† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11874328 and 11574276). The SXRD experiments were performed at the BL02B2 and BL04B2 of SPring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI; proposal Nos. 2019A1167, 2019A1095, and 2019A1340). We also acknowledge the help of Beamline Scientists Dr. Lirong Zheng (BSRF), Dr. Shogo Kawaguchi, and Dr. Koji Ohara (SPring-8).
Oxygen vacancies have a profound effect on the magnetic, electronic, and transport properties of transition metal oxides but little is known about their effect on thermal expansion. Herein we report the effect of oxygen defects on the structure formation and thermal expansion properties of the layered perovskite Ca2RuO4 (CRO). It is shown that the CRO containing excess oxygen crystallizes in a metallic L-CRO phase without structure transition from 100 K to 500 K and displays a normal thermal expansion behavior, whereas those with oxygen vacancies adopt at room temperature an insulating S-CRO phase and exhibit an enormous negative thermal expansion (NTE) from 100 K to about 360 K, from where they undergo a structure transition to a high temperature metallic L-CRO phase. Compared to the L-CRO containing excess oxygen, the S-CRO structure has increasingly large orthorhombic strain and distinctive in-plane distortion upon cooling. The in-plane distortion of the RuO6 octahedra reaches a maximum across 260 K and then relaxes monotonically, providing a structure evidence for the appearance of an antiferromagnetic orbital ordering in the paramagnetic phase and the Ag phonon mode suppression and phase flip across the same temperature found recently. Both the L- and S-CRO display an antiferromagnetic ordering at about 150–110 K, with ferromagnetic ordering components at lower temperature. The NTE in S-CRO is a result of a complex interplay among the spin, orbital, and lattice.
In recent years, the negative thermal expansion (NTE) of materials has received considerable attention due not only to its curious physics and chemistry behind but also to its potential implications for a variety of applications to meet the rapid advances in modern technology.[1–6] To date, the NTE could be driven by several mechanisms: (i) anharmonic phonon vibrations such as in framework oxides,[7–9] ReO3-type fluorides,[10] cyanides,[11,12], etc; (ii) magnetovolume effect such as in Invar alloys and manganese nitrides,[13,14] La(Fe, Si, Co)13,[15,16] and CrAs and Hf1−xTaxFe2 based compounds;[17,18] (iii) ferroelectric transition in PbTiO3 based materials;[19] and (iv) intermetallic charge transfer in SrCu3Fe4O12[20] and BiNiO3.[21] Oxygen vacancies have a profound effect on the magnetic, electronic, and transport properties of transition metal oxide materials and play a vital role in metal–insulating transition (MIT),[22,23] magnetoresistance,[24] catalysis,[25] fuel cells,[26] and supercapacitors.[27] Nevertheless, few studies about the effect of oxygen vacancies on the thermal expansion have been reported.[28,29]
Ca2RuO4 (CRO) is a prototype Mott insulator, where all of the degrees of freedom charge, spin, orbital, and lattice show robust interactions in distinctive phase transitions.[30–34] It undergoes a paramagnetic metal–insulator transition at TMI = 357 K, followed by a well-separated antiferromagnetic order at TN = 110 K.[30,35] The investigation of thermal expansion of CRO can be traced back to Braden et al.[30] and Friedt et al.[31] They observed an increase of lattice volume by 1% upon cooling from room temperature (RT) to 150 K for the stoichiometric crystalline CRO, whereas Alexander et al.[32] reported a strong decrease of lattice volume by 1.3% as the temperature was lowered from 400 K to 70 K for the single crystal CRO. Subsequently, Qi et al. found that M substituting for Ru produces a negative thermal expansion in Ca2Ru1−xMxO4 (M = Cr, Mn, Fe, or Cu).[36,37] Takenaka et al. reported a giant NTE for the reduced ruthenate and attributed it to microstructural effects consuming open spaces in the sintered body on heating.[29] However, the reason for the conflict results in lattice volume thermal expansion reported in crystalline CRO and single crystal CRO remains unclear and the understanding about the intrinsic role of oxygen vacancies played in the thermal expansion property and the MIT of the material is still lacking.
