Project supported by the National Natural Science Foundation of China (Grant Nos. 11772069 and 11872003).
Project supported by the National Natural Science Foundation of China (Grant Nos. 11772069 and 11872003).
† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11772069 and 11872003).
The pre-Bötzinger complex (pre-BötC) in mammalian brainstem is essential for the generation of respiratory rhythms. Most dynamic studies on the pre-BötC neuron have been focused on its firing activities modulated by the ion conductances rather than that by the electromagnetic radiation or the external forcing current. In this paper, by adding the electromagnetic radiation and external forcing current to Park and Rubin’s model, we mainly investigate the influences of those two factors on the mixed bursting (MB) of single pre-BötC neuron. First, we explore how the variation of external forcing current affects the MB patterns of the system with non-vanishing magnetic flux. We classify the MB patterns and show their dynamic mechanism through fast-slow decomposition and bifurcation analysis. Then, by modifying the feedback coefficient, we further analyze the sole effect of electromagnetic radiation on the firing activities of the system. Our results may be instructive in understanding the dynamical behavior of pre-BötC neuron.
Smith et al. confirmed that the pre-Bötzinger complex (pre-BötC) located in the brainstem is involved in the generation of respiratory rhythm in the mammalian nervous system.[1] To date, a number of studies have indicated that the pre-BötC is essential for normal breaths and sighs.[2–7] There is still no consensus, however, regarding the mechanism of the generation and modulation of the respiratory rhythm in humans, or furthermore, what causes respiratory rhythmic disease by interfering with these mechanisms. A better understanding towards the intrinsic dynamics of the pre-BötC can play an important role in addressing these issues.
Based on a single-compartment Hodgkin–Huxley (HH) formalism, Butera and his collaborators established two conductance-based models for both single as well as two-coupled cells in the pre-BötC.[8,9] Their models have been widely used to explore the general principles and mechanisms for rhythmic bursting of pre-BötC pacemaker cells. Based on Butera’s model, Best et al. explored the influences of parameters gtonic-e and gsyn-e on the network behavior.[10] Duan et al. compared the bursting patterns between the single and two-cell model network using dynamic analysis methods.[11]
Toporikova and Butera created a two-compartment model of an inspiratory pre-BötC neuron (TB model), whose somatic compartment developed from the previously described model and the dendritic compartment used the Li–Rinzel model which described the dynamics of Ca2+.[12,13] Considering that the voltage and the calcium-activated nonspecific cationic current can reciprocally regulate each other, Park and Rubin simplified the TB model and proposed a single-compartment model of pre-BötC inspiratory neuron.[14] They also showed five activity patterns in the two-parameter ([IP3], gNaP) space, among which we are mostly interested in the somatic–dendritic bursting that is usually presented as the mixed bursting (MB) in experimental observations.[15] Researchers have attempted to explain how ion conductances influence the MB patterns, and tried to explore the underlying dynamical mechanisms.[14,16,17]
According to the Faraday’s law of induction and the feedback effect, the electrical activities of neurons and the distribution of electromagnetic field inside and outside of neurons can influence each other. In fact, the neuronal system is sensitive to even slight stimulation applied to the neuron, and the continuous exchange of chemical ions (such as Na+ and K+) between the inside and outside of the nerve cell membrane can be triggered by an external stimulation thus putting out electrical pulses. Consequently, the dynamical behavior of neurons can change under the effect of magnetic flow or external forcing current. Li et al. built a mathematical model of neuron membrane current generated by electromagnetic radiation based on neuronal energy theory and then introduced it into the HH model.[18] Lv and Ma introduced the additive variable as magnetic flux into neuron model and discussed the electrical activities.[19,20] On the basis of Lv, Liu and Zhan explored the electrical activities of an improved M–L neuron model equipped with electromagnetic radiation and phase noise.[21] They found that the suprathreshold spiking of system is gradually reduced with the feedback coefficient k, which indicates that the coupling strength between magnetic flux and membrane potential is increased. Duan et al. investigated the effects of direct and alternating currents on dynamical activities of single pre-BötC neuron model featuring magnetic flux, and showed that regular bursting patterns are of different types with the direct current changed.[22] However, their analysis did not take into account the calcium dynamics in the model, nor did they examine the impact of the feedback coefficient of magnetic flux on the firing patterns. On the other hand, Gu et al. studied how the excitatory stimulus affects the number of spikes within a burst.[23] The difference is that we mainly focus on the effect of inhibition stimulus on the burst patterns.
