† Corresponding author. E-mail:
Project supported by the National Key Research and Development Program of China (Grant No. 2016YFB0701002), the Joint Fund of the National Natural Science Foundation of China and China Academy of Engineering Physics (NSAF) (Grant No. U1530152), the National Natural Science Foundation of China (Grant Nos. 91222112 and 61475177), and Shanghai Rising-Star Program (B type), China (Grant No. 14QB1401600).
The influence of Er3+ ions on the spectra of Yb3+, Er3+: Sc2SiO5 (SSO) single crystal, which was obtained by Czochralski (Cz) method, is discussed. The absorption coefficient at 980 nm was 13.36 cm−1 with a peak absorption cross-section of 1.46 × 10−20 cm2. The emission cross-sections at 1034 nm and 1062 nm were 5.5 × 10−21 cm2 and 4.9 × 10−21 cm2, respectively. The fluorescence lifetime was estimated as 1.24 ms at 1061 nm. The mechanical properties of SSO single crystal were also presented.
High-performance InGaAs diode laser operating in the range of 900–1100 nm raises the development of Yb3+ doped materials for all-solid-state ultrafast laser, high power laser, and thin disk laser.[1–5] Yb3+ doped laser materials possess advantages over Nd3+ doped ones, including long radiative lifetime, small quantum defect between absorption and emission, and absence of deleterious effects such as excited state absorption, up-conversion, and concentration quenching. The combination of Er3+ and Yb3+ in up-conversion is well studied. Upon excitation at the 4I11/2 level, the cross-relaxation process of [Er3+ (4I11/2 → 4I15/2); Yb3+ (2F7/2 → 2F5/2)] could occur.[6] Therefore, in this study, we choose Er3+ as the sensitizer in Yb3+ activated laser crystal.
However, the three-energy-level scheme of Yb3+ laser could lead to a high laser threshold due to the high thermal population of the terminal level. Quasi-four energy levels of Yb3+ could be introduced by selecting a crystal host with low symmetry and multi-crystallographic sites that would improve the emission band broadening and energy level splitting.[7,8] In the silicate crystal family, scandium silicate crystal (Sc2SiO5, SSO) possesses the low-symmetry crystal structure of monoclinic C2/c space group and two non-equivalent crystallographic sites.[9–11] In addition, SSO crystal possesses high thermal conductivity (κ = 7.5 W·m−1 · K−1) and negative thermo-optical coefficient with the dn/dT value of −6.3 × 10−6 K−1.[8] With the benefits from strong coupling between ytterbium ions and crystal field, quasi-four-level Yb3+: SSO single crystal exhibits large energy stark splitting of 1027 cm−1, which is beneficial for generating short pulse laser[12–14] and reducing heat generated in high power laser system.[3,15,16] Li et al. reported a diode-pumped Yb3+: SSO chirped pulse amplifier with 1 ps pulse duration.[17] SSO crystal host has been widely used in the fields of high power lasers, ultra-fast lasers, and amplifiers.
In this paper, the mechanical properties of pure Sc2SiO5 bulk crystal will be demonstrated for the first time. The influence of Er3+ ions on the near infrared spectroscopy of Yb3+, Er3+: SSO will also be discussed.
Undoped SSO crystal was grown for the measurement of fracture toughness and flexure strength. The raw materials of Sc2O3 (4N), SiO2 (4N), Yb2O3 (4N5), and Er2O3 (4N5) were used for SSO and Yb3+, Er3+: SSO single crystals growth. The mixture powders were compressed and loaded into an alumina container before sintering at 1250 °C for 12 h in a muffle furnace. The crystals were grown by the Czochralski method in an inductively heated iridium crucible under nitrogen ambient atmosphere. SSO crystal seed oriented in (010) direction was used. The pulling rate was set at 1–2 mm/h and the rotation rate was 15–20 rpm. After pulling apart from the melting, the crystals were cooled down to room temperature in 40 h. SSO and Yb3+, Er3+: SSO crystals with diameters of 25 mm and lengths of 65 mm were obtained as shown in Fig.
