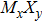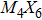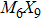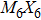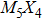† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 51702146), the College Students’ Innovation and Entrepreneurship Projects, China (Grant No. 201710148000072), and Liaoning Province Doctor Startup Fund, China (Grant No. 201601325).
Transition-metal chalcogenide nanowires (TMCN) as a viable candidate for nanoscale applications have been attracting much attention for the last few decades. Starting from the rigid building block of M6 octahedra (M = transition metal), depending on the way of connection between M6 and decoration by chalcogenide atoms, multiple types of extended TMCN nanowires can be constructed based on some basic rules of backbone construction proposed here. Note that the well-known Chevrel-phase based 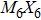
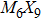
One-dimensional materials, such as nanotubes and nanowires, have been attracting considerable attention for decades for the significance on exploring fundamental physics at low dimensions and also as building blocks for materials in nanoscale electronic and mechanical device applications.[1–5] Carbon nanotubes (CNTs) in particular as almost perfect 1D systems have been studied intensively in multidisciplinary research and for multifunctional applications. However the drawback of CNTs has remained for long, such as difficulty in chirality-controlled synthesis, catalysis contamination, non-Ohmic electrode contact, and difficulty to separate bundled tubes.[6] Combining subnanometer size with structural stability, uniform electronic properties, and many advantages over CNTs, transition-metal chalcogenide nanowires (TMCN) have been studied widely as a potentially viable alternative to CNTs for many applications.[7–9]
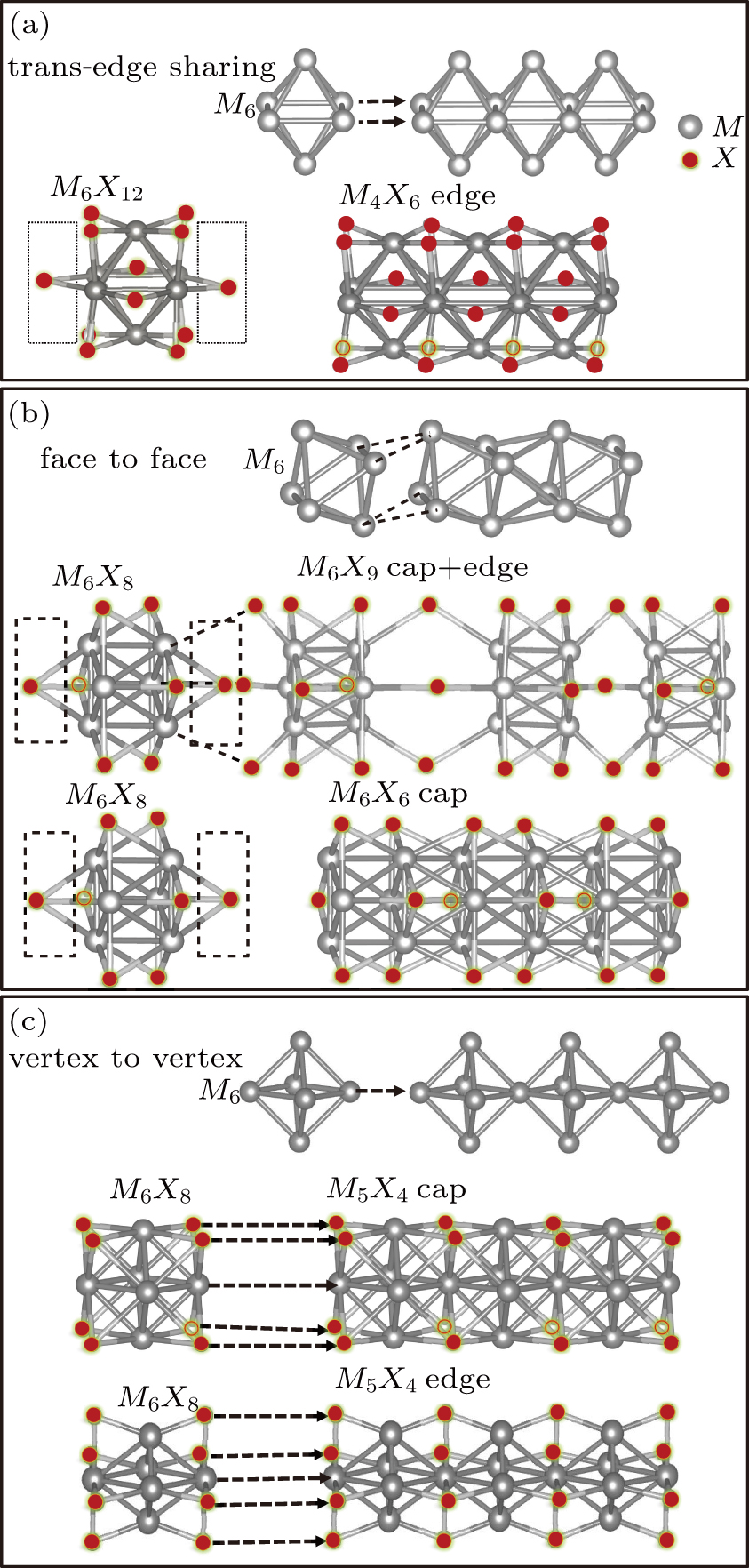
So far, a plethora of TMCN structures have been synthesized in experiments. All the possible forms of atomic structures can be summarized in Fig. 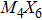
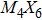
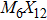
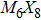
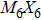
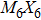