As we have mentioned, most of the researches on pre-BötC dynamical systems focus only on the impact of ion channels on the firing activities of the systems, however, the dynamics of pre-BötC systems under magnetic flow effect are less considered. In this paper, based on a modified single-compartment pre-BötC model[14] appended with an external direct current and a magnetic flux, we would like to investigate the MB patterns of the system by changing the direct current. We would also elucidate the sole effect of magnetic flow on the system.
This paper has been organized as follows. Section
In this paper, we consider the following single compartment model of pre-BötC inspiratory neuron, which is a modification of Park and Rubin’s model by introducing the external forcing current I and the magnetic flux φ
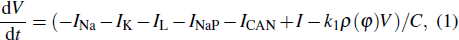 |
 |
 |
 |
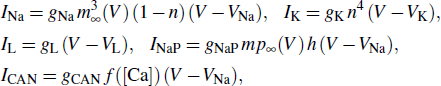 |
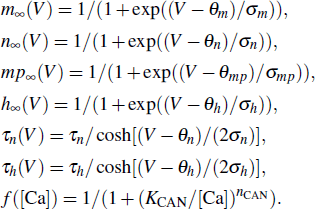 |
The calcium dynamics is given as
 |
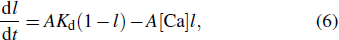 |
