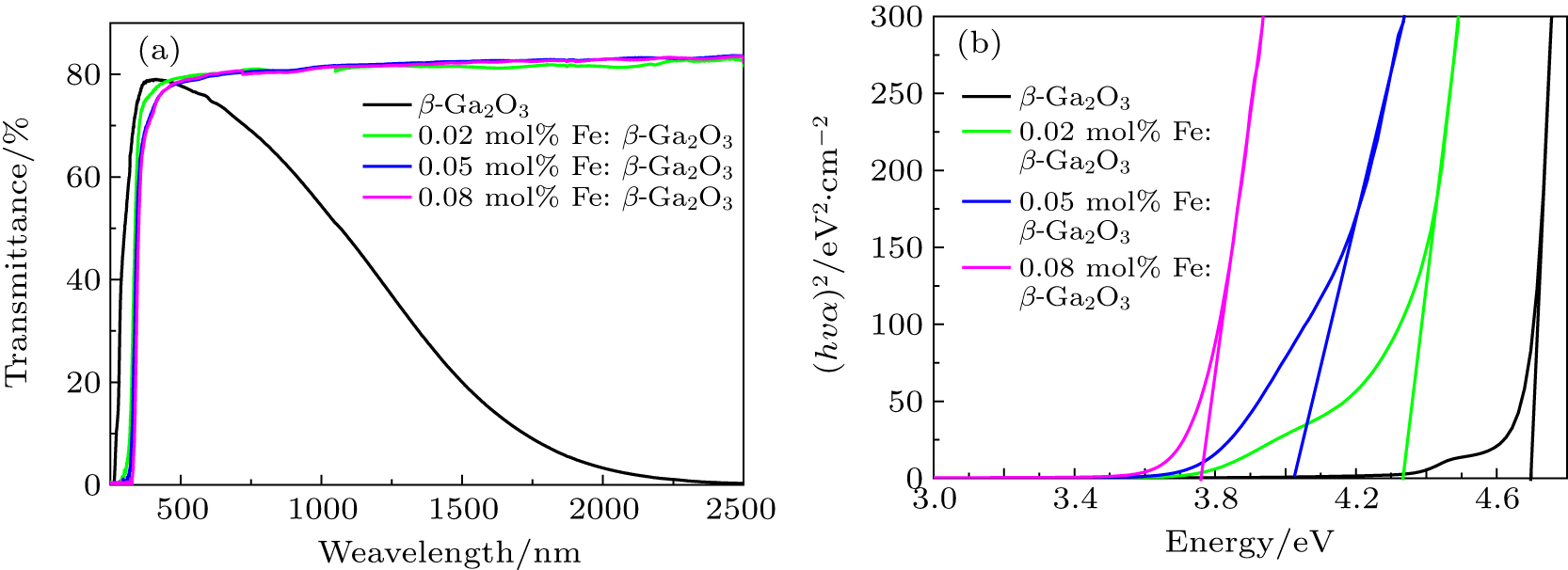 | Fig. 4. Room-temperature optical transmission spectra (a) and absorption edges (b) of 0.02 mol%, 0.05 mol%, and 0.08 mol% Fe-doped β-Ga2O3 crystals. |
The optical bandgap energy can be obtained by plotting (αhν)2 against hν, where α is the absorption coefficient and hν is the photon energy.[24,27] As shown in Fig.
| Table 1. Influences of the Fe dopant on the optical bandgap and resistivity. . |
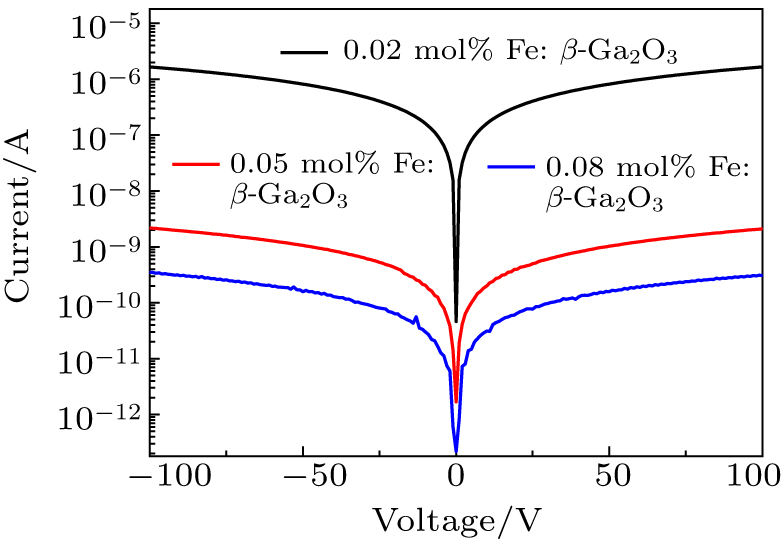
The root mean square roughness (RMS) of the polished samples is about 0.5 nm in the area of 7 μm × 7 μm tested by atomic force microscopy. The samples were cleaned by acetone and Ti/Au (20 nm/50 nm) electrodes were deposited by electron-beam evaporation with an area of 4 mm × 4 mm. In order to obtain Ohmic contact between Ti and Fe: β-Ga2O3, a rapid thermal annealing at 850 °C for 30 s in the nitrogen atmosphere was performed. Figure
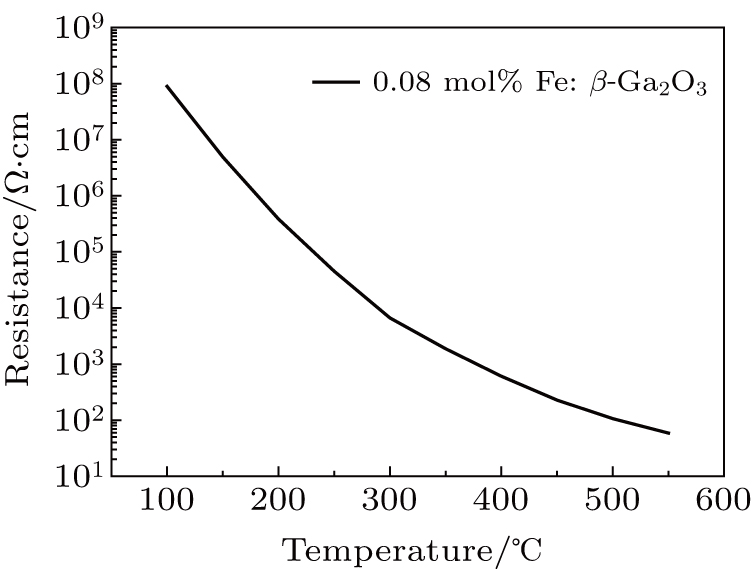
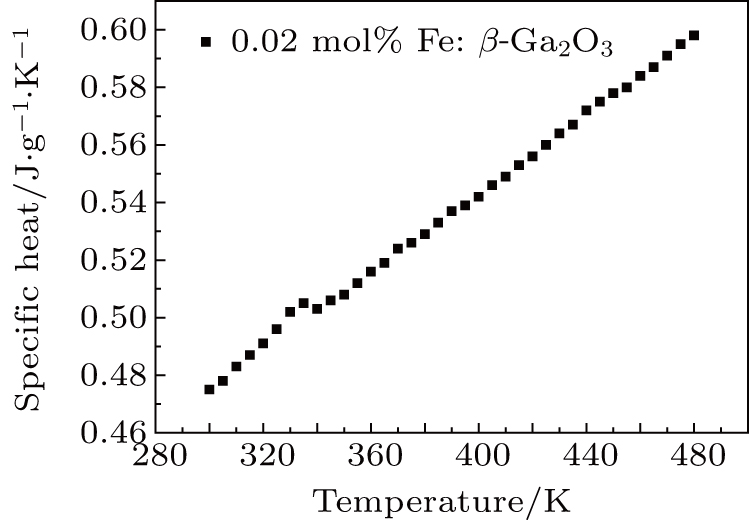