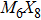
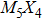
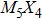
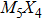
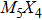
The nanowires arising from the vertex sharing of M6 attracted our most attention, not only due to the success in synthesis with a low X/M ratio which might be environmentally friendly, but also because of the smallest cross-section area among all TMCN nanowires available.[7,23–26] The cross-section size of nanowires is intimately related to quantum confinement, a more pronounced confinement effect than its sibling ribbon or wire structures is therefore anticipated and may give rise to much enhanced electronic properties than its bulk counterpart.[27–29] However, its structural stability and electronic properties are still unclear. How is it compared to the other TMCN nanowires reported? What are exactly their electronic structure? Is is possible to obtain a more stable geometry by making full use of the construction rules proposed here?
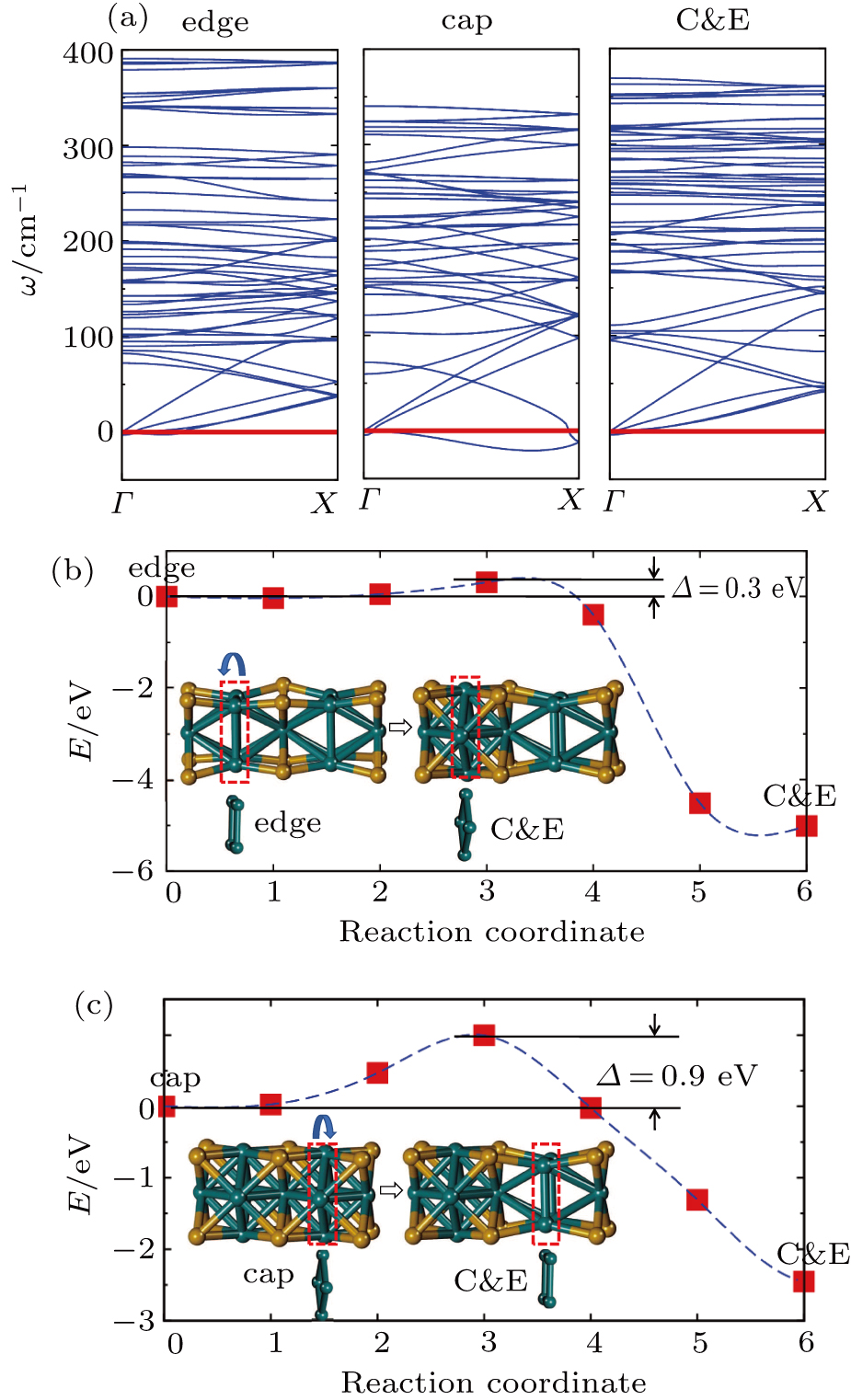
To address these problems, we select Mo and S elements and study the structural stability and electronic properties of vertex-sharing Mo5S4 nanowires and compare them with similar nanostructures in terms of convex hull of binding energy and phonon dispersion relation. We confirm that the experimentally obtained edge structure is stable from the binding strength and phonon dispersion relation. Interestingly, a new structure (the so-called C&E) with mixed cap and edge units is predicted here and appears more stable than the cap or edge structures. The calculated energy barriers of Peierls phase transitions from cap and edge to C&E are 0.9 eV and 0.3 eV per unit cell, respectively, suggesting that it is likely to achieve by a structural phase transition. Crystal orbital overlapping population (COOP) is also calculated to study the bonding character. COOP shows that the cap structure is unstable due to the partial occupation of anti-bonding states, while both the edge and C&E structures are stable from the COOP analysis. All three Mo5S4 phases are semiconductors with indirect band gap. Note that a band nesting appears in the band gap region of the C&E structure, and can be well fit for the optoelectronic sensor in the infrared spectrum.