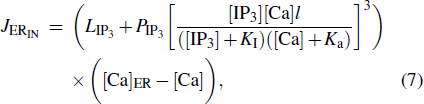 |
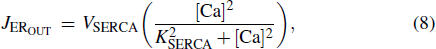 |
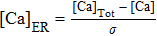
If setting k1 = 0, I = 0, the system in Eqs. (
In order to explore the influence of the external forcing current I under the magnetic flux on every MB solution, we firstly focus on the somatic part of each burst. In this section, we set parameter gCAN = 14 nS, k1 = 0.1. The bifurcation diagram of V with respect to the parameter I is given with fixed [Ca] = 0.022 in Fig.
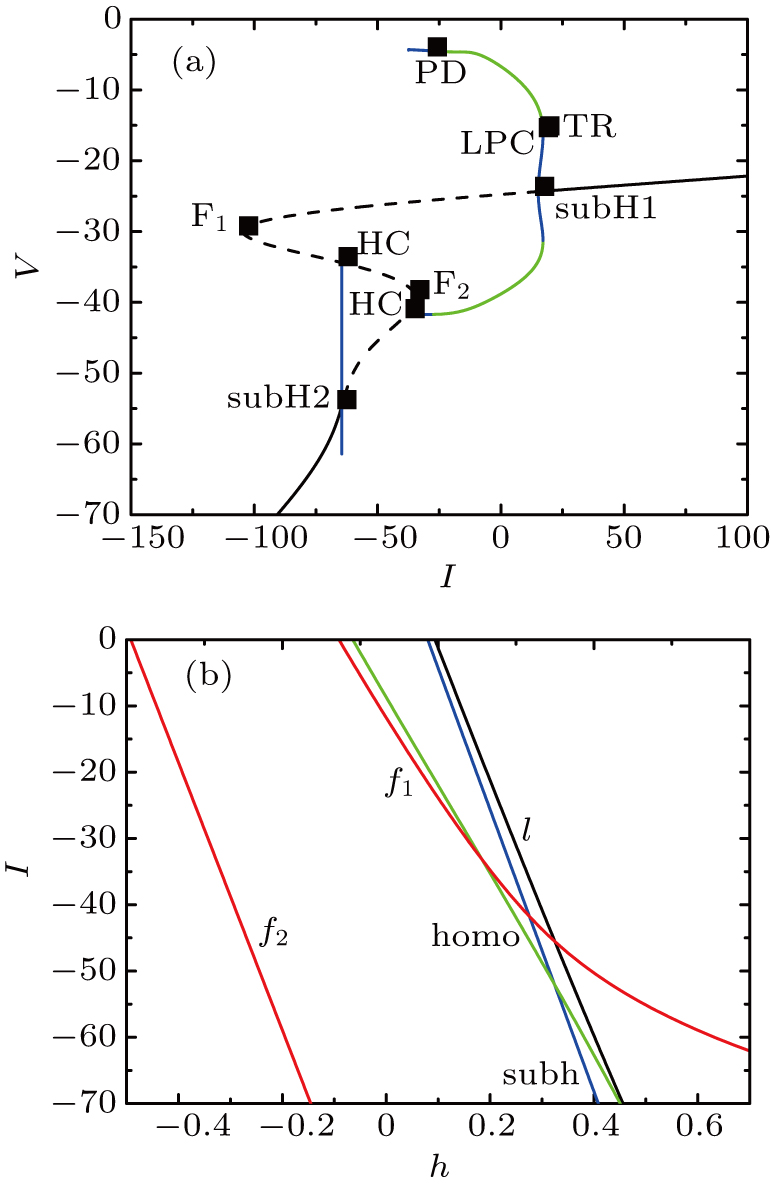
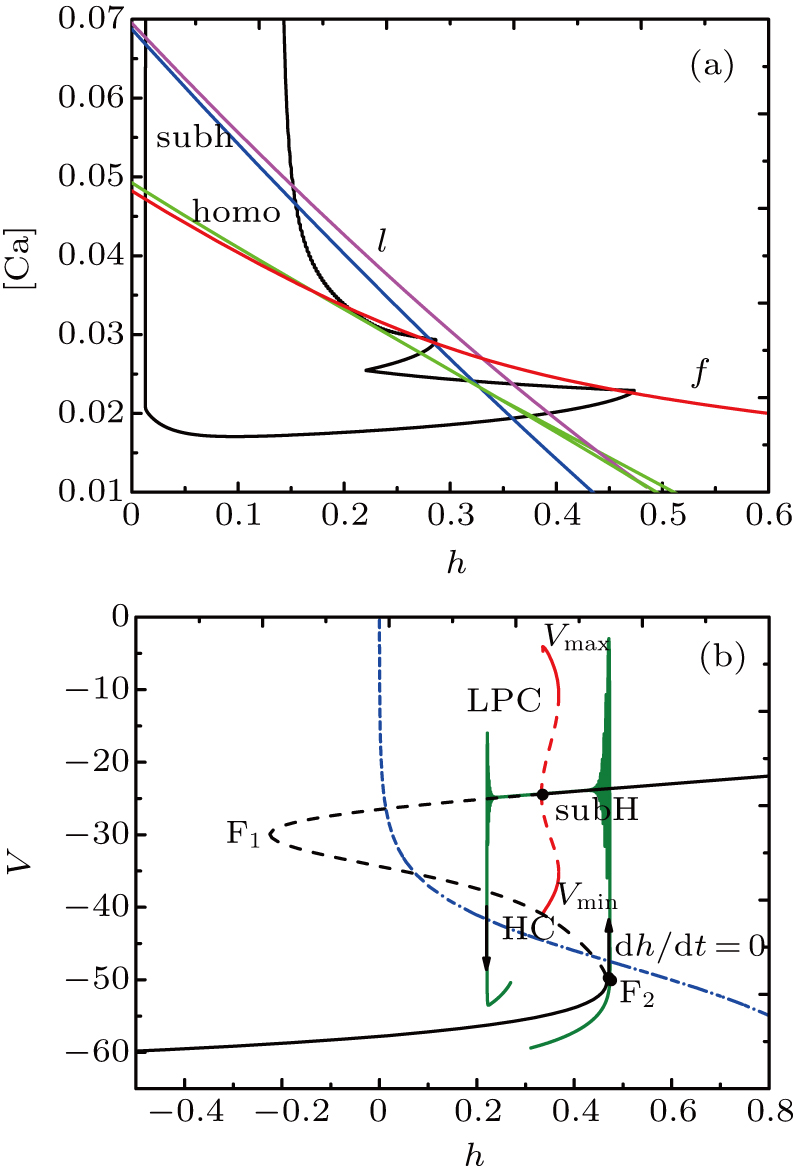
At [Ca] = 0.022, the two-parameter bifurcation diagram with respect to the slow variable h and the parameter I is shown in Fig.
Since the bifurcation structure is more complex when I∈[−55, 0], in the following discussions, we will restrict the range of external forcing current I to the interval [−55, 0]. As I changes, we will numerically simulate the dynamic system and perform a detailed bifurcation analysis for several types of MB.
The firing pattern without I is shown in Fig.
