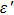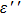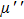† Corresponding author. E-mail:
Large-area and flexible reduced graphene oxide (rGO)/Fe3O4 NPs/polyurethane (PU) composite films are fabricated by a facile solution-processable method. The monolayer assembly of Fe3O4 nanoparticles with a high particle-stacking density on the graphene oxide (GO) sheets is achieved by mixing two immiscible solutions of Fe3O4 nanoparticles in hexane and GO in dimethylformide (DMF) by a mild sonication. The x-ray diffraction and Raman spectrum confirm the reduced process of rGO by a simple thermal treatment. The permittivity value of the composite in a frequency range of 0.1 GHz–18 GHz increases with annealing temperature of GO increasing. For 5-wt% rGO/Fe3O4 NPs/PU, the maximum refection loss (RL) of over −35 dB appears at 4.5 GHz when the thickness of film increases to 5 mm. The rGO/Fe3O4 NPs/PU film, exhibiting good electromagnetic properties over GHz frequency range, could be a potential candidate as one of microwave absorption materials in flexible electronic devices.
In recent years, with the rapid development of the modern communication technologies, especially 5G wireless communication, electronic components such as miniature filters, integrated inductor, microchip antenna, and electromagnetic interference (EMI) devices have been required to have the characteristics such as miniaturization, high-speed, low energy-consumption, good electromagnetic wave absorption, and flexibility.[1–5] There is a growing demand for the magnetic materials with high resonance frequency (fr), large permeability (μ), and suitable magnetic loss in a GHz frequency range to be used as inductor cores, antenna substrates, and a wide range of other microwave devices in size reduction and minimizing the electromagnetic energy trapped in the devices.[6] For example, magnetic materials can be used to reduce the volume of microchip antennas because it is roughly inversely proportional to the refractive index 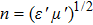
In addition, carbon nanotube (CNT)- and graphene-based composites have become new candidates for electromagnetic functional materials including microwave absorbing materials.[10,11] Generally, the enhanced microwave absorption of these materials can be mainly attributed to the following aspects: the residual defects on the surface of the graphene oxide (GO) sheets, the large aspect ratio and high conductivity, interfacial polarizations in these composites. Good EM absorption properties of reduced graphene oxide (rGO) or its related materials have been demonstrated. Zhang et al. have reported that an ultralight and highly compressible graphene foam has a broadband and tunable high-performance microwave absorption property.[12] Chen et al. have reported that novel reduced GO (RGO) composites with hematite nanoparticles embedded in RGO layers and wrapped by RGO sheets exhibited remarkably improved electromagnetic absorbing performance.[13] Many other composites, such as rGO/α-Fe2O3,[14] PVDF/GO,[15] and rGO/ZnO hollow spheres[16] have been reported as EM absorption materials. Though they usually have good EM absorption properties, poor dispersity and stability, and large loading content of GO or rGO, are the severe drawbacks to impede their practical applications. In addition, most of researchers use paraffin as matrix to measure the EM absorption, which cannot provide the practical EM parameters in the electronic devices to demonstrate their real EM absorption properties.
Here in this work, we prepare monodisperse Fe3O4 nanoparticles (NPs) and fabricate large-area rGO/Fe3O4 NPs/PU flexible film by using a facile solution-processable method. Fe3O4 NPs has recently attracted much attention due to its high Curie temperature (

The monodispersive Fe3O4 NPs were synthesized on a gram scale according to our reported method.[23] In a typical experiment for 7-nm NPs, 0.528 g (6 mmol) of 
GO was synthesized from natural graphite powder by a modified Hummers method.[24,25] Exfoliation was carried out by ultrasonicating the GO dispersion under ambient condition. The as-prepared GO was dispersed in DMF solution. The general procedure to assemble Fe3O4 NPs on GO sheets is adopted as follows: 15-mg Fe3O4 NPs were dispersed in 20-mL hexane and added into 20-mL DMF solution containing GO (0.5 mg/mL), and then the resulting mixture was dramatically stirred for 5 min and sonicated for another 1 h. After adding 10-mL ethanol, the suspension was centrifuged at 9500 rpm for 10 min to separate the GO/Fe3O4 NPs from mixed solvents. The GO/Fe3O4 NPs product was annealed at different temperature under Ar flow to obtain rGO/Fe3O4 NPs. For the preparation of rGO/Fe3O4 NPs/PU film, PU (5 g) was dissolved in DMF (25 mL) at 50 °C under a dramatic mechanical stirring. In another baker, 25 mL of a 10-mg/mL rGO/Fe3O4 NPs suspension in DMF was ultrasonicated for 30 min. Then, the black rGO/Fe3O4 NPs suspension was slowly added into the PU solution and sonicated for another 30 min. Then, the homogeneous rGO/Fe3O4 NPs/PU solution was scraped into a smooth film by adjusting its viscosity. The film was dried in vacuum drying box at 60 °C until no weight lost.
The x-ray diffraction (XRD) data were collected on a D2 PHASER x-ray diffractometer (Cu 


