† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11674235, 11374218, and 21204058), the National Laboratory of Solid State Microstructures, China (Grant No. M30036), and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions, China.
Films of poly(9,9-dioctylfluorene) (PFO) are of great importance in fabricating light emitting diodes. In practice, the (β-phase of PFO is expected due to its high efficiency in the transport of charge carrier. To promote the formation of (β-phase, PFO films are immersed and annealed in the mixture of solvent/nonsolvent. The effects of temperature, solvent/nonsolvent ratio, and annealing time are examined systematically. It is found that the fraction of (β-phase can be highly improved by increasing the ratio of solvent/nonsolvent. The reconfiguration of PFO molecules for (β-phase in annealing is generally finished in 10 min. The finding in this study demonstrates that solvent-assisted annealing offers a fast and economic approach for mass annealing.
Conjugated polymers are emerging as promising materials for fabricating elastic and flexible electronic and photonic devices.[1] Among them, poly(9,9-dioctylfluorene) (PFO) as a blue-light-emitting polymer has been extensively studied due to its excellent charge transport properties and highly efficient photoluminescence. Optoelectronic devices based on PFO have a wide range of potential applications in organic light-emitter diodes (OLEDs),[2] lasers,[3] solar cells,[4] and field effect transistors.[5] In these applications, the molecular conformation of PFO plays a critical role in determining the performance of the devices. There are two distinct conformations of PFO in films, namely: α-phase and β-phase. In the β-phase, the polymer chains adopt an extended coplanar conformation which allows a high charge-carrier mobility[6] and a high efficiency in photoluminescence.[7–9] For applications such as light emitting diodes and electrically pumped organic lasers, the high charge-carrier mobility is critical.[10] Therefore, improving the fraction of the β-phase in PFO products is of particular interest in fabricating organic semiconductors.[8]
The most widely used methods in preparing thin polymer films are spin-coating, vapor deposition, and solvent evaporation. In these methods, the conformation of the polymer chains in the products is generally arrested in the amorphous glass state because of the fast dynamics. To improve the crystallinity, films are often processed by post-annealing. In the case of PFO, strategies including substrate thermal annealing[11] and solvent vapor treatment[12] have been exploited. Nevertheless, the efficiency of these approaches is limited.[13,14] PFO films with high crystallinity are still a big challenge in practice.
Recently, solvent-assisted annealing has been exploited, in which the PFO film is immersed in the mixture of solvent/nonsolvent vapor for annealing.[15] In this case, the annealing time is as long as tens of hours. Alternatively, Lu immersed the pristine PFO film in a mixture of solvent/nonsolvent solution.[16] The dynamic reconfiguration processes of polymer toward β-phase were highly accelerated via the so called self-dopant[17] mechanism. However, in their study, the effect of solvent/nonsolvent ratio and the temperature of annealing were not addressed. This greatly impeded its practical application.
In this study, PFO films are processed with solvent-assisted annealing. The effects of the volume ratio of solvent/nonsolvent, temperature, and annealing time on the fraction of β-phase are systematically studied. We find that an optimal ratio of solvent/nonsolvent exists in promoting the formation of β-phase, the result of solvent-assisted annealing is not sensitive to the temperature, and the typical time scale of the dynamic process of β-phase formation is around 10 min. The microscopic mechanism underlying these observations is addressed.
The PFO (ADS129BE) was purchased from American dye source company and used as received.
The PFO powder was dissolved in toluene and maintained at 60 °C overnight for sufficient dissolution. The solution was then filtered with 
The pristine PFO films were immersed into the mixture of toluene (solvent) and ethanol (nonsolvent) for annealing. The annealing processes were conducted at three different temperatures: 30 °C, 40 °C, and 50 °C. To quantify the effect of the temperature, the samples and the annealing solution were heated to the annealing temperature separately. The pristine PFO film was then immersed in the solution mixture to prevent the occurrence of a temperature-gradient-induced crystallization.