In order to investigate the dynamical mechanism of this MB, we separate the full system into two subsystems, i.e., the fast subsystem in Eqs. (
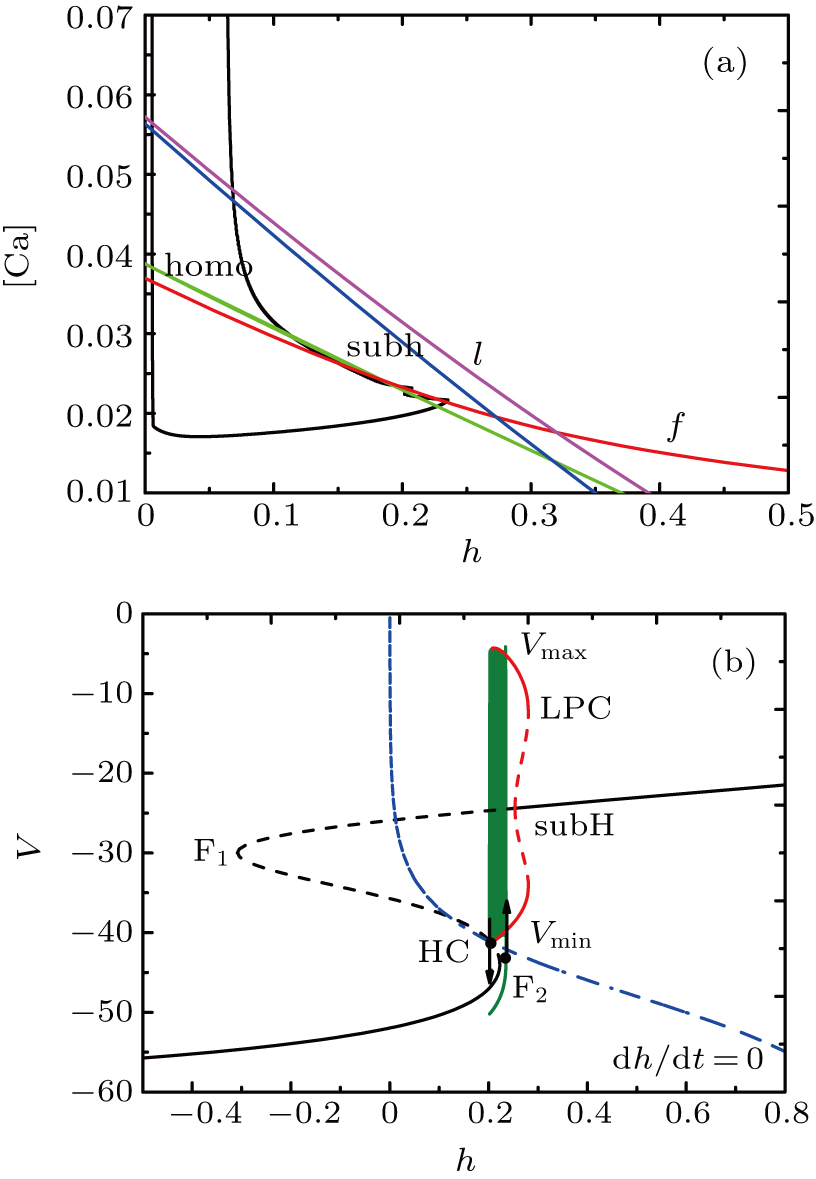
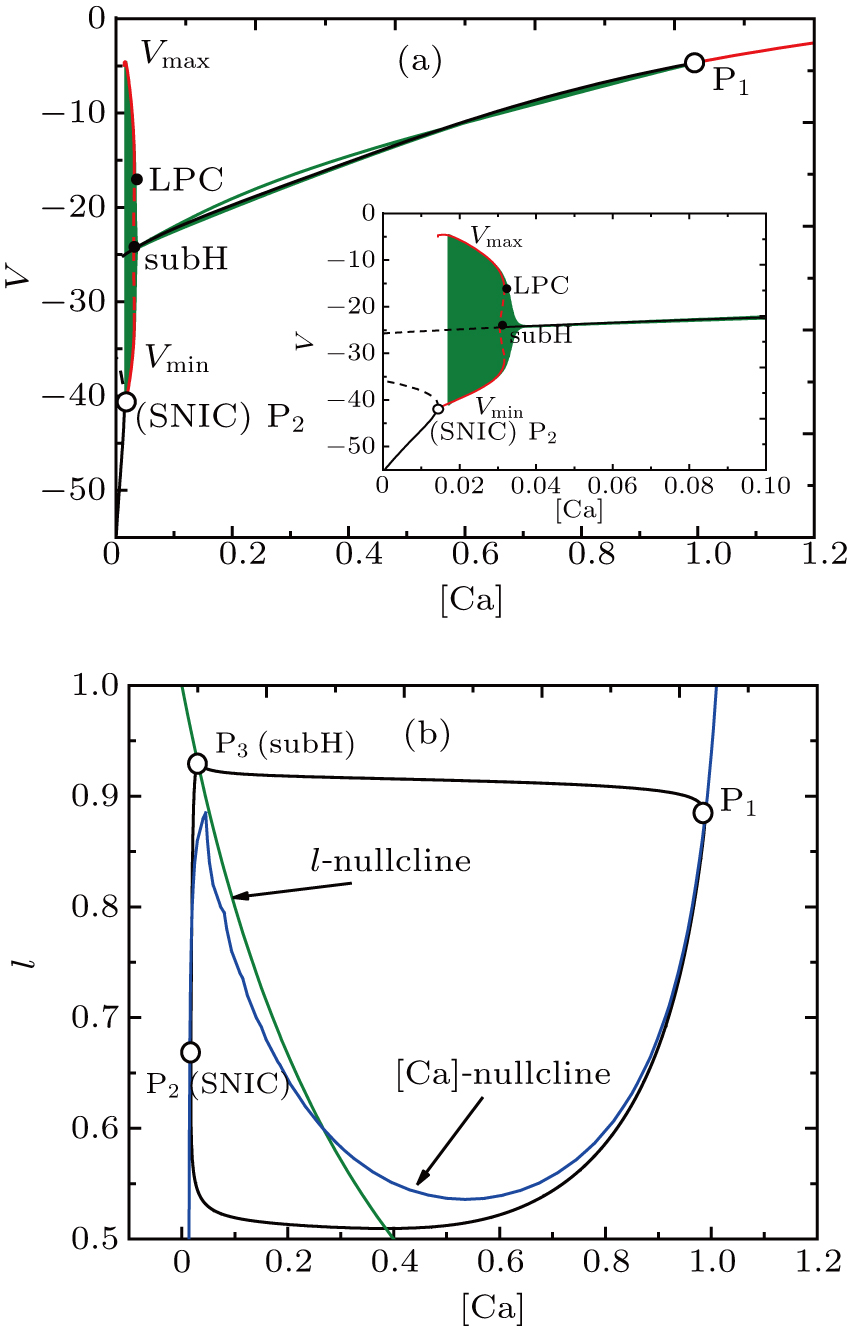 | Fig. 3. The generation of full system bursts can be triggered by dendritic relaxation oscillations.[13] (a) Bifurcation diagram of the fast subsystem with bifurcation parameter [Ca]. The solution of the full system when I = 0 (olive) is also superimposed. The bursting arises through a saddle-node on an invariant circle bifurcation and disappears via subcritical Hopf bifurcation. (b) The trajectory in the dendritic