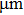† Corresponding author. E-mail:
Natural aluminum particles have the core–shell structure. The structure response refers to the mechanical behavior of the aluminum particle structure caused by external influences. The dynamic behavior of the structural response of aluminum core–shell particles before combustion is of great importance for the aluminum powder burning mechanism and its applications. In this paper, an aluminum particle combustion experiment in a detonation environment is conducted and analyzed; the breakage factors of aluminum particles shell in detonation environment are analyzed. The experiment results show that the aluminum particle burns in a gaseous state and condenses into a sub-micron particle cluster. The calculation and simulation demonstrate that the rupture of aluminum particle shell in the detonation environment is mainly caused by the impact of the detonation wave. The detonation wave impacts the aluminum particles, resulting in shell cracking, and due to the shrinkage-expansion of the aluminum core and stripping of the detonation product, the cracked shell is fractured and peeled with the aluminum reacting with the detonation product.
Aluminum powder can greatly increase the potential energy of composite energetic material, such as explosives and propellants.[1–4] However, much of the potential cannot be released due to incomplete combustion. The study of aluminum powder combustion theory in detonation environment is of great significance to solve the above problem and enrich the theoretical model of metal powder combustion.
Natural aluminum particles have the core–shell structure.[5] The structure response is the mechanical behavior of the aluminum particle structure caused by external influences. An aluminum ignition and combustion occurs after oxide shell separation, so the response mechanism of the core–shell structure of aluminum particle before ignition is the focus of aluminum powder combustion theory. The study of core–shell structural response of aluminum particle under slow heating conditions is conducted in detail.
Eisenreich[6] researched the slow thermal response of aluminum powder for certain size ranges with different stages of aluminum oxidation being observed. His conclusions were supported and referenced by other scholars, such as Hasani and Korshunov.[7–9] Further, Eisenreich[10] noted that the alumina layer has a phase change in oxidation, but there was no quantitative correlation between the dynamic changes of phase transformation and oxidation.
Dreizin[11] studied Al particle combustion in air using a pulsed micro-arc discharge. He found that the Al particle combustion corresponds to different temperatures, internal particle compositions, and flame shapes. Trunov[12,13] also noted that the growth of the crystal phase change caused by the micron aluminum oxide layer is directly related to the aluminum oxidation behavior.
Trunov[14,15] proposed a reaction model for the aluminum slow weighting process as affected by the shell phase transformation for micro-aluminum. The phase change of the alumina was divided into four stages. For stage 1, the reaction temperature is less than 800 K, the natural amorphous state of the alumina shell grows, the crystal shape changes to γ-Al2O3, and the oxide layer thickens. During stage 2, between 800 K and 910 K, the transfer rate of the active material through the oxide layer increases, further promoting the phase change. At stage 3, between 910 K to 1320 K, the γ-Al2O3 oxide layer thickens and changes to θ-Al2O3 and then α-Al2O3. For stage 4, the layer continues to change to α-Al2O3 and the thickness grows further.
Bradstant[16] studied the slow oxidation behavior of micro/nano aluminum powder in a CO2 atmosphere. The maximum reaction temperature was 1250 °C and the results showed that the aluminum of less than 
Researchers also studied the combustion of aluminum particles. Glassman[18,19] first suggested that the metal particle burning was similar to droplet combustion, with the ignition depending on the melting and boiling point of the metal and oxide. He speculated that the stable aluminum combustion condition was melted alumina when the reaction temperature reached the boiling point of aluminum.
Further research confirmed the model proposed by Glassman.[20,21] Olsen[22] found that during the combustion, the generated liquid alumina would gather on the surface of particles, forming an alumina cap. Allen[23] studied nano-aluminum powder under high temperature combustion heat, verifying the Altman hypothesis. But previous research has also shown that the burning of aluminum began before the temperate reached the boiling point when the shell burst.[24] The reason for this may be different temperature and pressure environments[25] or mechanical stresses.[26]
In summary, the previous research focused on the structure response of aluminum particle in the thermal analysis (below 1500 K), and the combustion model of aluminum particles in the high temperature combustion environment (above 3000 K and below 10 MPa). However, the investigation on aluminum particle combustion in detonation environment (above 3000 K and more than 5 GPa) is deficient, which is significantly important to increase the efficiency of aluminum particle combustion. The purpose of this paper is to understand the mechanism of the structural response of aluminum core–shell particles in detonation environment.
Table
| Table 1.
Formulas of detonation test. . |
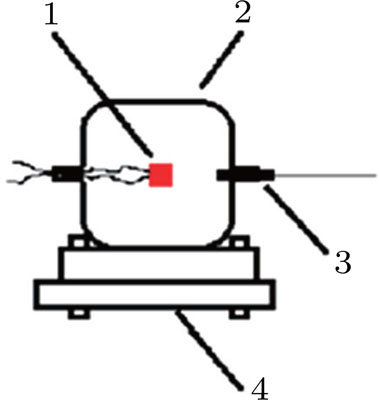
Aluminum was mixed with explosive to form charges. Three kinds of micron aluminum powder were used with particle sizes of 