The morphologies of the PFO films were first visualized by atomic force microscope (AFM, MFP-3D-SA, Asylum Research) in the tapping mode on Pt-coated Si cantilevers (0.2 N 
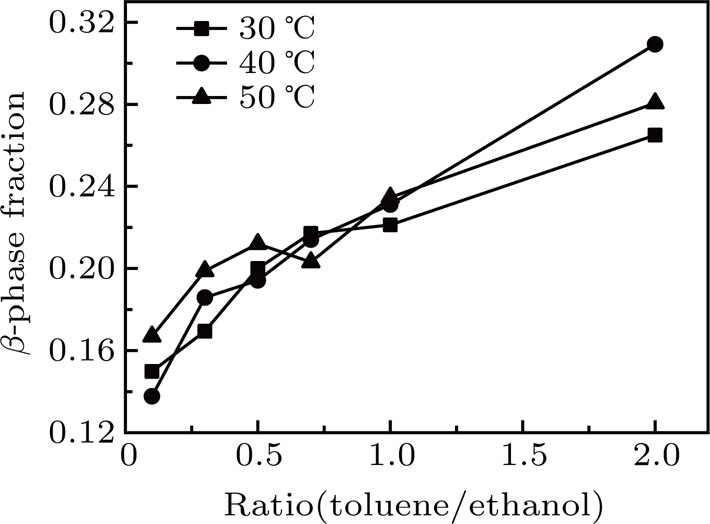
To examine the effect of the ratio of solvent/nonsolvent and the temperature, PFO films on ITO substrates were processed in the mixture of toluene (solvent)/ethanol (nonsolvent) with different mixing ratios (R) at different temperatures (T) for different processing time (t).
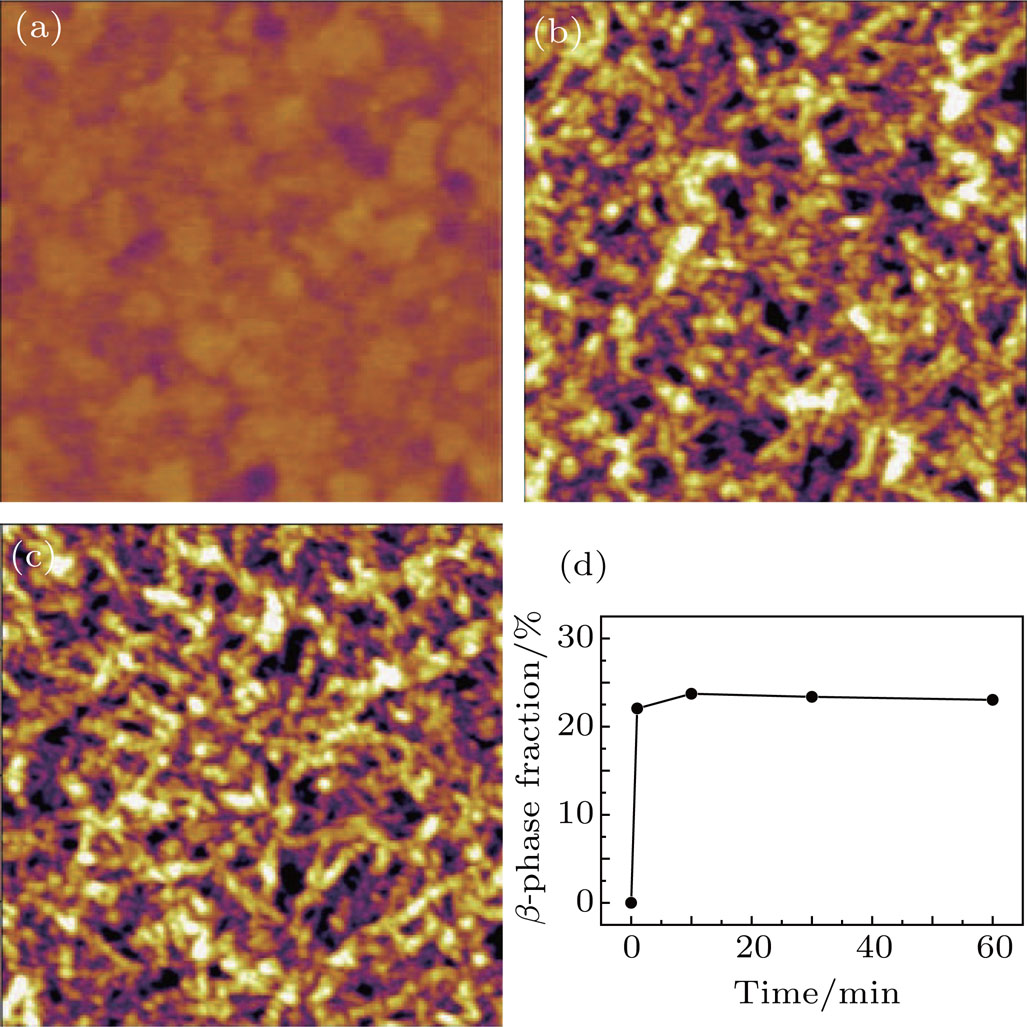
The morphologies of the products after annealing were first investigated by AFM. Figure
At the ratios of 0.5:1 (Figs.