In this paper, we explore the effect of oxygen defects on the intrinsic thermal expansion properties in CRO. For this purpose, we prepared samples with different concentrations of oxygen defects by solid state reactions under different O2/Ar ratios and under ambient atmosphere. It is found that the type of oxygen defects plays a decisive role in the formation of the structure type of CRO, either in a metallic L-CRO or in an insulating S-CRO, and hence affects their temperature dependent behaviors such as structure evolution, thermal expansion, and electrical transportation. Only the S-CRO structure exhibits NTE from 100 K to about 360 K, where it transforms to a high temperature L-CRO phase. However, the two L-CRO phases with different types of oxygen defects are found to be different not only in lattice constant and RuO6 octahedral deformation but also in a series of physical properties. This work provides not only valuable information on the effect of the types of oxygen defects on the structural formation, evolution, and correlated changes in physical properties of CRO, but also a stage for future experimental and theoretical investigations to settle the debating on the nature of the Mott metal--insulator transition based on renewed models taking the oxygen vacancies into account.
The CRO samples containing different oxygen contents were prepared by a solid state reaction method with different O2/Ar ratios (0.5%, 1.0%, 1.5%, 2.0%) and in ambient atmosphere conditions. CaCO3 (purity 99%) and RuO2 (purity 99.9%) were used as raw materials which were dried and mixed with 2 : 1 molar ratio. The mixed powders were ground for approximate 2 h and then heated in a furnace at 1643 K for 24 h with a heating and cooling rate of 5 K/min. After cooling naturally, the powders were reground and pressed into cylinders with a diameter of 5 mm under 10 MPa. The cylinders were sintered at 1643 K for 48 h either in ambient atmosphere or in an Ar atmosphere with different percentage of oxygen. To avoid the reaction of the cylinders with the Al2O3 crucible, non-compressed powder of the same composition was used as the bedding powder.
