† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 11474280).
This article reviews the research progress of measurement techniques and materials on the mechanocaloric effect over the past few decades. Mechanocaloric materials can be divided into elastocaloric and barocaloric materials depending on the applied uniaxial stress or hydrostatic pressure. Elastocaloric materials include non-magnetic shape memory alloys, polymers, and rare-earth compounds. Barocaloric materials include magnetic shape memory alloys, ferroelectric ceramics, superionic conductors, and oxyfluorides. The mechanocaloric effects of these classes of materials are systematically compared in terms of the isothermal entropy change and adiabatic temperature change. In addition to the thermal effects, other characteristics closely related to the application of mechanocaloric materials are also summarized. Finally, perspectives for further development of mechanocaloric materials in the solid-state cooling area are discussed.
Traditional refrigeration technology is represented by air conditioners and refrigerators, with are mainly driven by steam compression and liquid vaporization. The technology could cause ozone depletion and/or greenhouse effects. Because of environmental concerns, research on solid-state cooling as a next-generation refrigeration technology is expanding. Solid-state refrigeration technology is mainly related to three thermal effects that refer to the reversible thermal response of a given material when subjected to changes in an externally applied mechanical, electric, or magnetic field, namely the mechanocaloric effect (MCE), including the elastocaloric effect (ECE) and barocaloric effect (BCE); the electrocaloric effect (eCE); and the magnetocaloric effect (mCE). The advantages of solid-state refrigeration are that it is efficient and clean, with no emissions and easy recycling.
The search for mechanocaloric materials with giant caloric effect has become a major challenge for the development and application of mechanocaloric cooling equipment. Approximately 200 years ago, the mechanocaloric effect was first described by the natural philosopher John Gough.[1] The elastocaloric effect of various materials was reported by Joule in 1859.[2] In the last three decades, many mechanocaloric materials have been discovered. These mechanocaloric materials can be divided into various classes including alloys, magnetic materials, ceramics, organic–inorganic hybrid perovskites, polymers, oxyfluorides, superionic conductors, and plastic crystals. Of the vast array of mechanocaloric materials, which is a good refrigerant? On the basis of research on mechanocaloric materials, we identify the following main requirements that mechanocaloric materials should meet for solid-state cooling application: (i) good cooling efficiency under low pressure, namely, a large and reversible entropy or/and temperature change; (ii) adequate cooling temperature span; (iii) environmentally friendly; (iv) chemically stable with excellent reproducibility of caloric effects; (v) small hysteresis to rapid cooling cycles; (vi) economical for wide applications. These parameters affect the selection of a refrigerant. The temperature and entropy changes are important parameters, which reflect the cooling efficiency of a mechanocaloric material. It is more economical to use a refrigerant with high cooling efficiency. The cooling temperature span is closely related to the relative cooling power (RCP): RCP = |ΔSmax |δ TFWHM, where ΔSmax is the maximum of entropy changes and is the full width at half maximum (FWHM) of the ΔS versus T curves. An appropriate pressure can increase the cooling efficiency for many mechanocaloric materials if it can be achieved and the materials can support it. In addition, some mechanocaloric materials contain toxic elements that may damage the environment. The refrigerant must have good reproducibility for the long-term use. Furthermore, the stability of the refrigerant, including its thermal and chemical stability, must be considered in designing cooling equipment. Hysteresis restrains the work frequency and cooling capacity. Economical mechanocaloric materials are beneficial for widespread applications. Although there have been many review papers on solid-state cooling materials,[3–6] a review that summarizes and compares the various mechanocaloric materials from the application viewpoint, especially for recent materials with colossal mechanocaloric effects, is lacking. The present paper aims to introduce measurement methods, summarize recent progress, and discuss further development in this area.
The two typical quantities used to characterize the MCE are the isothermal entropy change (ΔS) and adiabatic temperature change (ΔT). The measurement methods can be broadly classified into three types: direct, quasi-direct, and indirect methods.[3] Different methods can be used to obtain different values of ΔS and/or ΔT for the same material. For example, indirect methods usually overestimate ΔS, and direct methods usually underestimate ΔT because of heat leakage.
