† Corresponding author. E-mail:
A highly reliable and selective ethanol gas sensor working in realistic environments based on alpha-Fe2O3 (α-Fe2O3) nanorhombs is developed. The sensor is fabricated by integrating α-Fe2O3 nanorhombs onto a low power microheater based on micro-electro-mechanical systems (MEMS) technology. The α-Fe2O3 nanorhombs, prepared via a solvothermal method, is characterized by transmission electron microscopy (TEM), Raman spectroscopy, x-ray diffraction (XRD), and x-ray photoelectron spectroscopy (XPS). The sensing performances of the α-Fe2O3 sensor to various toxic gases are investigated. The optimum sensing temperature is found to be about 280 °C. The sensor shows excellent selectivity to ethanol. For various ethanol concentrations (1 ppm–20 ppm), the response and recovery times are around 3 s and 15 s at the working temperature of 280 °C, respectively. Specifically, the α-Fe2O3 sensor exhibits a response shift less than 6% to ethanol at 280 °C when the relative humidity (RH) increases from 30% to 70%. The good tolerance to humidity variation makes the sensor suitable for reliable applications in Internet of Things (IoT) in realistic environments. In addition, the sensor shows great long-term repeatability and stability towards ethanol. A possible gas sensing mechanism is proposed.
Ethanol sensors have been extensively applied in numerous fields, such as chemical industries, the development of fungi and bacteria in foodstuffs, ethanol breath analyzers in drivers’ breath, and so on. Gas sensors based on metal-oxide-semiconductors have attracted widely attention in the last decades, due to their small size and low cost.[1–5] However, the miniaturized and low-power gas sensors remain deployment for reliable applications in Internet of Things (IoT) in realistic environments.
Hematite (α-Fe2O3), n-type semiconductor (Eg = 2.1 eV), is the most thermodynamically stable iron oxide under ambient conditions.[6] Because of its environmentally friendly, high resistance to corrosion and low cost, α-Fe2O3 has widespread applications in various fields, such as magnetic devices, catalysts, electronic materials, gas sensors, biological and medical fields.[7–12] Sensor devices based on α-Fe2O3 nanostructures have been reported, for different gas detections, such as volatile organic compounds (VOCs),[7] ethanol,[8, 13, 14] H2S,[15] and NO2.[16, 17] Noteworthily, α-Fe2O3 could provide rapid response/recovery gas sensing properties due to its special valence state.[13, 18, 19] However, the pristine α-Fe2O3-based gas sensors for reliable applications have not received much attention, especially the ones with immunity to humidity in realistic environments. Furthermore, limited reports on the great selectivity of the pristine α-Fe2O3 to ethanol are available.[20, 21]
Generally, the ambient humidity varies greatly in realistic environments from case to case, and even in different seasons. The vulnerability to humidity not only makes drift of the gas sensor response, but also deteriorates the long-term stability of gas sensors. For the application in realistic environments, a great immunity to humidity of gas sensors is vital. In previous reports,[22–24] ambient humidity had no obvious effect on the gas sensing properties when the sensor device worked at high temperatures. This is due to that water adsorption is too small at high temperature.
So as to provide control over the sensing temperature and keep the low-power consumption of a device while heating the sensing material, a microfabricated heater is needed. With the developments of microelectronics and IoT, the device sizes shrink continuously as predicted by Moore’s law.[25, 26] The miniaturized, low cost, and low-power (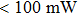
Given the challenges of gas sensors for reliable applications in realistic environments and the relative lack of knowledge of the pristine Fe2O3-based gas sensors, an exploration of the gas sensing properties of α-Fe2O3 nanorhombs is in order. Here, we report the characterizations of a highly reliable and selective ethanol sensor with immunity to humidity based on α-Fe2O3 nanorhombs. The α-Fe2O3 nanorhombs are synthesized using solvothermal method. Owing to control the sensing temperature and enhance the sensing response and recovery, α-Fe2O3 is integrated onto a microheater. The sensor devices are tested in different humid environments. The relative humidity (RH) dependence on ethanol sensing is experimentally investigated. A cross-sensitivity to other VOCs and ammonia was also performed. A reasonable gas sensing mechanism is discussed.
