† Corresponding author. E-mail:
Project supported by the Natural Science Foundation of Zhejiang Province, China (Grant No. LQ19E010006), the National Natural Science Foundation of China (Grant Nos. 51671048 and 91963123), the Ten Thousand Talents Plan of Zhejiang Province, China (Grant No. 2018R52003), and the Fundamental Research Funds for the Provincial University of Zhejiang Province, China (Grant No. GK199900X022).
The electronic structures, magnetic properties, and martensitic transformation in all-d-metal Heusler-like alloys Cd2MnTM (TM = Fe, Ni, Cu) were investigated by the first-principles calculations based on density-functional theory. The results indicate that all three alloys are stabilized in the ferromagnetic L21-type structure. The total magnetic moments mainly come from Mn and Fe atoms for Cd2MnFe, whereas, only from Mn atoms for Cd2MnNi and Cd2MnCu. The magnetic moment at equilibrium lattice constant of Cd2MnFe (6.36 μB) is obviously larger than that of Cd2MnNi (3.95 μB) and Cd2MnCu (3.82 μB). The large negative energy differences (ΔE) between martensite and austenite in Cd2MnFe and Cd2MnNi under tetragonal distortion and different uniform strains indicate the possible occurrence of ferromagnetic martensitic transformation (FMMT). The minimum total energies in martensitic phase are located with the c/a ratios of 1.41 and 1.33 for Cd2MnFe and Cd2MnNi, respectively. The total moments in martensitic state still maintain large values compared with those in cubic state. The study is useful to find the new all-d-metal Heusler alloys with FMMT.
The ferromagnetic Heusler-based shape memory alloys (FSMAs)[1] which is a large family of multifunctional materials have been well studied both theoretically and experimentally. Introducing the external fields, the martensitic transformation (MT) will happen with the change of magnetic states from ferromagnetic (FM) state and antiferromagnetic/paramagnetic (AFM/NM) state. In the process of FMMT, there are usually accompanied by various physical properties, such as magnetic-field-induced recovery, magnetocaloric effect, barocaloric effect, exchange bias, magnetoresistance, and magnetostrain.[2–19]
Recently, the all-d-metal Heusler-like alloys based on d–d hybridization by substituting transition metal (TM) with less valence electrons for main group element in classic Heusler alloys were reported.[20,21] The metamagnetic MTs and corresponding multifunctional properties (magnetic-entropy change,[20] field-induced strain,[21] giant elastocaloric effect[11,22,23]) were gained in MnNiTi(Co) system. Wei[22] reported a reversible elastocaloric effect with ΔTad = 9.0 K at a strain level of 4.6% for Ni35Co15Mn35Ti15. Aznar[23] reported a giant barocaloric effect for Ni50Mn31.5Ti18.5, which has maximum value of adiabatic temperature (12 K) and isothermal entropy changes (74 J⋅kg−1 ⋅ K−1) under 4 kbar (1 bar = 105 Pa). More ferromagnetic MT behaviors[24–26] were also reported in other MnNiTi(Co) system. The exploration and design of new all-d-metal Heusler alloys has become a research hotspot.
Afterward, the crystal and electronic structures and MTs were predicted in Mn-based, Ni-based, and Pd-based all-d-metal Heusler alloys by first-principles calculations.[27–30] Among the Heusler-based alloys, the Zn-based and Cd-based materials are less investigated in comparison with the other d-metals. This is probably due to the high vapor pressure (low boiling point)[31–33] of these metals which makes them difficulty to be fabricated. Thus the first-principles calculations become an effective method to predict crystal structure and phase stability. Recently, Han et al.[34] found that the Zn2MnTM crystalize L21-type structure with ferromagnetic state, Zn2RuMn, Zn2RhMn, Zn2OsMn, and Zn2IrMn have possible MT. Yang et al.[35] reported the atomic configurations, electronic, magnetic, mechanical, and dynamic properties for Cr2ZnSi/Cr2ZnGe.
In this work, we investigated the electronic structures, magnetic properties, and martensitic transformation in Cd-based all-d-metal Heusler-like alloys Cd2MnTM (TM = Fe, Ni, Cu) by first-principles calculations. And we found that three samples reveal the ferromagnetic state with L21-type structure, and the Cd2MnFe and Cd2MnNi may possess the ferromagnetic martensitic transformation.
The Cambridge Serial Total Energy Package (CASTEP) code, based on pseudopotential method with a plane-wave basis set, was used to investigate the electronic structures, magnetic properties, and MT of all-d-metal Heusler-like alloys Cd2MnTM (TM = Fe, Ni, Cu).[36,37] The generalized-gradient-approximation (GGA)[38] and ultra-soft pseudo-potential[36] were selected to carry out the electron-exchange-related energy and interaction between atomic core and valence electrons, respectively. A 500-eV plane basis set energy cut-off and the Monkhorst–Pack special 16×16×16 k-point mesh were used in the irreducible Brillouin zone. The selected energy convergence and self-consistent field tolerances were within 1 × 10−6 eV/atom and 1 × 10−7 eV/atom, respectively.