To study the structural stability and potential applications of transition metal chalcogenides nanowires, we carried out density functional calculation with the projector augmented wave method[30] as implemented in the Vienna ab initio simulation package (VASP).[31] The exchange correlation potential was approximated by generalized gradient approximation (GGA) proposed by Perdew, Burke, and Ernzerhof.[32] To describe well-isolated nanowires while using periodic boundary conditions, we used a tetragonal lattice with a large interwire separation of 20 Å. We sampled a 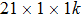
On the basis of the equilibrium structures, the phonon dispersion relation of nanowires was calculated based on density functional perturbation theory[33] as implemented in the PHONOPY code.[34] A 3 ×1 ×1 supercell size and 10 ×1 ×1 q mesh were used.
Crystal orbital overlapping population[35,36] was used to evaluate all pairwise interactions between two orbitals (i
th and j
th) of two atoms (at R and 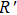
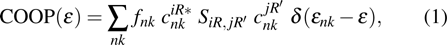 |
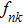
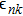
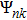
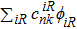
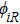
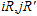
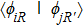
To form a stable extended nanowire structure, Mo6 octahedral unit and how sulfur decorates the rigid unit are both essential. In regard to how sulfur decorates, the two most stable cluster structures, Mo6S12 and Mo6S8, can give us a good hint. As shown in Fig.
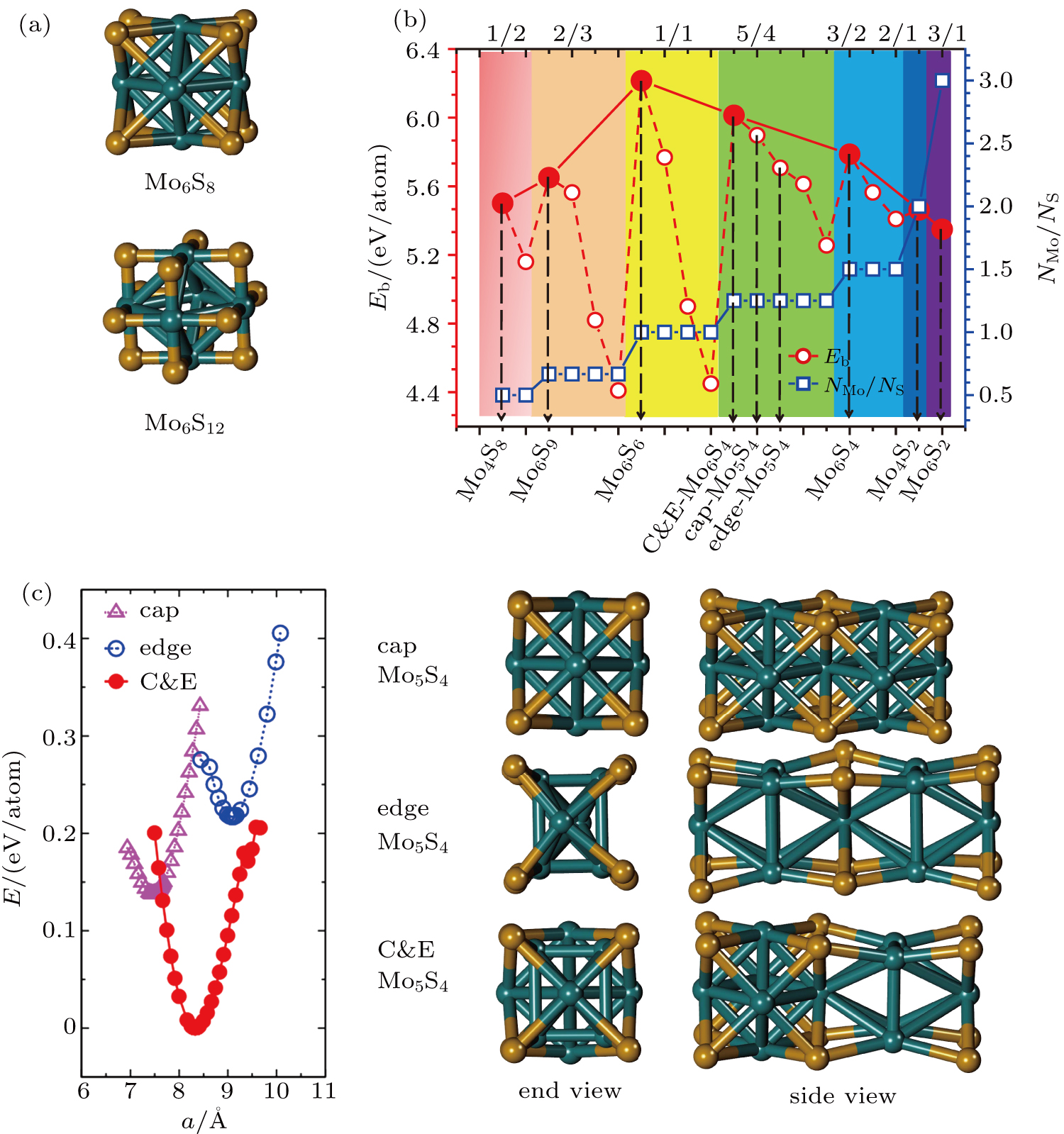 | Fig. 2. (a) Two primary cluster structures Mo6S8 and Mo6S12 with face-bridged and edge-bridged chalcogenides, respectively. (b) Binding energy as a function of stoichiometry and structure of MoxSy nanowires. Stoichiometry ratio x/y is grouped into different color zones, the most stable structures are represented in filled red dots, while the rest in empty red dots. Square blue is to show the value of x/y, or 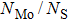
|
Guided by the strategy, we constructed possible atomic structures at different stoichiometries, i.e., 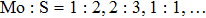
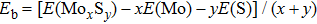
More interesting is the stoichiometry Mo:S = 5:4 given in the green zone in Fig.