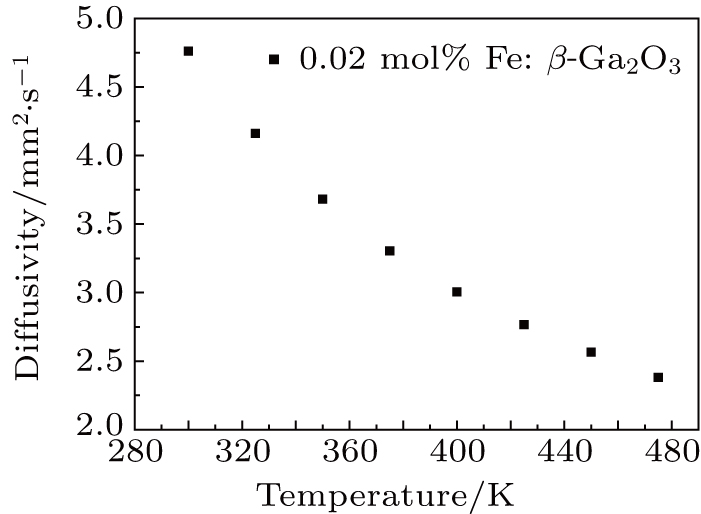
The thermal conductivity of the substrate plays an important role in the β-Ga2O3 power devices. 0.02 mol% Fe: β-Ga2O3 crystal was cut into 4 mm × 3 mm × 1 mm to test the specific heat. Figure
Fe: β-Ga2O3 crystals with different doping concentrations were grown by the FZ method. With the increase of the Fe doping concentration to 0.08 mol%, the optical bandgap reduces to 3.760 eV. For the unintentionally-doped β-Ga2O3 crystal, the optical bandgap is 4.695 eV. Fe: β-Ga2O3 crystals have high resistivity at room temperature. The room temperature resistivity of 0.08 mol% Fe: β-Ga2O3 crystal is 3.63 × 1011 Ω ⋅cm, and the resistivity reduces rapidly to 59 Ω ⋅ cm when the temperature raises up to 550 °C. The thermal conductivity of the 0.02 mol% Fe: β-Ga2O3 crystal is 12.780 W/m⋅K at room temperature, slightly lower than the reported result of β-Ga2O3 crystal. Fe: β-Ga2O3 crystal is a good candidate for high-power devices.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] |