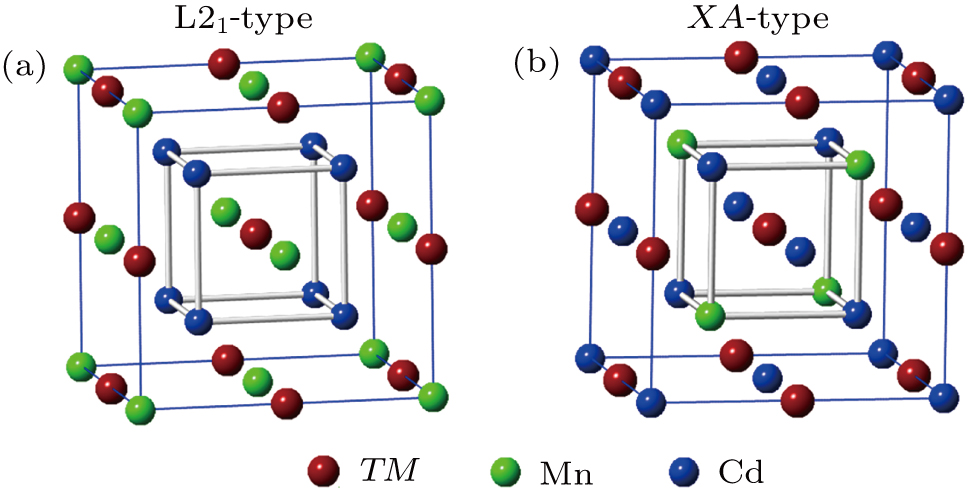
The classic full-Heusler alloys X2YZ (X and Y are TM atoms, Z is main group atom) have the highly ordered structures, there are four atom sites occupied the atoms, namely, A (0,0,0), B (0.25,0.25,0.25), C (0.5,0.5,0.5), and D (0.75,0.75,0.75). Normally, there are two possible atomic-site orderings on the basis of the site rule of Heusler alloys.[39,40] The first is L21-type structure, in which two X atoms with higher valence electrons prefer to occupy sites A and C, Y atom with fewer valence electrons prefers to occupy site B, main group atom Z prefers to occupy site D. The other is XA-type structure, in which two X atoms with fewer valence electrons prefer to occupy sites A and B, Y atom with higher valence electrons prefers to occupy site C, main group Z atom prefers to occupy site D. In our work, the main group atom Z is replaced by TM atom. The two crystal structures of Cd2MnTM (TM = Fe, Ni, Cu) are shown in Fig.
The calculated total energy as a function of lattice constant for Cd2MnTM (TM = Fe, Ni, Cu) is shown in Fig.
 | Fig. 2. Calculated total energy as a function of lattice constant for (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu with L21-type and XA-type structures in FM, AFM, and NM states, respectively. |
| Table 1. Crystal structure, equilibrium lattice constant (a), total and partial spin moments for Cd2MnTM (TM = Fe, Ni, Cu) at equilibrium lattice constant. . |
After geometric optimization, the band structures of L21-type Cd2MnTM (TM = Fe, Ni, Cu) alloys were calculated (see Fig.
 | Fig. 3. Calculated band structures of L21-type (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu at each equilibrium lattice constant. |
 | Fig. 4. Calculated total and partial density of states of L21-type (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu at each equilibrium lattice constant. |
Furthermore, the structure instability of Cd2MnFe comes from the sharp peak near EF due to the energy increasing of the system.[41] The structures of Cd2MnNi and Cd2MnCu are relatively stable duo to the valleys appear near EF based on Jahn–Teller effect,[42] the aspect of MTs will be discussed in the subsequent section. For the PDOS, the d-states of Cd locate below –7 eV and the p-states with low energy locate in the energy region from –4 eV to +2 eV. There are strong d–d orbital hybridizations between Mn and TM atoms from –4 eV to +2 eV. The hybridization between the p states of Cd and d states of other transition metal atoms can be expected. The Cd element acts as the main group element in the Heusler alloys, which is similar to Ga-based Heusler alloys.[43] From the Fig.
The total and atomic magnetic moments of L21-type (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu as a function of the lattice constant were calculated, as shown in Fig.