The α-Fe2O3 nanorhombs were prepared via a solvothermal route.[18] Typically, 0.202 g of Fe(NO3)3

Transmission electron microscopy (TEM) images were taken with a JEOL JEM-2100 microscope to characterize the morphology of α-Fe2O3 nanorhombs. Powder x-ray diffraction (XRD) analyses were performed on a Bruker D8 Advance diffractometer with Cu Kα radiation (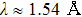
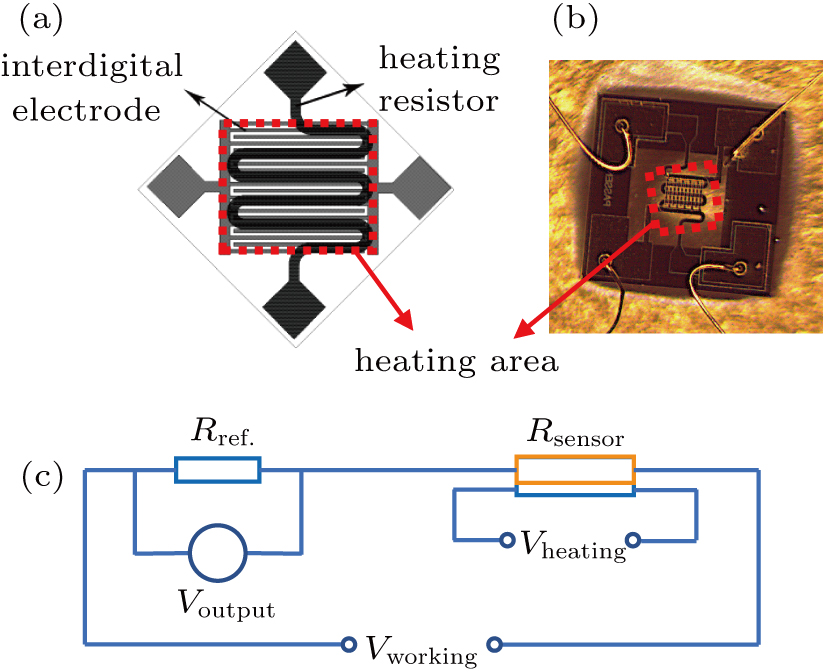
The gas sensor devices were based on an SOI (silicon on insulator) MEMS microheater technology. Figure 