The crystal structure was analyzed by high-resolution synchrotron radiation x-ray diffraction (SXRD, Japan, λ = 0.495724 Å). The powder XRD measurements at different temperatures were also performed with a Bruker D8 Advance x-ray diffractometer with Cu Kα radiation. The software used to analyze the XRD pattern was Fullprof. An AXIS ULTRA x-ray photoelectron spectroscopy (XPS, Shimadzu Kratos) was used to analyze the elemental composition and valence states in the samples. The XPS data analysis was performed by the fractional peak fitting using CasaXPS (v2.3.17).[38] The relative length changes of the bulk samples were measured by a LINSEIS DIL L76 dilatometer at a heating and cooling rate of 5 K/min. Temperature-dependent electrical resistivity and magnetic susceptibility were measured using a Quantum Design physical property measurement system.
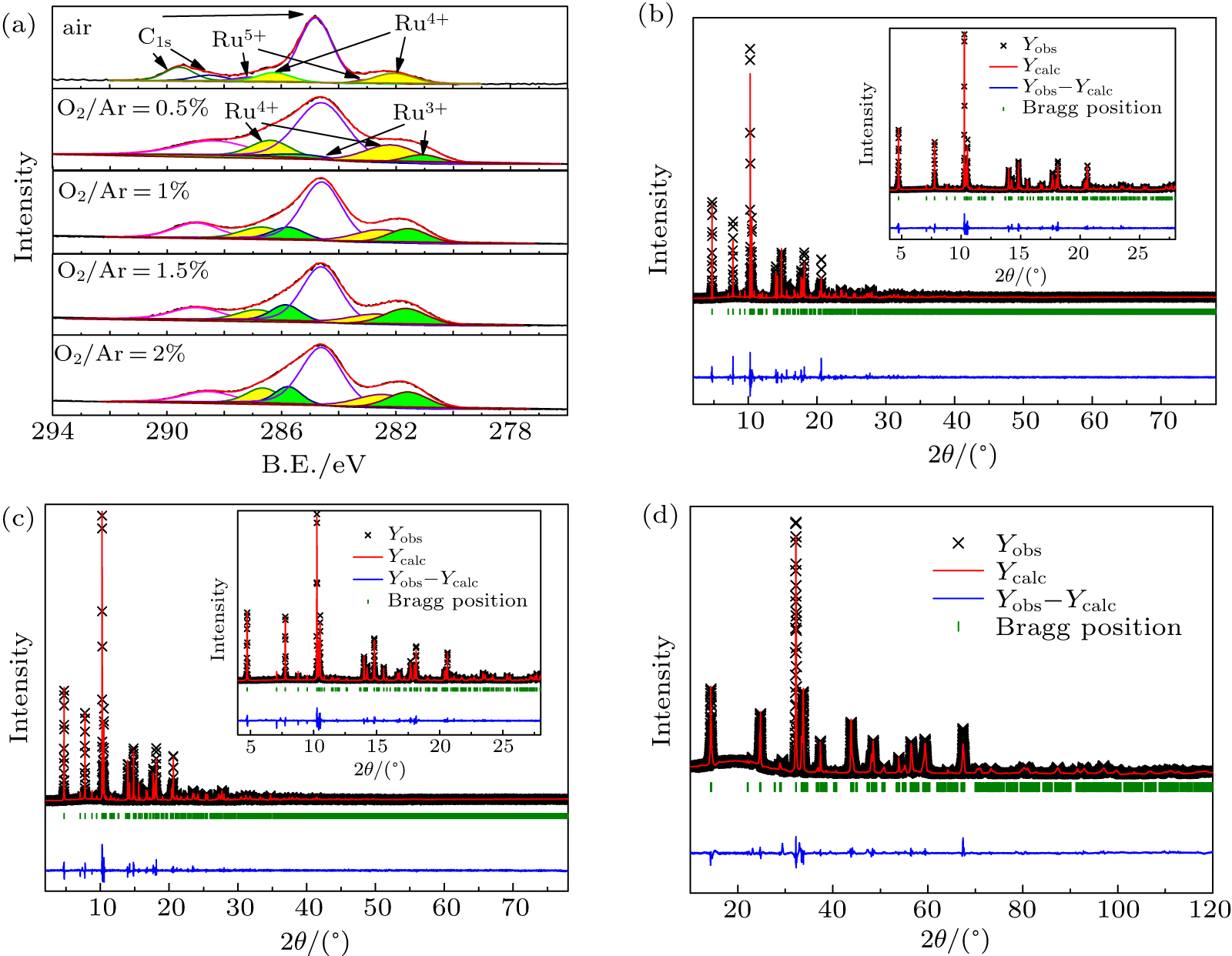
In order to determine the contents of oxygen defects in the samples prepared under different atmospheric conditions, we performed XPS analysis[39] for the Ru 3d orbital binding energies as shown in Fig.
The SXRD analyses for selected samples of Ca2RO4.09, Ca2RO3.87, and Ca2RO3.73 were performed to explore the effect of the oxygen vacancies on the structures as shown in Figs.
The above observations demonstrate that it is the oxygen content that governs the crystallization of CRO either in the S-Pbca or the L-Pbca phase, i.e., the types of oxygen defects play a vital role in the structure formation. It explains why the samples synthesized at different temperatures crystallized in different phases and the L-CRO phase could be obtained by annealing the powders of the S-CRO phase at 673 K for 50 h,[43] though the authors claimed the oxygen content of 4.0 per formula in both samples. In fact, the formation of the S-CRO or L-CRO phase could be very sensitive to the types of oxygen defects, even the contents could not be detected.