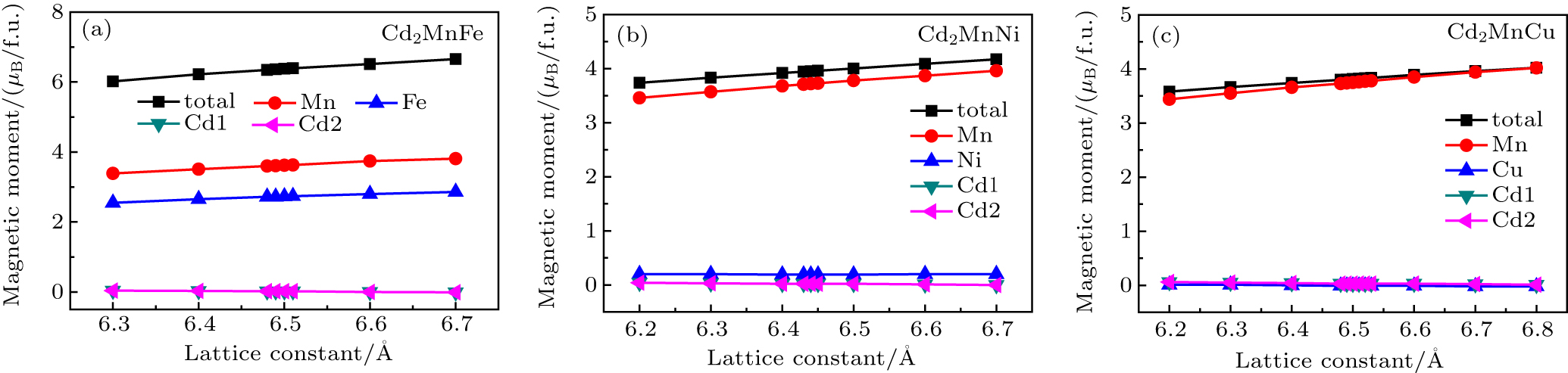 | Fig. 5. Calculated total and atomic magnetic moments of L21-type (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu as a function of the lattice constant. |
To study the possibility of the MTs in Cd2MnTM (TM = Fe, Ni, Cu) alloys, the energy difference (ΔE) between the tetragonal martensite and cubic austenite at the ground state as a function of c/a ratio (for c and a can be seen in Ref. [34]) with fixed crystal volumes, namely equilibrium cubic volume (Vopt) when c/a = 1, are given in Fig.
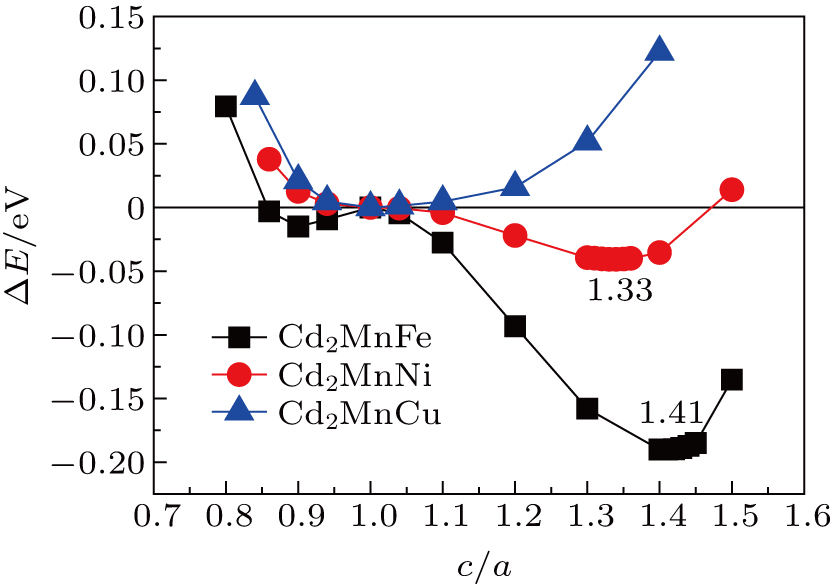 | Fig. 6. ΔE as a function of the c/a ratio for Cd2MnTM (TM = Fe, Ni, Cu). The zero point represents the energy of the austenite (c/a = 1). |
Considering the magnetic moments of Cd2MnTM (TM = Fe, Ni, Cu) with fixed volume, we carried out the total and atomic moments in the change of c/a. The band structures of Cd2MnFe with c/a = 1.41 and Cd2MnNi with c/a = 1.33 are shown in Figs.