The concentrations of Yb3+ and Er3+ in Yb3+, Er3+: SSO crystal detected by inductively coupled plasma atomic emission spectrometry (ICP-AES) were 7.07 wt.% and 0.065 wt.%, respectively. The segregation coefficient of Yb3+ in the Yb3+, Er3+: SSO crystal was calculated to be 0.85, while the segregation coefficient was 0.96 in Yb3+: SSO crystal.[3] The solubility of Yb3+ in the Yb3+, Er3+: SSO host lattice was reduced due to the competition of Er3+ ions with Yb3+ while substituting in the Sc3+ lattice. The influence of Er3+ on the spectra will be further discussed in Section
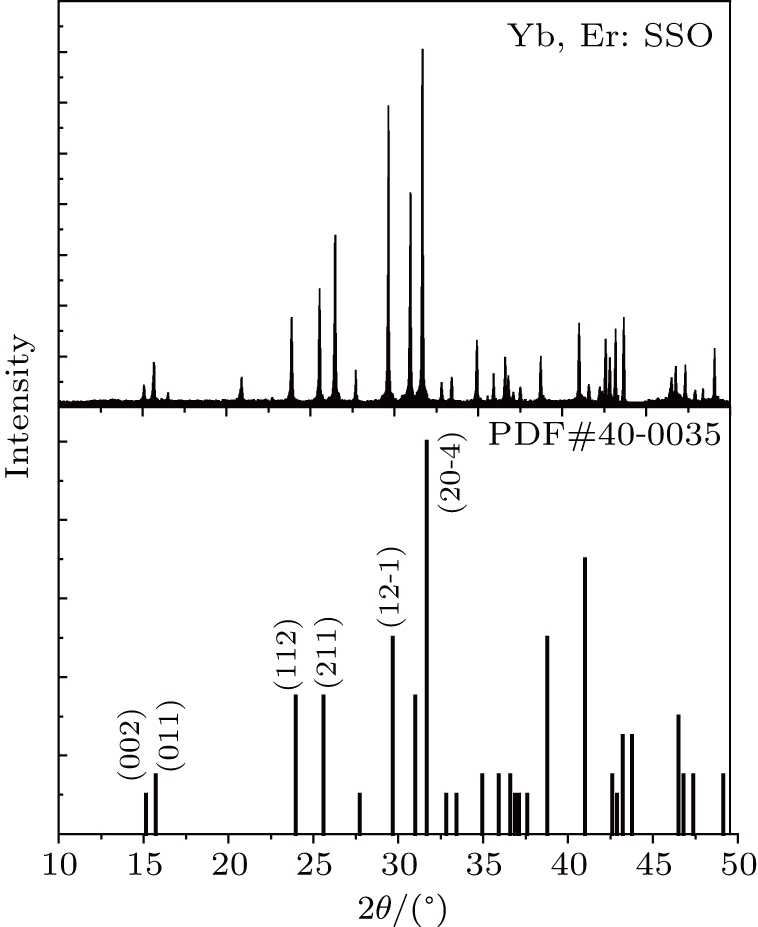
Crystal structure was confirmed by x-ray diffraction (XRD, Cu target, Kα, X-pert, Holland). Figure
To apply laser crystals in high power laser system, it is important to evaluate mechanical properties that could predict the energy storage capability of the laser host.
The Vickers hardness (HV) and fracture toughness of SSO crystal were determined by indentation technique in a digital Vickers hardness tester (Wilson-WolpertTukon2100B) at room temperature for the first time. Mechanical properties of Nd: YAG crystal were also tested for comparison.
Figure
 |
 |
The fracture toughness and flexure strength of SSO and Nd: YAG crystals are listed in Table
| Table 1. Fracture toughness and flexure strength of SSO, Nd: YAG, and other reported crystals. . |
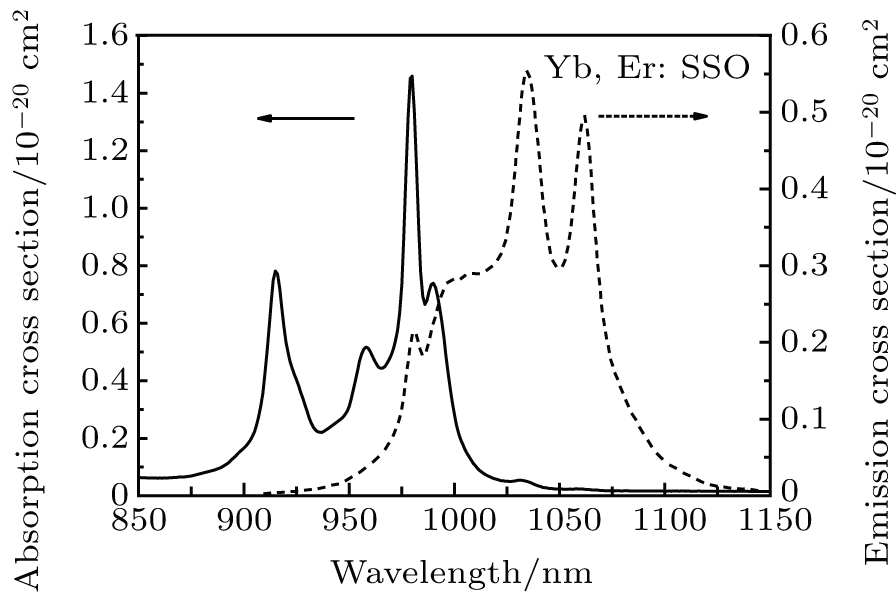
The absorption and emission spectra at room temperature of Yb3+, Er3+: SSO crystal in the wavelength region of 850–1150 nm are shown in Fig.
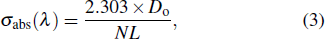 |
The emission spectrum was investigated under the excitation of Xe lamp (Jobin-Yvon TRIAX 550 spectrophotometer) in the range of 900–1200 nm. The fluorescence spectrum is mainly composed of three bands around 998 nm, 1034 nm, and 1062 nm in addition to the zero-line at 981 nm. The fluorescence intensities at 1034 nm and 1062 nm are stronger than that at 998 nm. The shapes of absorption and fluorescence spectra are broad due to the thermal spreading of the spectra at room temperature.