The gas sensing properties were evaluated in a homemade measuring system via a static process. For the test, a reference resistor (Rref.) was put in series to form a measurement circuit. The circuit diagram is illustrated in Fig.
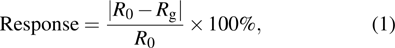 |
Morphology of the as-synthesized α-Fe2O3 is characterized using TEM. Figure
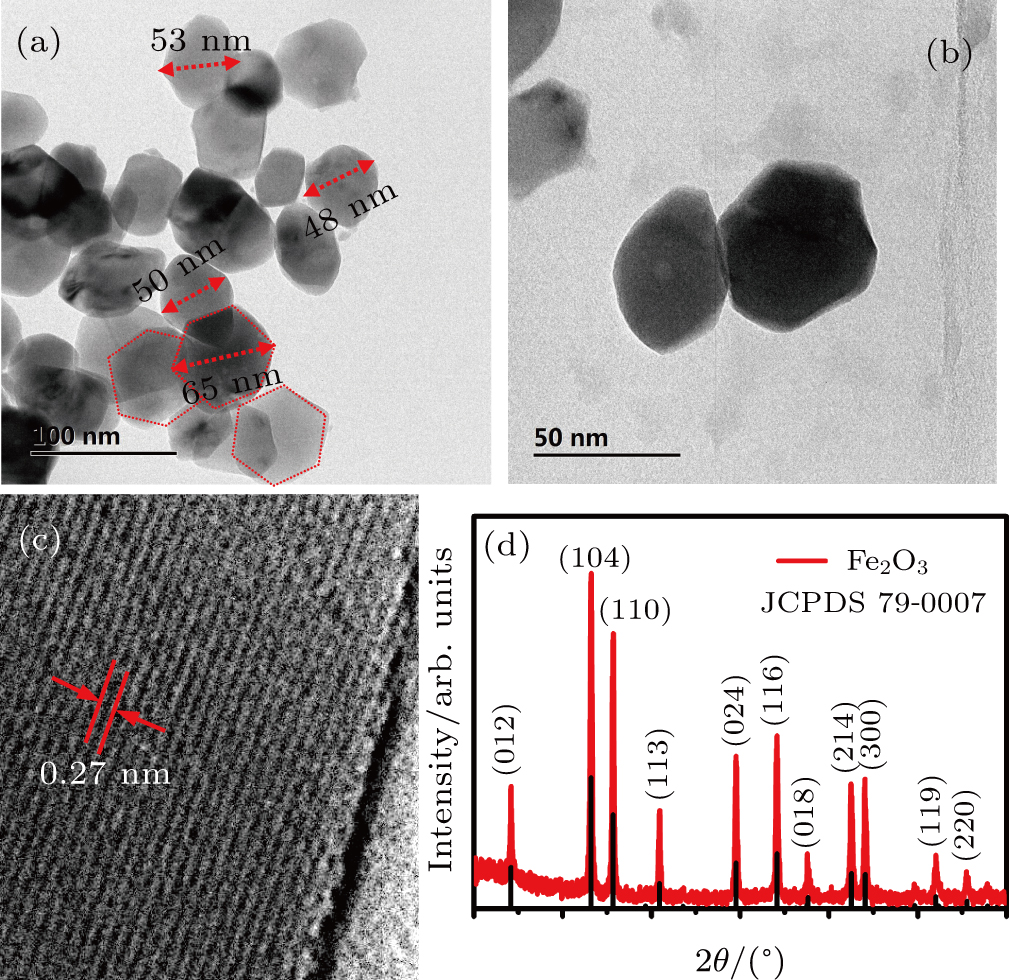 | Fig. 2. The α-Fe2O3 nanorhombs: (a) and (b) low magnification TEM images, (c) high resolution micrograph, (d) x-ray diffraction spectrum. |
The x-ray diffraction pattern (Fig. 
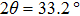
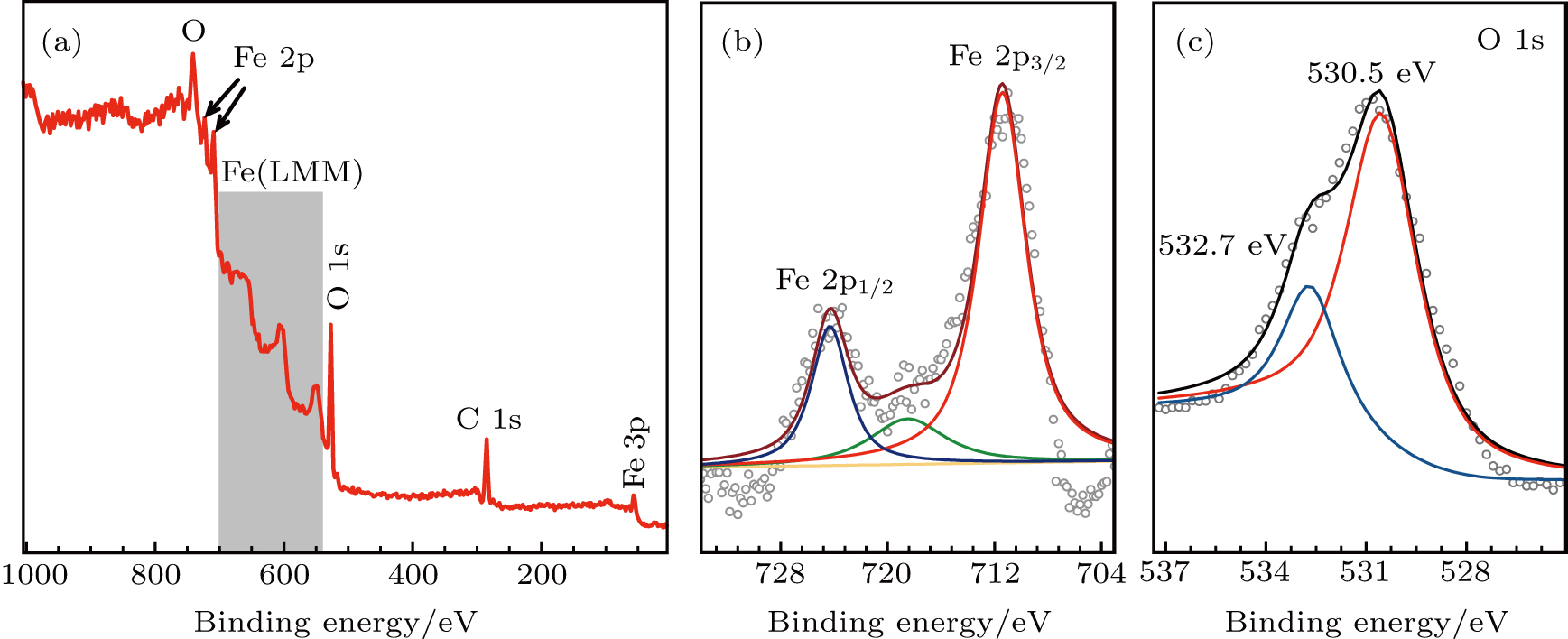
The XPS survey spectrum of the α-Fe2O3 nanorhombs is shown in Fig.