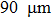

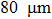
The explosive was initiated by the #8 electric detonator and 2.2-g composition RDX/wax (95/5) booster. The booster and charge were glued together and the detonator was inserted into the booster hole. The red sample in Fig.
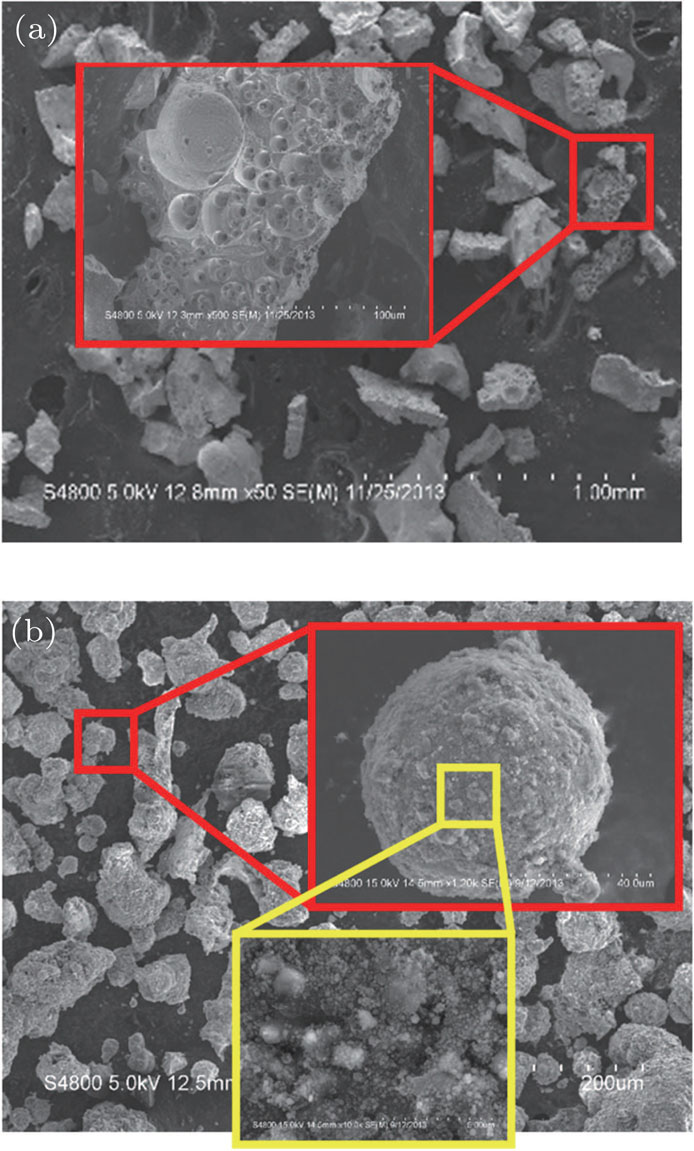
Solid detonation product is analyzed. The main solid detonation product is Al2O3. The appearance of the four formulas is similar. The typical SEM images of the products are shown in Fig.
The other one is like that as shown in Fig.
The x-ray diffraction (XRD) results are shown in Fig.
| Table 2.
Crystal phase compositions of condensed detonation products. . |
The product photo shows that the aluminum particle burns in a gaseous state and condenses into a sub-micron particle cluster. The premise of the aluminum powder gas phase reaction is that the particle shell is broken, and then the core is exposed to the high temperature gas, vaporizing, burning, and condensing into a sub-micron particle cluster.
Combined with the literature and experimental results, the potential mechanism for the separation of aluminum core–shell particles in detonation environment is as follows.
(I) Thermal stress. The density of solid aluminum is 2.7 g/cm3, and the density of liquid aluminum is 2.4 g/cm3. Therefore, when the aluminum core is melted, its volume expands by 12%. The aluminum shell swells and ruptures due to internal tensile stress.
(II) Detonation wave. The detonation wave pressure is greater than 10 GPa in the aluminized explosive reaction zone. Such high pressure causes a large deformation of the aluminum powder, which makes alumina shell fail to crack, forming cracks on the surface of the aluminum.
In the detonation environment, high temperature and strong impact are present simultaneously, and the two factors should be considered comprehensively. By calculating the separation time required for the two methods, the main cause of the core–shell deconstruction in the detonation environment can be determined.
(III) Crystal transformation and phase transformation of alumina. Thermal analysis results[27,28] show that as the temperature increases, the alumina changes from an amorphous state to γ and θ states, eventually forming a stable α state. When the crystal form of alumina changes, its density increases all the time, causing the volume of alumina to decrease, which causes the alumina to not completely cover the aluminum core, resulting in shell–core separation.
In summary, the crystal transformation and phase transformation of alumina are the main reasons for shelling in thermal analysis environment (300 °C–1100 °C); in the combustion reaction, thermal stress is the main reason; in the detonation environment, high temperature and strong impact are present simultaneously, and the two factors should be considered comprehensively. By calculating the separation time required for the two methods, the main cause of the core–shell deconstruction in the detonation environment can be determined.
The melting point of aluminum is 933 K.[29] TNT/Al (80/20) explosive detonation temperature is 3309 K by cast formula.[30] Taking 10-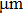