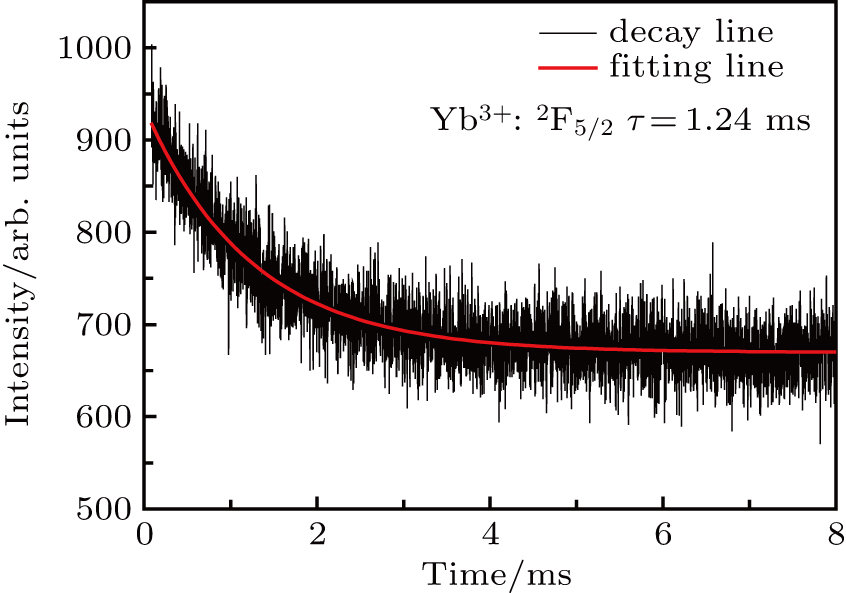
A fluorescence lifetime measurement was carried out by a computer-controlled transient digitizer decay curve of emission. The fluorescence lifetime at 1061 nm was estimated to be 1.24 ms, as shown in Fig.
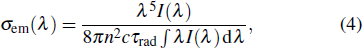 |
Table
| Table 2. Spectroscopic parameters of Yb3+, Er3+: SSO crystal. . |
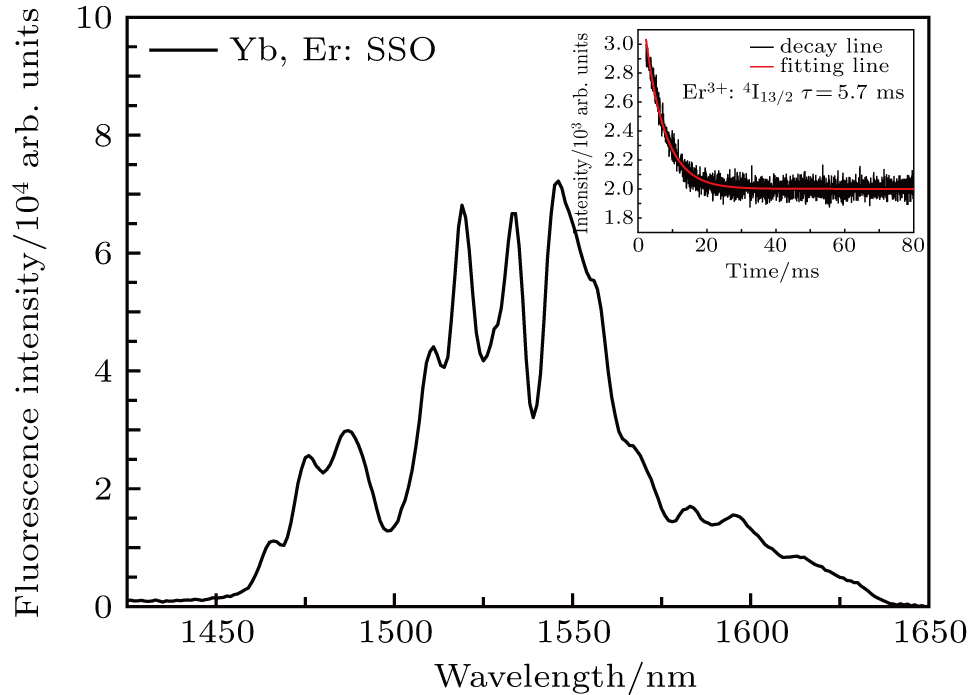
In order to better understand the effect of Er3+ in Yb3+, Er3+: SSO crystal, the emission spectrum and fluorescence decay curve of Er3+ around 1548 nm are presented in Fig.
In summary, the fracture toughness was obtained to be 0.83 MPa·m1/2 and flexure strength was 135.5 MPa in SSO crystal. Yb3+, Er3+: SSO crystal with Er3+ content of 0.065 wt.% shows broader emission bandwidth of 72 nm. The emission band at 1034 nm possesses the largest emission cross-section of 5.5 × 10−21 cm2, which is favorable for low threshold and high efficient laser operation. The effective regulation of optical parameters obtained in Yb3+, Er3+: SSO crystal demonstrated that cations-interaction is promising in investigating new laser candidates with higher emission cross-section for high power laser systems.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] |