The direct methods are simple and direct with the temperature determined using a thermocouple or non-contact infrared thermometry. The methods require adiabatic condition to obtain an accurate ΔT. When the speed of the external field changes is much faster than the heat exchange rate between the sample and environment, an adiabatic condition can be considered to be achieved. For example, for foil samples exposed to air in elastocaloric experiments, the adiabatic requirement is met if the strain rate is larger than 0.2 s−1.[7] In barocaloric experiments, the temperature change measured by direct methods is usually underestimated because the pressure medium around the samples leads to a heat leak. For direct measurement of isothermal entropy changes, there is currently no suitable device.
For the quasi-direct methods, ΔS can be determined by measuring the heat capacity or heat flux under high pressure. Differential scanning calorimetry (DSC) under high pressure is the most suitable technique for BCE studies of first-order phase transitions. DSC equipment provides hydrostatic pressure, which is generated by gas such as oxygen or nitrogen. For elastocaloric studies, currently, no suitable calorimetric device can work under uniaxial external loads. When a sample is on a heating run in DSC, the specific heat CL of the low-temperature phase can be measured. Then, the temperature increases to the initial temperature of phase transition (T1). The phase transition occurs, and the specific heat C can be determined. With continuous heating, the temperature reaches the final temperature of phase transition (T2). The specific heat CH of the high-temperature phase can be determined when the temperature is above T2, and we suppose that CL and CH are constant under different pressures. Then, the entropy (S), which refers to the value at a given temperature T0 for a heating run can be computed as[5]
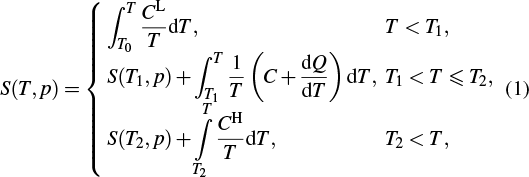

The entropy changes measured by quasi-direct methods (ΔSqd) can be expressed as
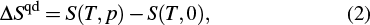
The temperature changes measured by quasi-direct methods (ΔTqd) can be expressed as

The indirect methods are based on the Maxwell relations or Clausius–Clapeyron equation. For the BCE, the volume changes of the unit cell can be determined using x-ray diffraction under high pressure and different temperatures. However, as for the ECE, the stress(σ)–strain(ε) curves of elastocaloric materials can be obtained using suitable strain gauges. The equation of entropy changes can be expressed as follows:
Elastocaloric effects:

Barocaloric effects:

Mechanocaloric materials can be classified into the following types of materials depending on their physical characteristics: non-magnetic shape memory alloys (non-MSMAs), magnetic shape memory alloys (MSMAs), magnetic materials, perovskite materials, oxyfluorides, polymers, superionic conductors, and plastic crystals.
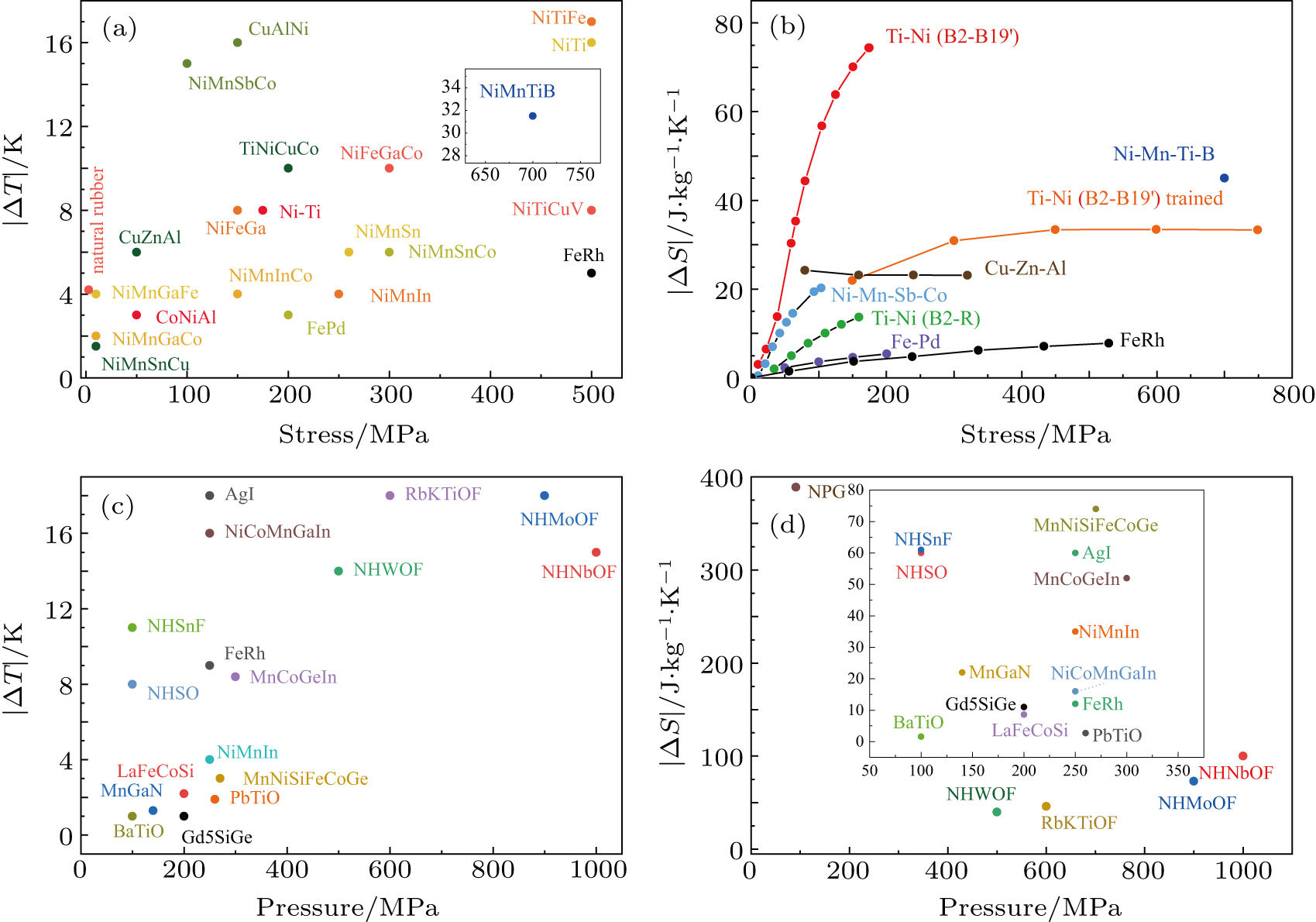