We already see from the binding energy that two new structures (cap and C&E) are more stable than the experimentally observed edge Mo5S4 nanowires. But it is still not clear whether they are stable dynamically. We are also curious whether it is plausible to obtain the new and stable structures from the one available. Moreover, the electronic properties, which have not been studied yet, need to be calculated before any potential applications can be conceived.
To check the dynamic stability and possible route for synthesis, we performed phonon dispersion relation and nudged elastic band (NEB) calculation. From the phonon dispersion relation shown in Fig.
Indeed, as mentioned in Fig.
To have an insight into the electronic properties of Mo5S4 nanowires, we calculated the electronic band structure and show it in Fig.
 | Fig. 4. Band structure, density of states (DOS), and crystal COOP of (a) cap, (b) edge, and (c) C&E Mo5S4 nanowires. Positive value of COOP represents bonding states and negative anti-bonding states. |
When looking into both the electronic density of states (DOS) in Fig.
It is worth noting that, different from the zone-folding feature of bands in Figs. 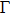
In summary, we present a theoretical investigation on the stability and electronic properties of new types of transition metal chalcogenides Mo5S4 nanowires. The stability of the predicted new structural phases (cap, edge, and C&E) are explored by the binding energy, phonon dispersion relation, and crystal orbital overlapping population analysis. The experimentally obtained edge structure is confirmed to be thermodynamically and dynamically stable. Moreover, a new and stable C&E structure having mixed cap and edge units and more stable than the edge structure is found dynamically stable. COOP analysis also substantiates the structural stability of both edge and C&E phases based on the maximally optimized bonding states. The calculated energy barrier of Peierls phase transition from the edge to C&E is about 0.9 eV per unit cell, indicating a likability to achieve the C&E phase from the experimentally available phase. All the Mo5S4 phases are semiconducting with indirect band gap. Band nesting appearing in band gap region of the C&E structure makes it well fit for optoelectronic sensor applications in the infrared spectrum.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] |