The gas sensing performances of the α-Fe2O3 sensor are related to the working temperature closely. This is because that the adsorption/desorption rates of oxygen molecules and target gas molecules on the sensing material surface are affected by temperature. The gas sensing properties of the α-Fe2O3 sensor are investigated at different operating temperatures of the heater. Nearly linear current versus voltage behaviors at different temperatures suggest an Ohmic contact between α-Fe2O3 and sensor electrodes (Fig. 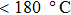
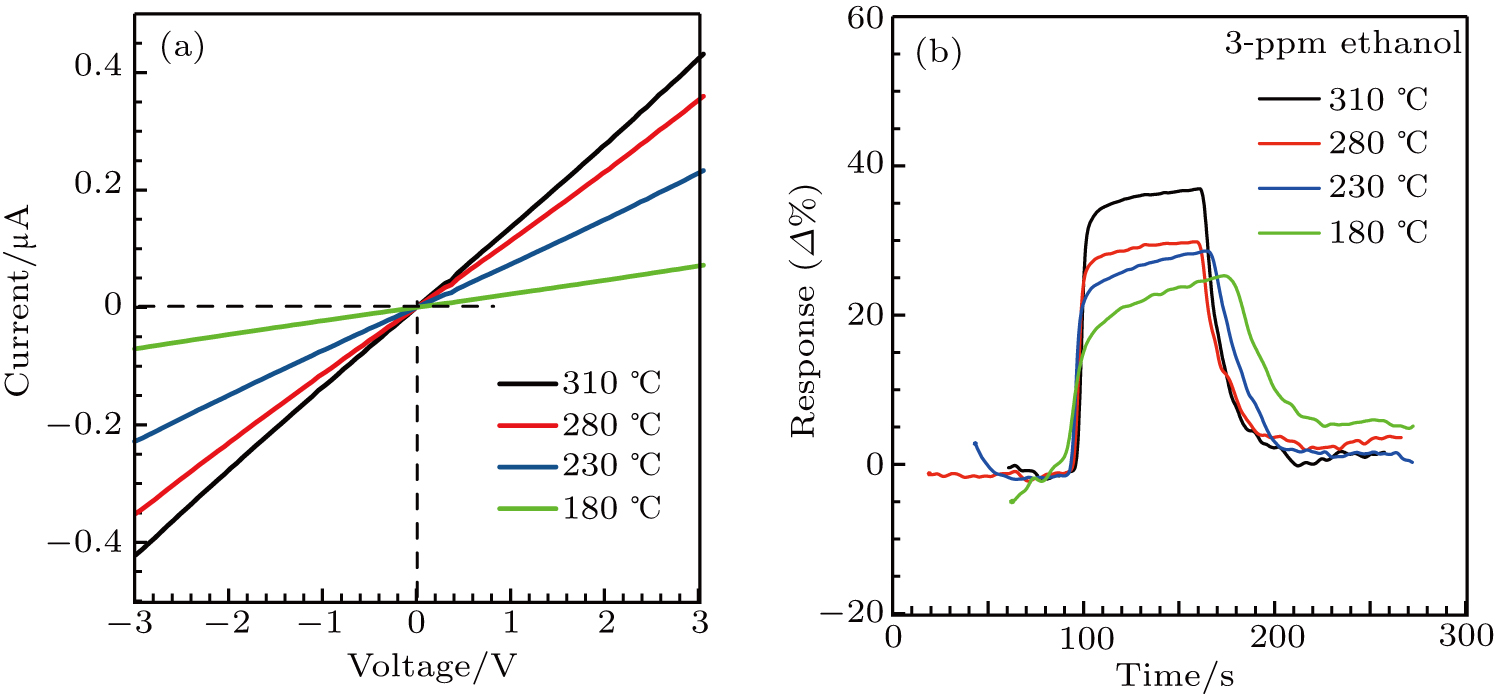 | Fig. 4. (a) I–V curves of the α-Fe2O3 nanorhombs-based sensor at different working temperatures. (b) Response values of the sensor to 3-ppm ethanol at different working temperatures. |
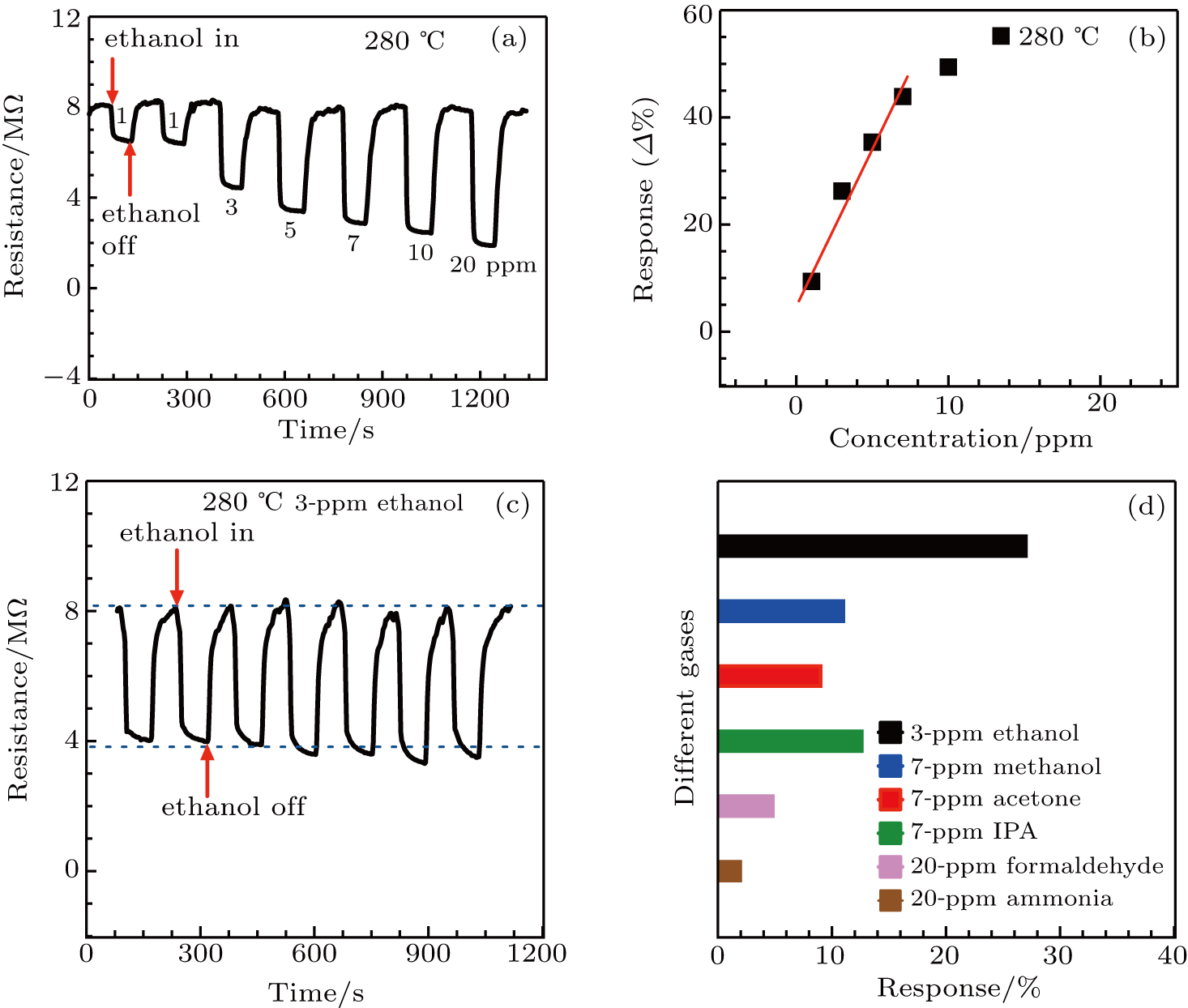
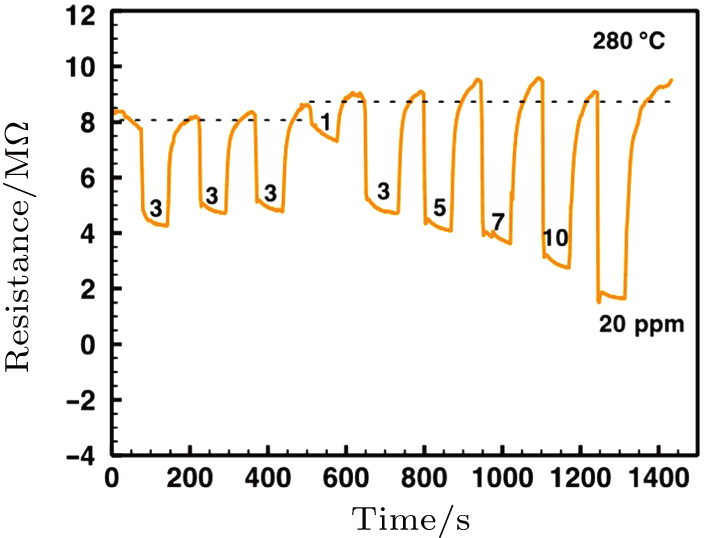
The α-Fe2O3 sensor response to varied concentrations of ethanol from 1 ppm to 20 ppm at 280 °C is shown in Fig.