Shape memory alloys (SMAs) have many unique thermomechanical characteristics such as the shape memory effect, pseudoelasticity, and superelasticity. These alloys can ‘memorize’ their deformation and return to their original form when subjected to thermomechanical or magnetic variations. The non-MSMAs under uniaxial stress undergoes a reversible martensitic transition, which is a first-order structural transition. Because of the large latent heat of the transition, these alloys exhibit the MCE. The lattice distortion associated with the transition is mostly caused by a pure shear. The transition temperature and the MCE are strongly sensitive to uniaxial stress but insensitive to hydrostatic pressure.[8] Recently, research on the MCE of non-MSMAs has focused on Cu-based and Ti–Ni-based alloys. The thermal effect in single crystals of β-CuZnSn was reported in 1981.[9] Later experiments[10–13] have proved that Cu-based alloys such as Cu–Zn–Al single crystal have the MCE. ΔS and ΔT for Cu-based alloys can exceed 20 J·kg−1·K−1 and 14 K, respectively. Because of the large value of ΔS and broad temperature span, the Cu–Zn–Al polycrystal has an enormous relative cooling power for the ECE.[11] Another important class of non-MSMAs is based on Ti–Ni.[7,14–22] These alloys transform from a B2 (Pm2m) to a B19′ (P2/m) martensitic phase upon cooling.[8] The reversible B2→ B19′ transition has a large latent heat, and the transition entropy change is quite high (in the range of 60 J·kg−1·K−1–80 J·kg−1·K−1) at moderate stress (∼ 170 MPa).[19] ΔT measured by direct methods can reach 25 K for Ni–Ti wires.[16,17] In addition, the cooling performance of Ni–Ti-based alloys has been studied. One study[7] revealed that Ni–Ti wires have a quite high cooling efficiency. Furthermore, the values of temperature changes for sputtered NiTi films, especially sputtered NiTiCu films, have good reproducibility even though the alloys have been trained for many cycles.[15,16] Recently, the ECE caused by the rubber-like behavior of the R-phase nanocrystalline wires of Ti–50.8Ni (at.%) has been reported. Because of the small energy dissipation, the ECE related to the R-phase is more efficient than that induced by the B2 → B19′ transition.[23] The thermodynamic characteristics are compared for selected non-MSMAs in Table
| Table 1. Thermodynamic characteristics for elastocaloric effect compounds. Temperature for the forward (cooling) transition (Tt); transition temperature shift with uniaxial stress (dT/dσ); applied stress (Δσ) to obtain the temperature and entropy changes in this table; adiabatic temperature change measured using direct methods (ΔTd) and estimated from the measured isothermal entropy change (ΔTe); isothermal entropy change determined using indirect methods (ΔSi) and estimated from measured temperature changes (ΔSe). . |