To quantify the fraction of β-phase in the final products, the UV–vis absorption spectra of the resulting films are investigated, and the fraction of β-phase is estimated according to Lamber–beer’s law[18]
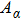
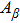
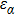
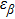
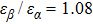
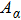
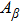
To examine the effect of processing time, the PFO films were processed at 50 °C with a solvent/nonsolvent ratio of 2:1 for different time. Figure
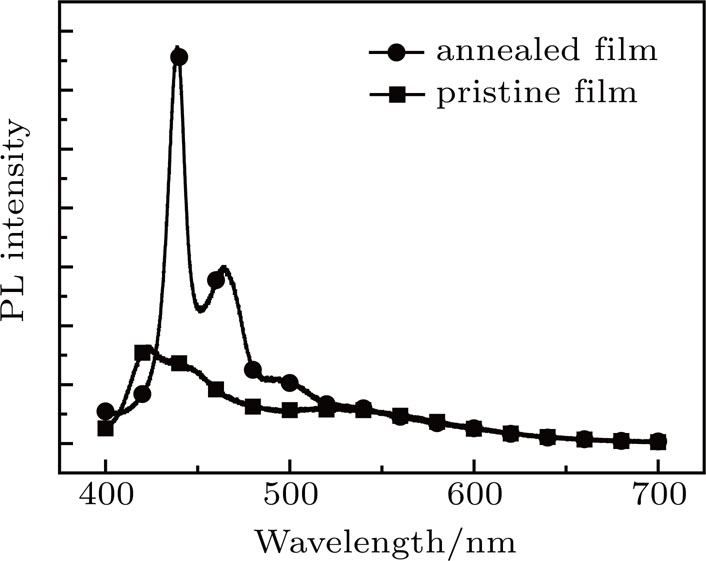
To verify the crystalline component of the fibrous structure, the PL spectrum of the PFO films was investigated. Figure
We suggest that the β-phase formation in the solvent-assisted annealing arises from the local reconfiguration of PFO macromolecular chains, which is facilitated by the presence of a good solvent. The good-solvent molecules diffuse into the PFO molecular mesh in the film and facilitate the relaxation of local molecular configurations. Due to the presence of the poor solvent molecules, the PFO chains cannot be completely dissolved but can relax to a lower free-energy state via local reconfiguration, giving rise to small crystalline domains.[19] The average size and number of the crystalline domains are determined by the number fraction of good-solvent molecules. Increasing the concentration of good solvent in the mixture leads to more and larger crystalline domains, which eventually interconnect to fibrous structures.
In summary, the β component in PFO films can be highly improved by the solvent-assisted annealing process. The annealing temperature does not impact the final results significantly. By contrast, the ratio of solvent/nonsolvent is critical and an optimal value exists to promote the formation of β component without dissolving the film. The annealing process can be accomplished within minutes. Compared with previous studies, our results clarify the effect of temperature, ratio of solvent/nonsolvent, and annealing time. These results offer a general guideline for practical applications of solvent-assisted annealing.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] |