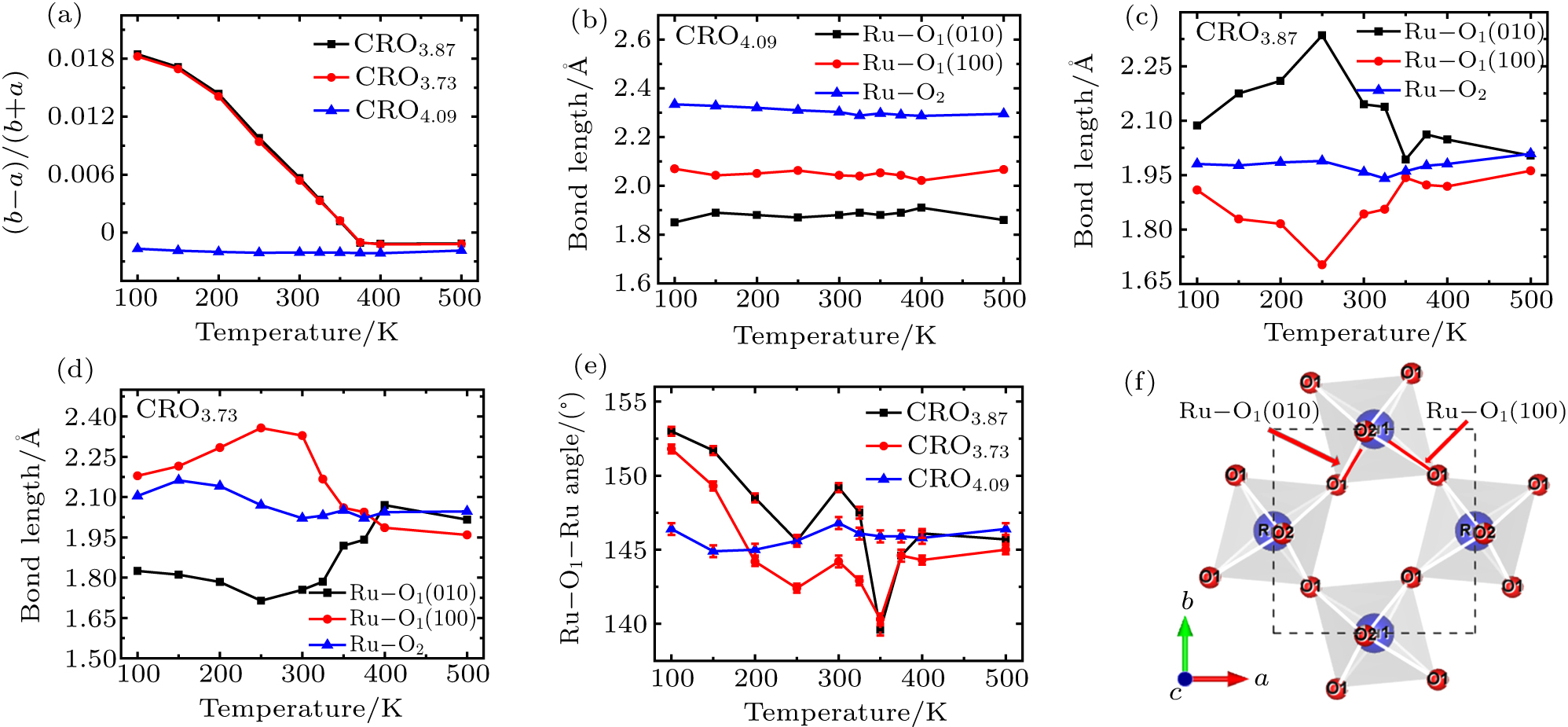
Though the samples with oxygen vacancies have a smaller c axis, but they have a larger lattice volume with respect to that containing excess oxygen. A large cell volume favors the appearance of NTE since it enhances the flexibility for the polyhedra movement. We show in Fig.
In order to see the effect of oxygen vacancies on the intrinsic thermal expansion properties, we carried out XRD measurements for the samples of Ca2RuO4.09, Ca2RuO3.87, and Ca2RuO3.73 from 100 K to 500 K and obtained the structure parameters by Rietveld refinements. Typical data at selected temperatures are presented in Table S3. Figures
| Table 1. Coefficients of axial and lattice volume expansion. . |
The a and b axes shrink concomitantly for the sample containing excess oxygen upon cooling (Fig.
In order to get insight into the effect of the octahedral distortion on the thermal expansion properties, we depict in Figs.