Magnetic shape memory alloys (MSMAs) are interesting materials with mechano-caloric or multi-caloric effects. In MSMAs, the caloric effect is usually induced by a magnetostructural phase transition. Many characteristics of MSMAs are similar to those of non-MSMAs such as the martensitic transition and shape memory effects; however, the structural transition in MSMAs is accompanied by changes in the magnetism from the antiferromagnetic (AFM) state to the ferromagnetic (FM) state. MSMAs can be divided into two categories: Fe-based alloys, such as Fe–Pt and Fe–Pd, and Ni-based and some Co-based alloys, which are usually Heusler alloys. For MSMAs, the high-temperature phase is not martensitic but cubic and FM. As for the magnetism of the martensitic phase, it depends on whether the martensitic transition temperature is above or below the Curie temperature Tc in a specific material. The martensitic and magnetic transition in Fe-based MSMAs such as Fe–Pt and Fe–Pd were studied many years ago.[24–26] The martensitic transition from a cubic structure (Fm3m) to a face-centered tetragonal martensitic structure (P4/mmm) occurs on cooling for Fe-based MSMAs. In Fe–Pd alloys, the martensitic transition is weak first-order with a low entropy and small transformation hysteresis.[26] Because of the small volume changes of the unit cell in this transition, the BCE is not expected. The ECE was studied in Fe–Pd single crystal[26] and Fe7Pd3 polycrystal.[25] In Fe–31.2Pd (at.%) alloy, a significant ECE was reported. Because of lattice softening caused by a weak first-order martensitic transition near room temperature, an adiabatic temperature change of more than 3 K and ΔS of 4 J·kg−1·K−1–5 J·kg−1·K−1 with excellent reproducibility were observed under uniaxial stress of 200 MPa in this alloy. Apart from Fe-based MSMAs, the MCE for Ni-based MSMAs belonging to Heusler alloys has been heavily investigated.[14,27–43] Early in their study, these investigated alloys were usually very brittle and could only support very low stresses (≈ 10 MPa).[42,43] Later, tougher alloys were synthesized using suitable doping or other methods with the maximum applied stresses exceeding 300 MPa. The magnitude of the MCE in these MSMAs depends on the sensitivity of the martensitic transition to hydrostatic pressure or uniaxial stress. The MCE was observed in Ni-based magnetic shape memory alloys and can be compared with giant magnetocaloric effects. Upon uniaxial compression of 100 MPa, the stress-induced entropy change ΔS is 21 J·kg−1·K−1 and the estimated ΔT is approximately 15 K in polycrystalline Ni–Mn–Sb–Co MSMA. The result is better than other reports on the ECE in Ni-based MSMAs.[24,29,39] For applied hydrostatic pressure, the BCE for Ni–Mn–In MSMA resulted from discontinuities of both the volume of the unit cell and entropy at a magnetostructural transition. This alloy exhibited both barocaloric and magnetocaloric effects. Under hydrostatic pressure of 260 MPa, ΔT can reach 4.5 K and ΔS is approximately 24 J·kg−1·K−1. However, when a magnetic field of 0.94 T is applied, the change of temperature for mCE is only 1.3 K.[27] It is worth noting that the peak values of ΔT and ΔS increase as pressure increases in the range of 20 MPa–260 MPa. These studies suggest that some magnetocaloric materials could exhibit the BCE. Thermodynamic characteristics are compared for selected MSMAs in Tables