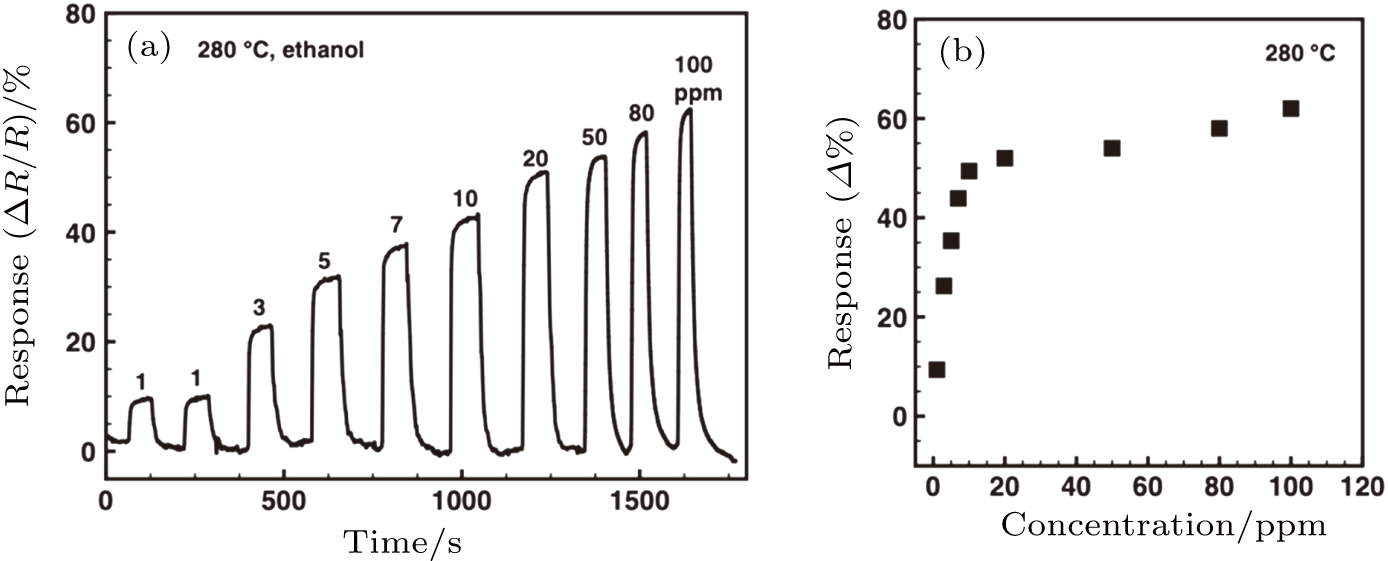 | Fig. A1. The α-Fe2O3 nanorhombs-based sensor working at 280 °C: (a) Resistance versus time for various ethanol concentrations (1 ppm–100 ppm), (b) the corresponding response values. |
Repeated response/recovery measurement of the α-Fe2O3 sensor to 3-ppm ethanol gas for 7 cycles are shown in Fig.