It is noted that an antiferromagnetic orbital ordering even in the paramagnetic phase below TOO = 260 K was suggested from resonant x-ray scattering experiments[44,45] and a strong suppression of oscillation-amplitude for an Ag symmetric phonon mode and a phase flip over 180° across the same temperature were observed by a recent optical pump–probe experiment.[46] We believe that the Ru–O(1) bond length and bond angle changes observed in Figs.
The excess oxygen in the interstitials will oversaturate the Ca coordination and lead to a charge transfer towards the RuO2 planes which requires oxidation of Ru higher than 4+ as evidenced by the XPS analysis. It consequently results in smaller Ru–O(1) and larger Ru–O(2) equilibrium distances and a relaxation of the strain implied by the misfit of smaller radius of Ca in the structure. At the same time, it also modulates the orbital polarization and the interlayer van der Waals interactions. As a result, it leads to the crystallization of the L-CaRO phase at RT with longer c axis and shorter a and b axes with respect to the counterparts of the S-CRO phase with oxygen deficiencies (a: 5.3112 Å vs. 5.4070 Å, b: 5.2990 Å vs. 5.4685 Å, c: 12.3770 Å vs. 11.9842 Å). The inserted oxygen atoms play a decisive role in the formation of the L-Pbca structure with lower orthorhombic strain at RT and in retaining the structure over a wide temperature range.
By contrast, an oxygen vacancy plays the role of double electron donors which requires the reduction of Ru lower than 4+ as revealed by the XPS analysis and results in a shorter Ru–O(2) equilibrium distance and a larger Ru–O(1) equilibrium bond length difference, making the RuO6 octahedra more distorted and flattened. It leads to the formation of S-CRO phase with much higher orthorhombic strain than that in the case with additional interstitial oxygen atoms. The expanding of the b axis in the S-CRO upon cooling is responsible for the lattice volume NTE, which is a complex interplay between the Ru–O(1) bond length and bond angle as well as the RuO6 octahedral rotation and tilting. However, the NTE mechanisms below and above 260 K may be not completely the same as indicated by the distinct changes of octahedral distortion across the temperature. The formation of the antiferromagnetic orbital ordering in the paramagnetic phase may make a contribution to the NTE below 260 K since the paramagnetic to antiferromagnetic phase transition is also accompanied by a volume expansion in Mn3AN upon cooling.[13,14]
Besides the enormous NTE, the samples with oxygen vacancies are also characterized by a distinct structure transition from the low-temperature orthorhombic S-CRO to a high-temperature L-CRO phase as indicated by the marked increase in the c axis around 350 K (Figs.
Figure
In Fig.
The types of oxygen defects play a decisive role in the formation of structure type in Ca2RuO4 and hence affect a series of their physical properties. The sample containing excess oxygen adopts the metallic L-CRO structure, whereas those with oxygen vacancies take the insulating S-CRO phase at RT. While the L-CRO structure containing excess oxygen displays a normal thermal expansion property without structure transformation from 100 K to 500 K, the S-CRO structure exhibits enormous NTE from 100 K to about 360 K, where it transforms to a high temperature metallic L-CRO phase which differs from its counterpart with excess oxygen in lattice parameters, RuO6 octahedral deformation, and a series of physical properties. Compared to the L-CRO with excess oxygen, the S-CRO has a distinctively larger in-plane RuO6 octahedral distortion which reaches the maximum across about 260 K and then relaxes monotonically as indicated by the progressive reduction of the in-plane bond length difference and extension of the bond angle. This might be taken as a structural evidence for the antiferromagnetic orbital ordering in the paramagnetic phase and the Ag mode suppression and phase flip found recently. All the samples display antiferromagnetic ordering at about 150–110 K, below which a ferromagnetic orbital ordering component appears in the antiferromagnetic phase. The NTE in S-CRO arises from a complex interplay among the spin, orbital, and lattice. In addition to the knowledge presented, this work might set up an stage for future experimental and theoretical investigations to settle the debating on the nature of Mott metal–insulator transition by taking oxygen vacancies in to account and prompt the exploring of NTE materials along the line of oxygen vacancies.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [53] |