| Table 2. Thermodynamic characteristics for the barocaloric effect compounds. . |
Some magnetic materials also exhibit the MCE because of a first-order magnetostructural transition. The transition can cause changes in the symmetry of the crystal structure or just a uniform expansion or contraction of the unit cell. These compounds, which do not involve changes in the symmetry of the crystal structure, mainly include Fe–Rh, La–Fe–Si, and Mn–Ga–N compounds. The ECE in Fe–Rh alloys was reported early on.[44] The magnetic transformation from AFM to FM is accompanied by a negative temperature change of 5.2 K under stress of 529 MPa. Later, the BCE with good reproducibility in Fe–Rh alloys was observed.[45,46] Large values of |ΔT| ∼ 8 K–10 K and |ΔS| ∼ 12 J·kg−1·K−1 were reported under hydrostatic pressure (250 MPa). Another class of these compounds is the La–Fe–Si-based itinerant ferromagnets. The LaFe13 – xSix compounds have the cubic NaZn13 structure (

Perovskite materials have the same crystal structure as calcium titanium oxide, known as the perovskite structure. The general chemical formula for perovskite materials is ABX3, where A and B are two cations of very different sizes and X is an anion that bonds to both A and B.[55] For cubic structure stability, the requirement of relative ion size is very stringent. Therefore, slight distortion and deformation can result in several lower-symmetry structures, in which the coordination number of A or B cations or both is reduced. These characteristics lead to a rich phase diagram in perovskite materials. Furthermore, many perovskite ceramics such as BaTiO3 exhibit ferroelectricity because the complex structure produces electric dipoles. The perovskite materials mentioned in this section are also ferroelectric materials, which were investigated as eCE materials. Because of the strong coupling between polarization and structure, ferroic phase transitions may occur in perovskite materials. Therefore, the MCE is expected in perovskite materials. The MCE of perovskite materials such as BaTiO3, (Ba0.85Ca0.15)(Zr0.1Ti0.9)O3, and PbTiO3 has been investigated.[56–64] At high temperature (> 400 K), BaTiO3 has a centrosymmetric ABO3 perovskite crystal structure (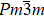
As special perovskites, organic–inorganic hybrid perovskites exhibit pressure-induced solid-state cooling effects. This family of materials has the perovskite structure ABX3, where A is an alkylammonium cation, B is a transition metal cation, and X is a halide or polyatomic bidentate-bridge ligand. A prior review summarized the BCE in more than a dozen hybrid perovskites.[65] These hybrid perovskites have the following characteristics: (i) a large and reversible entropy change, (ii) a large shift of transition temperature with pressure, and (iii) an adequate transition temperature. A typical example of hybrid perovskites is [TPrA]Mn[dca]3 (TPrA = (CH3CH2CH2)4N+, dca = [N(CN)2]−).[66] This material exhibits giant BCEs near room temperature under a pressure of 70 bar (1 bar = 105 Pa).