 | Fig. 8. (a) Calculated band structure of Cd2MnFe with c/a = 1.41. (b) Calculated band structure of Cd2MnNi with c/a = 1.33. |
Figure
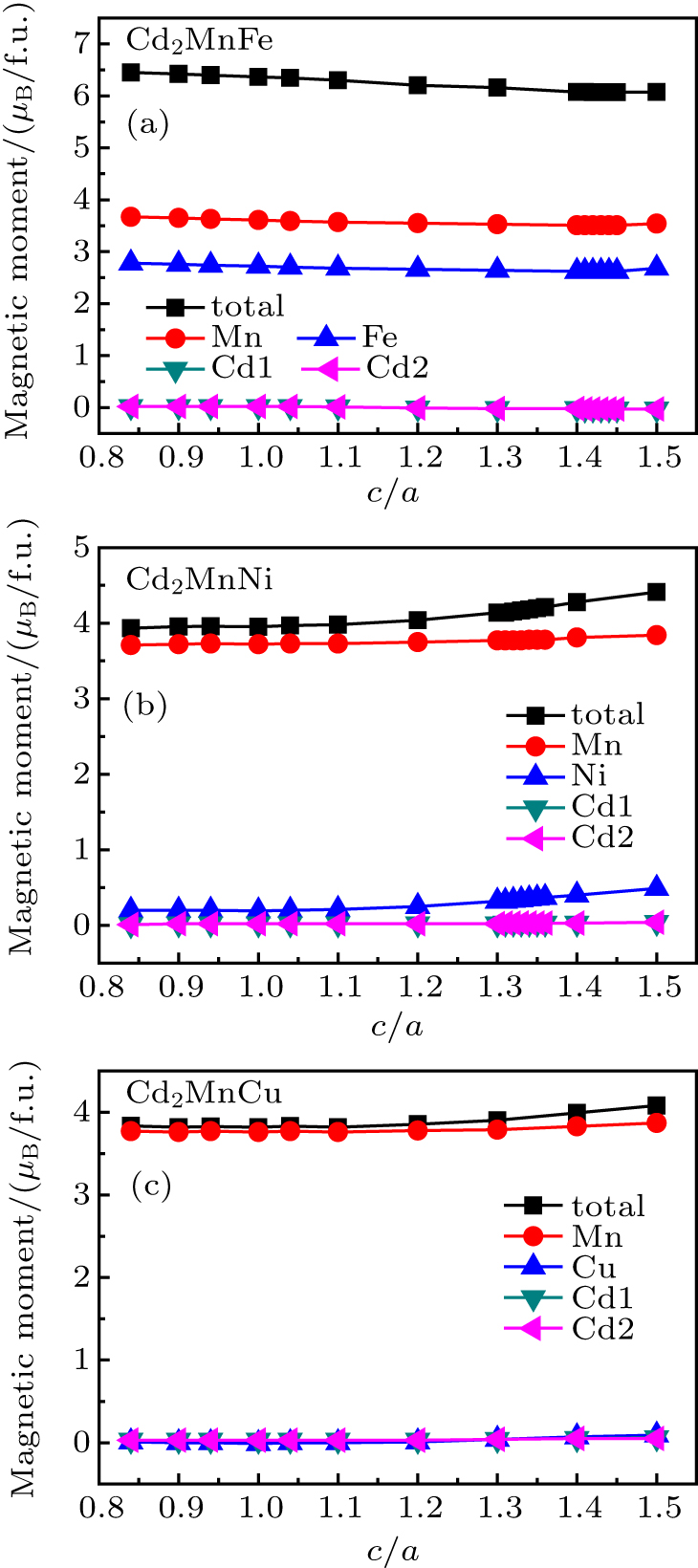 | Fig. 9. Calculated total and atomic magnetic moments of L21-type (a) Cd2MnFe, (b) Cd2MnNi, and (c) Cd2MnCu as a function of c/a. |
| Table 2. Energy difference ΔE between martensite and austenite, c/a ratio, total and partial magnetic moments of Cd2MnTM (TM = Fe, Ni) in their martensitic phases. . |
In summary, we have studied the electronic structures, magnetic properties, and martensitic transformation in all-d-metal Heusler-like alloys Cd2MnTM (TM = Fe, Ni, Cu) by first-principle calculations. The ferromagnetic L21-type cubic structure is the most stable phase. Applying site rule of Heusler alloys, the two Cd atoms prefer to occupy the equilibrium A, C sites, Mn and TM atoms prefer to occupy the equilibrium B and D sites, respectively. The total magnetic moment at equilibrium lattice constant of Cd2MnFe (6.36 μB) is larger than Cd2MnNi (3.95 μB) and Cd2MnCu (3.82 μB). The energy differences (ΔE) between martensite and austenite are negative in Cd2MnFe and Cd2MnNi under tetragonal distortion and different uniform strains, indicating the possible occurrence of the ferromagnetic martensitic transformation. The present theoretical investigation in Cd2MnTM alloys would provide some valuable information for exploring new all-d-metal Heusler alloys.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] |