Project supported by the National Basic Research Program of China (Grant No. 2016YFA0202302), the National Natural Science Foundation of China (Grant Nos. 61527817, 61875236, 61905010, and 61975007), and the Overseas Expertise Introduction Center for Discipline Innovation, 111 Center, China.
Project supported by the National Basic Research Program of China (Grant No. 2016YFA0202302), the National Natural Science Foundation of China (Grant Nos. 61527817, 61875236, 61905010, and 61975007), and the Overseas Expertise Introduction Center for Discipline Innovation, 111 Center, China.
† Corresponding author. E-mail:
Project supported by the National Basic Research Program of China (Grant No. 2016YFA0202302), the National Natural Science Foundation of China (Grant Nos. 61527817, 61875236, 61905010, and 61975007), and the Overseas Expertise Introduction Center for Discipline Innovation, 111 Center, China.
As an important member of the two-dimensional layers of metal dichalcogenides family, the two-dimensional (2D) group IV metal chalcogenides (GIVMCs) have been attracting intensive attention. However, the growth of monolayer tin disulfide (SnS2) remains a great challenge contrasted to transition metal dichalcogenides, which have been studied quite maturely. Till date, there have been scant reports on the growth of large-scale and large-size monolayer SnS2. Here, we successfully synthesized monolayer SnS2 crystal on SiO2/Si substrates via NaCl-assisted CVD and the edge can be as long as 80 μm. Optical microscope, Raman spectroscopy, x-ray diffraction, atomic force microscopy (AFM), and energy-dispersion x-ray (EDX) were performed respectively to investigate the morphology, crystallographic structure, and optical property of the 2D SnS2 nanosheets. In addition, we discussed the growing mechanism of the NaCl-assisted CVD method.
Recently, two-dimensional (2D) layered metal dichalcogenides (LMDs) such as molybdenum disulfide (MoS2) have attracted significant attention due to their electronic structure, super-large specific surface, quantum effect compared to the bulk materials.[1,2] They have great application prospects in the fields of optics, electronics, catalysis, etc. Until now, more 2D LMDS such as MoS2[3] and WS2[4] have been extensively studied in terms of preparation technology and potential applications in field effect transistors (FETs), photodetectors, solar cells, and flexible devices.[5–13] However, the investigation of 2D SnS2 which is an important number of IV–VI A group is still in its nascent stage.[14,15]
SnS2 is consisted of planar three-fold layers (TLs), where strong covalent bonding exists in plane but weak van der Waals interaction dominates out of plane. SnS2 has a large bandgap of ∼ 2.5 eV and an octahedral lattice made of two atomic layers of sulfur and one atomic layer of tin (shown in Fig.
Up to now, most of the reported SnS2 nanosheets are synthesized via exfoliation from bulk crystals,[22] chemical vapor deposition (CVD),[17,23,24] hydrothermal methods,[14] and spin coating techniques.[25] The most common methods are exfoliation and CVD. However, the size of layered SnS2 obtained by exfoliation is usually limited because the thickness and size are difficult to control in this way. More importantly, the resultant drawbacks, such as less controllability of the uniform morphology, sever clustering, poor crystallinity, or poor harvest, may impact the device performance (low responsivity or slow response speed).[15]
Chemical vapor deposition has been proposed as an effective way to synthesize various large-scale atomic layered 2D materials because of the advantage of the precise control on morphology, defects, and structure of the final products, particularly on large-area growth of 2D materials such as MoS2 and graphene.[26] Herein, we report a facile and repeatable method to synthesize large-size and high-quality monolayer SnS2 crystal. Recently, NaCl-assisted CVD has been proved to lower the reaction requirements effectively for synthesizing some 2D layer materials.[27] In the reports so far, no one has used this method to synthesize 2D group IV metal chalcogenides (GIVMCs). Motivated by this, by mixing NaCl with tin oxalate (SnC2O4)[28] powder as the precursor, we successfully synthesized large-size and high quality monolayer SnS2 on SiO2/Si substrates. The monolayer SnS2 can be as long as 80 μm. The Raman peak position shift and intensity change are observed as a function of the thickness and illustrate that the synthesized SnS2 crystals have a 2H phase.[15] Atomic force microscopy (AFM) was used to prove that the synthesized SnS2 nanosheets are of single atom layer.[20] The x-ray diffraction (XRD) and energy-dispersion x-ray (EDX) images can be well indexed to a pure SnS2 crystal phase without any detectable impurities such as SnS, Sn, Sn2S3, Na and Cl elements.[29] In summary, we have used NaCl-assisted CVD method to synthesize the large-size and high quality monolayer SnS2. Until now, there are only a few reports on the growth of atomically thin SnS2 layers. These jobs will provide inspiration for future researchers.
Large-size SnS2 monolayer was synthesized via an optimized CVD method (diagrammatic sketch shown in Fig.
Oxides and iodides like SnO2 and SnI2 are difficult to evaporate because of their extremely high melting points about 850 °C and 700 °C.[27,31,32] During the heating process, SnC2O4 will decompose into tin oxide. Mixing tin oxide and NaCl will produce a molten solution. The vapor pressure of the molten salt is much higher than that of many metal oxides. A general reaction can be written as
 |
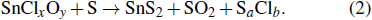 |
Figures
Two-dimensional SnS2 were synthesized on SiO2/Si substrates by chemical vapor deposition method inside a high-temperature horizontal tube furnace with a 4.5-cm inner diameter and a 98-cm-long heating zone. A mixture of SnC2O4 powder (purity 99.9%, Alfa Aesar) and NaCl powder (purity 99.9%, Alfa Aesar) with a mass ratio of 8:1 in an alumina boat with a top face down SiO2/Si substrate was placed at the center of the quartz tube (the heating zone). The sulfur (500 m g) was placed in an alumina boat 15 cm upstream from the Sn precursor, where the temperature was lower, which was around 200 °C during the growth. Before heating, high purity Ar was flow through the system at a rate of 600 standard cubic centimeter per minute (sccm) for 40 min to eliminate oxygen in the furnace. Then the temperature of the center zone was heated to 600 °C in 25 min and maintained at this temperature for 5–10 min. Argon (100 sccm) was used as the carrier gas and to maintain an inert atmosphere. In the cooling step, the furnace was cooled to 200 °C with a cooling rate of 3 °C/min and then rapidly cooled to room temperature.
In summary, we synthesized large-size and high-quality monolayer SnS2 with the edge length of up to 80 μm via an CVD method by NaCl-assistant. As we know, no method for stable preparation of monolayer SnS2 has been reported. Optical microscope, AFM, SEM, XRD, and EDX were used to characterize the size, semblance, thickness, and composition of the sample. NaCl can lower the melting points of Sn precursors and increase the vapor pressure of the Sn precursor. These changes lead to a higher nucleation rate. This method might pave the way for the electronics and optoelectronics applications and inspire other researchers.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] |