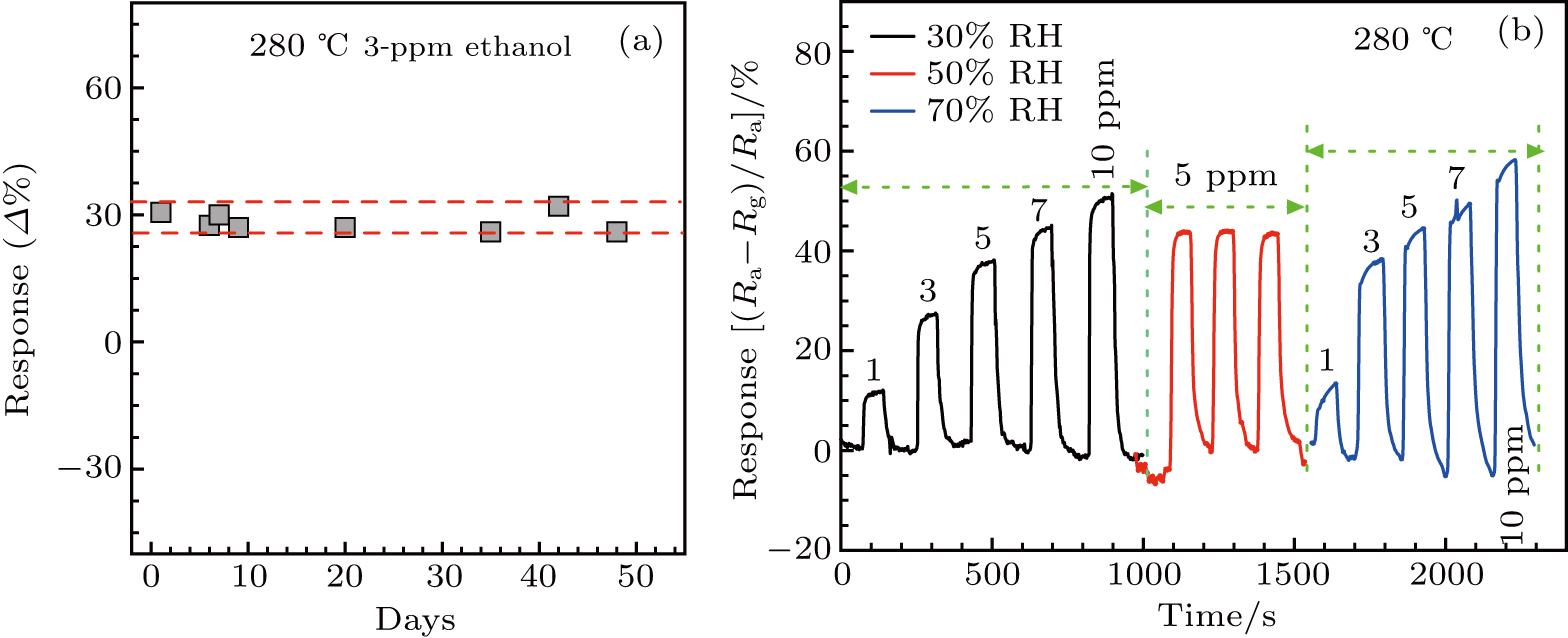
Excellent long-term stability of gas sensors is another essential requirement for reliable applications. The responses of the α-Fe2O3 sensor to 3-ppm ethanol gas were measured over a period of 50 days, while the sensor is always on at the working temperature of 280 °C. The response values and the reproducible resistance 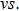
 | Fig. A2. Repeatability for 3-ppm ethanol of the fresh α-Fe2O3-based sensor and the one over working at 280 °C for 50 days. |
Humidity is a critical issue for all semiconducting solid-state sensor devices due to the varying humidity in realistic environment. As shown in Fig.
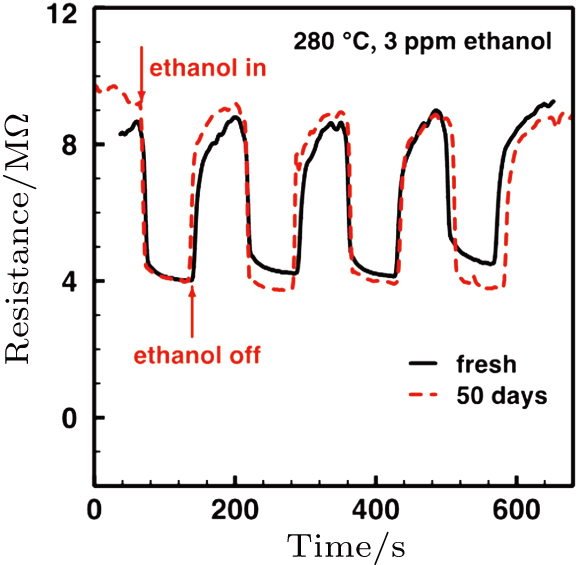
The gas sensing mechanism of α-Fe2O3 sensor has been clarified in many previous reports.[13, 31] The model is based on the change in resistance caused by the adsorption and desorption of gas molecules on the surface of sensing films. In air ambient, the oxygen molecules are adsorbed on the α-Fe2O3 surface. The adsorbed oxygen molecules capture electrons from the conduction band of the α-Fe2O3 surface layer, which results in the formation of chemisorbed oxygen ions O− at the operating temperature of 280 °C, according to the following equation:[32]
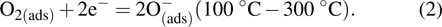 |
Hence, an electron depletion layer is formed on the surface of α-Fe2O3, and a potential barrier is generated contributing to a high resistance state.