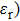
Figure
 | Figure 1. TEM images of (a) GO sheet, with inset showing SAED pattern; (b) monodisperse Fe3O4 NPs; (c) low- and (d) high-stacking-density monolayer assembly of Fe3O4 NPs on GO nanosheet. |
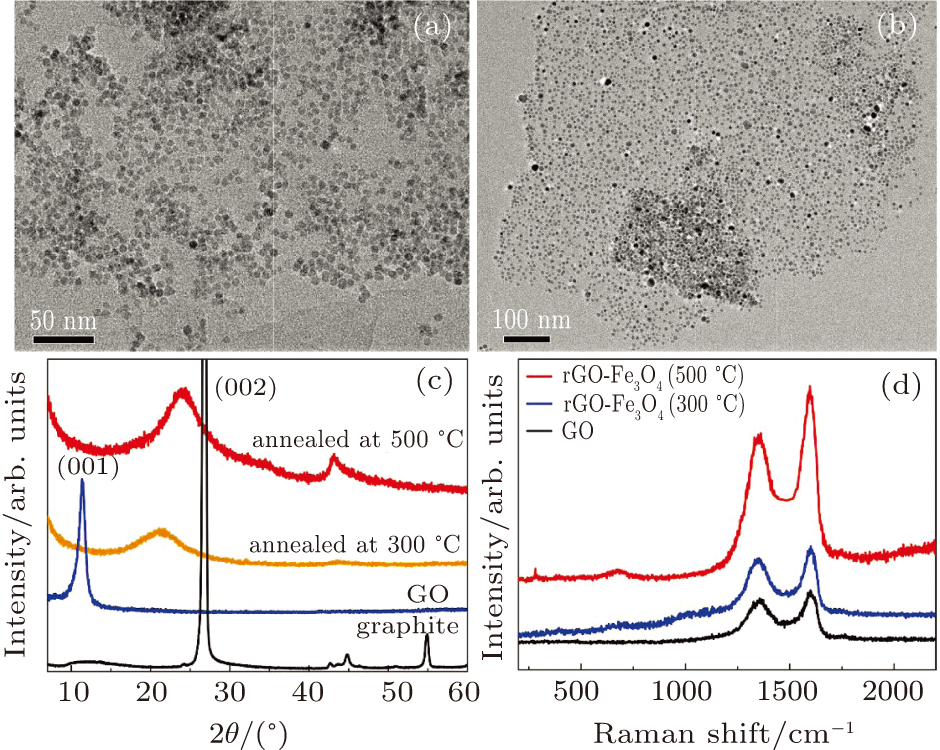
To improve the interface binding strength and permittivity properties, GO/Fe3O4 NPs are annealed at moderate temperatures to form rGO / Fe3O4 NPs. If being annealed at above 600 °C, Fe3O4 NPs will aggregate and result in a wide size distribution. As shown in Figs. 

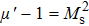


Figure
 | Figure 3. Pictures of (a) 15-cm-long rGO/Fe3O4 NPs/PU film, (b) our home-made scraping film facility, and (c) freely folding rGO/Fe3O4 NPs/PU film. |
Figures 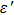

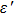




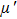


According to the transmission line theory, for a single layer absorber with a backed metal plate, the refection loss (RL) curves are simulated from the electromagnetic parameters at various sample thickness values by means of the following expressions[31,32]
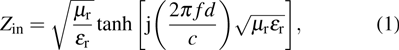 |
 |
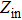
| Table 1.
Comparison of EM absorption properties of rGO/Fe3O4 NPs/PU film with other reported results. . |
In this work, monodisperse magnetic Fe3O4 NPs are prepared by a facile high-temperature organic solution method and loaded on the GO with a high monolayer stacking density. A simple and robust solution-processable method is used for preparing the rGO/Fe3O4 NPs/PU flexible films. The rGO and PU can form a strong hydrogen bonding due to a lot of hydroxy groups, which results in a well-dispersed rGO in PU instead of agglomeration or restack. The permittivity value of the composites in a frequency range 0.1 GHz–18 GHz increases with annealing temperatures of GO increasing. For 5-wt% rGO/Fe3O4 NPs/PU, the maximum RL of over −35 dB appears at 4.5 GHz when the thickness of film increases to 5 mm. The main microwave absorption mechanism of rGO/Fe3O4 NPs/PU film is dielectric loss produced by relaxation process that includes interfacial polarization and electronic dipole polarization between rGO and PU, and good impedance match from the introduce of permeability of Fe3O4 NPs. Such an rGO/Fe3O4 NPs/PU flexible film can be used as effective microwave absorption material in the future flexible electronic devices.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] |