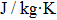
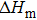

According to Fourier’s law

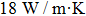
Bring the heat capacity, phase change enthalpy, and other parameters[29] into the formula, and tmel can be calculated.
When the diameter of aluminum particle is 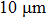
| Table 3.
Heating time of different aluminum particle sizes reaching the aluminum melting point. . |
To investigate the mechanical response of aluminum particle in the detonation shock wave, the finite element software AUTODYN[31] is employed. The aluminum with core diameter of 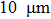
The simulation model consists of TNT, aluminum, and alumina. The location of the detonation of the TNT is to the left of the surface. The relationship between pressure and the relative volume of the TNT product is as described by the Jones–Wilkins–Lee (JWL) state equation.[32] The constitutive equations of aluminum are described by the Von Mises model[33] and the constitutive equations of alumina are described by the Johnson–Holmquist (JH) model.[34]
The detonation shock wave impacts the aluminum particle with a force of tens of GPa. The different mechanical properties of alumina and aluminum lead to different types of deformation: the alumina shell fails and fractures under the shock wave and the aluminum core experiences elastic-plastic deformation. Figure
Figure
Based on this simulation, the shell breakage times for different particle sizes are obtained, and the results are shown in Table
| Table 4.
The breakage time of alumina shell for different particle sizes. . |
At the same particle size, the alumina shell breakage of the detonation wave impact is significantly less than tmel caused by the thermal stress. Therefore, the rupture of the aluminum particle shell in the detonation environment is mainly caused by the impact of the detonation wave.
The velocity curves of four gauges are calculated, as shown in Fig.
According to the detonation product and simulation results, the structure response of aluminum particle can be divided into three stages. In the first stage, the spherical particles are squeezed by the detonation wave, and the windward side shell cracks. During the second stage, the particle rebounds, the shell breaks and is peeled by the detonation product stripping with internal aluminum core stress, and the internal aluminum steam burns with the detonation products. For the third stage, the detonation product alumina cannot be attached to the surface of the core, so the aluminum keeps evaporating and burning with some product being attached to the container wall and some condensing into sub-micron particle clusters. The structural response of aluminum particle is shown in Fig.
The analysis of the aluminum particle product for detonation environments demonstrates that the aluminum particle burns in a gaseous state and condenses into a sub-micron particle cluster.
The rupture of the aluminum particle shell in the detonation environment is mainly caused by the impact of the detonation wave. The result shows that the detonation wave impacts on the aluminum particles, resulting in shell cracking and then, due to the shrinkage-expansion of the aluminum core and stripping of the detonation products, the cracked shell is fractured and peeled with the aluminum reacting with the detonation product.
The core–shell structure response of the aluminum particle in a detonation environment provides a foundation for studying the combustion behavior and revealing the burn mechanism of aluminum particles in such environments.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] |