Upon exposure to ethanol gas, the ethanol molecules react with the adsorbed O− ions on α-Fe2O3 surface and release the trapped electrons back to conduction band of α-Fe2O3, resulting in a reduced potential barrier energy. Accordingly, the sensor’s resistance is decreased. These can be expressed according to the following reactions:
 |
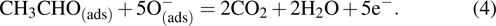 |
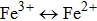
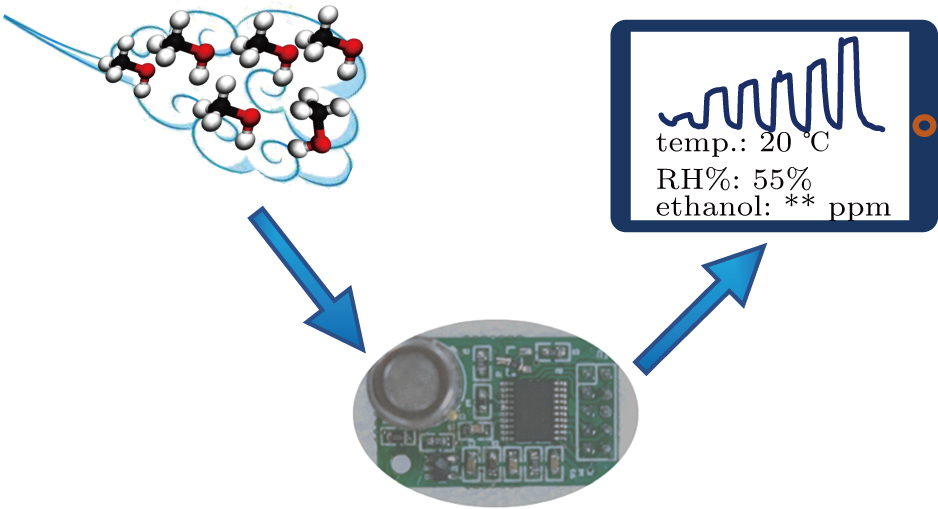
To demonstrate the feasibility of real application of the α-Fe2O3 sensor, the sensor was integrated with a prototype of electronic circuit, which can transmit sensing data to mobile terminal and show the ethanol gas concentration values of the atmosphere. Figure
Highly reliable and selective ethanol sensors based on α -Fe2O3 nanorhombs integrated with MEMS microheater were successfully prepared. The α-Fe2O3 nanorhombs were synthesized solvothermally. The MEMS gas sensors are found to be power-efficient and on-chip electronic circuit integration is possible. The sensing performances were investigated in the temperature range 180 °C–310 °C, while the corresponding power of the microheater was in the range 44 mW–70 mW. The optimum operating temperature was found to be at 280 °C, with the heater at 60 mW. The sensor showed great selectivity to ethanol gas. The ethanol response was found to be 9%–56% for the concentration range 1 ppm–20 ppm. The response and recovery times were around 3 s and 15 s for all the concentrations (1 ppm–20 ppm) of ethanol, respectively. In the humid conditions (RH of 30%–70%), the α-Fe2O3 ethanol sensor was reliable with acceptable immunity to humidity. The sensor showed excellent repeatability and stability for a long term (50 days) towards ethanol. The MEMS sensors based on α-Fe2O3 nanorhombs are expected to have numerous applications and develop low cost smart ethanol sensor in realistic environments.
The authors acknowledge M Ling for help with the TEM, XRD, and XPS characterizations. The authors acknowledge X B Li for support on microheater. The authors acknowledge the 2011 Zhejiang Regional Collaborative Innovation Center for Smart City.
Some figures for better understanding the text are given below.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] |