Unlike the above MCE materials, the MCE materials discussed in this section are amorphous. This type of material can support a dramatic reversible elongation or retraction. The huge deformation results in a huge entropy change. In fact, the first reported material with the ECE is Indian rubber, a randomly oriented polymer.[1] However, this material was not valued until recently.[67–70] In natural rubber, the highest temperature change for the ECE is 13.7 K at 674% elongation and the estimated ΔS is 67 J·kg−1 K−1. There are many polymers with the MCE based on ferroelectric polymers such as polyvinylidene fluoride (PVDF).[71–74] According to results of x-ray diffraction experiments, a crystal transformation from the anti-polar α to the polar β phase was observed in PVDF terpolymers. The transformation can be induced by pressure or an electric field. The elastocaloric properties of PVDF trifluoroethylene chlorotrifluoroethylene (PVDF–TrEE–CTFE) were reported using an infrared imaging camera. ΔT of 2.15 K and ΔS of 11 J·kg−1·K−1 were measured at a strain of 12%. For the BCE, ΔT of 6 K in this polymer was observed under a pressure of 200 MPa at room temperature and increased to approximately 19 K at the phase transition temperature of 368 K.
The MCE of rare-earth compounds as solid-state cooling materials was studied. Because of their 4f electron hybrids, most rare-earth materials have rich magnetic properties that are sensitive to pressure. Some rare-earth compounds such as Cex(La, Y)1 − xSb, HoAs, Ce3Pd20Ge6, and EuNi2(Si1-xGex)2 have been reported as MCE materials.[75–78] In these compounds, the entropy changes must take into account that the crystalline electric field splits the (2J + 1)-fold degenerated ground-state J-multiplet of the rare-earth ion into discrete energy levels. In Cex(La, Y)1 − xSb, the PM–AFM transition induced by uniaxial stress was expected to result in a huge entropy change. The in situ experiments revealed ΔT of 0.42 K upon releasing a uniaxial stress of 240 MPa in Ce0.85(La0.95Y0.05)0.15Sb and ΔT of 2 K upon releasing a uniaxial stress of 500 MPa in CeSb at low temperature (∼ 20 K). The monopnictide HoAs exhibits the ECE resulting from a magnetic–structural phase transition. ΔT for the ECE is over 0.4 K upon the application of uniaxial stress of 300 MPa along the [001] direction. The third material introduced is a heavy-fermion system Ce3Pd20Ge6. The |ΔT| of 0.75 K for the ECE was obtained in the Kondo lattice compound Ce3Pd20Ge6 because release of the uniaxial stress of 300 MPa leads to a positive shift of the Kondo temperature and splitting of the ground state at 4.4 K. The rare-earth compound EuNi2(Si1 − xGex)2 undergoes a first-order valence transition with a thermal hysteresis of 10 K and a huge entropy change from an Eu2+ state toward an Eu3+ state near 42 K. The sharp valence transition is expected to lead to the BCE.
In the year 2010, the MCE in a series of oxyfluorides was reported. These oxyfluorides have four types, namely (NH4)2NbOF5, (NH4)2WOF4, (NH4)2MoO2F4, and Rb2KTiOF5.[79–83] The ammonia compound (NH4)2NbOF5 undergoes two phase transitions accompanied by entropy changes. For (NH4)2NbOF5, the disordered phase I transforms into the partially ordered phase II on cooling at 258 K and then transforms to a fully ordered phase III at 220 K.[84] The strengths of the BCE and ECE are almost equal. The maximum of ΔT and ΔS for the MCE can reach 15 K and 60 J·kg−1·K−1, respectively, upon the application of hydrostatic pressure or uniaxial stress of 1 GPa near the phase transition temperatures. Later, the MCE in (NH4)2WOF4 and (NH4)2MoO2F4 oxyfluorides was studied. Two phase transitions were reported at 271.4 K and 180 K for the molybdate and at 201.5 K and 161 K for the tungstate. ΔT of 10 K and ΔS of 40 J·kg−1·K−1 for the molybdate and ΔT of 12 K and ΔS of 50 J·kg−1·K−1 for the tungstate were calculated using differential thermal analysis under a force field of 0.5 GPa near the first-order structural phase transitions. Later, the MCE in Rb2KTiOF5 was observed near the structural phase transition at 215 K under pressure between 0 and 0.6 GPa. The Rb2KTiOF5 undergoes a first-order ferroelastic phase transition that can result in huge entropy changes. ΔT of 16 K and ΔS of 43 J·kg−1·K−1 were calculated from the differential thermal analysis under a pressure of 0.4 GPa. In 2016, a review of the MCE in ferroelastic fluorides and oxyfluorides was published.[83] This paper reviewed the effects of uniaxial stress and hydrostatic pressure on phase transitions, the MCE, and the experiment methods used to obtain the temperature–pressure phase diagram and entropy under high pressure. The thermodynamic characteristics of selected materials are compared in Table
Superionic conductors, also known as fast ionic conductors, are a class of solid materials with extremely high ionic conductivity.[85] According to recent studies of the BCE in fluoride-based and AgI superionic conductors, superionic conductors are expected to be good candidates for MCE materials.[86,87] The superionic transition in fluorite-structured fast ion conductors such as CaF2 and PbF2 results in a large entropy change of over 100 J·kg−1·K−1 under hydrostatic pressure or uniaxial stress. |ΔT| of 20 K in PbF2 and 3 K in CaF2 were calculated using density functional theory upon releasing the stress of 0.6 GPa. When the hydrostatic pressure increases to 5 GPa, calculations for the BCE in CaF2 indicate that |ΔT| and |ΔS| can reach 152 K and 186 J·kg−1·K−1, respectively. Other materials with giant MECs include AgI, which consists of α, β, and γ phases at different temperatures. The structure of the α phase is a body-centered-cubic structure (
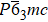

In 2019, colossal BCEs were observed in plastic crystals.[88,89] Plastic crystals, also known as orientation-disordered crystals, are oriented randomly, whereas the mass center forms symmetric lattices. The reversible first-order transition induced by pressure in plastic crystals induces large amounts of heat changes. In plastic crystals such as 2,2-dimethyl-1,3-propanediol (NPG), 2-methyl-2-nitro-1-propanol (MNP), and pentaglycerin (PG), the solid-state transition enthalpy is equivalent to or even higher than the melting enthalpy of some common solids such as Fe, Ti, and Mn. For the BCE in NPG, MNP, and PG, the maximum of |ΔS| calculated by indirect methods can reach 389, 404, and 493 J·kg−1·K−1, respectively. The value of entropy changes in plastic materials is dozens of times as large as that in previous materials with the MCE. The microscopic origin of colossal BCEs in plastic crystals was revealed through high-resolution quasi-elastic neutron scattering and inelastic neutron scattering experiments under high pressure. These experiments verified that orientational disorder in plastic crystals is restrained by the pressure. Thus, the transition results in huge entropy changes.[88] Plastic crystals have excellent cooling efficiency; however, the organic part leads to quite large hysteresis.
To date, the MCE has been investigated in SMAs including non-magnetic SMAs and MSMAs, ferroelectric perovskite materials, polymers, rare-earth compounds, oxyfluorides, superionic conductors, plastic crystals, and other materials. Here, we list thermodynamic characteristics for the elastocaloric effect (in Table
Figure
In conclusion, to boost the application of mechanocaloric materials, we reviewed the measurement techniques for the mechanocaloric effect and reviewed mechanocaloric materials. From an application viewpoint, we propose requirements that a good refrigerant should meet. Through analysis of the mechanocaloric effect in various mechanocaloric materials, we summarize and compare the characteristics of these materials. Finally, we suggest that a material with the disorder–order transition and small hysteresis can exhibit a more significant caloric effect and is more suitable for application in solid-state cooling.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [52] | |
| [53] | |
| [54] | |
| [55] | |
| [56] | |
| [57] | |
| [58] | |
| [59] | |
| [60] | |
| [61] | |
| [62] | |
| [63] | |
| [64] | |
| [65] | |
| [66] | |
| [67] | |
| [68] | |
| [69] | |
| [70] | |
| [71] | |
| [72] | |
| [73] | |
| [74] | |
| [75] | |
| [76] | |
| [77] | |
| [78] | |
| [79] | |
| [80] | |
| [81] | |
| [82] | |
| [83] | |
| [84] | |
| [85] | |
| [86] | |
| [87] | |
| [88] | |
| [89] | |
| [90] | |
| [91] | |
| [92] | |
| [93] | |
| [94] | |
| [95] | |
| [96] | |
| [97] | |
| [98] | |
| [99] |