subsystem when [IP3] = 0.96. [Ca]-nullcline is the S-shaped curve in blue and l-nullcline is the olive diagonal curve cutting through the figure. |
The corresponding periodic orbit in ([Ca], l)-space is shown in Fig.
In this subsection, we show how the dynamical behaviours of the model vary with the change of I ∈ [−55, −20]. We use fast-slow decomposition and bifurcation analysis to classify each bursting in somatic part of every MB pattern and further reveal the mechanism of MB. When considering the somatic part of MB, we treat h as a slow variable and settle [Ca] as constant. Therefore, equations (
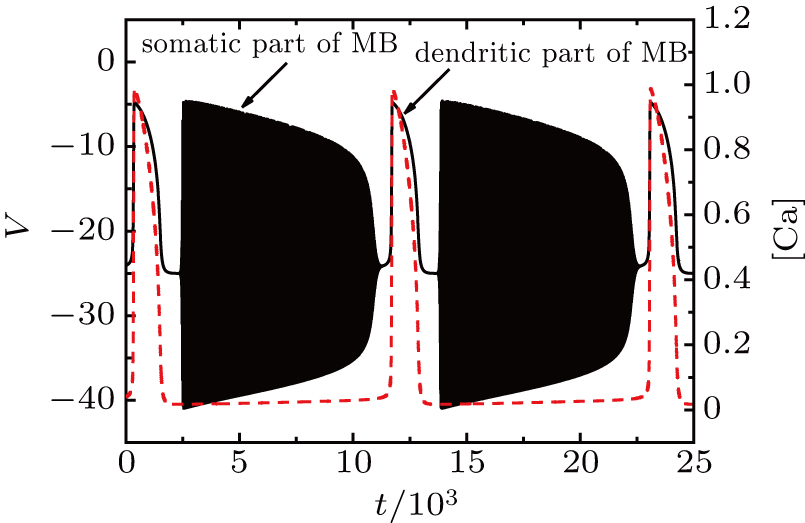
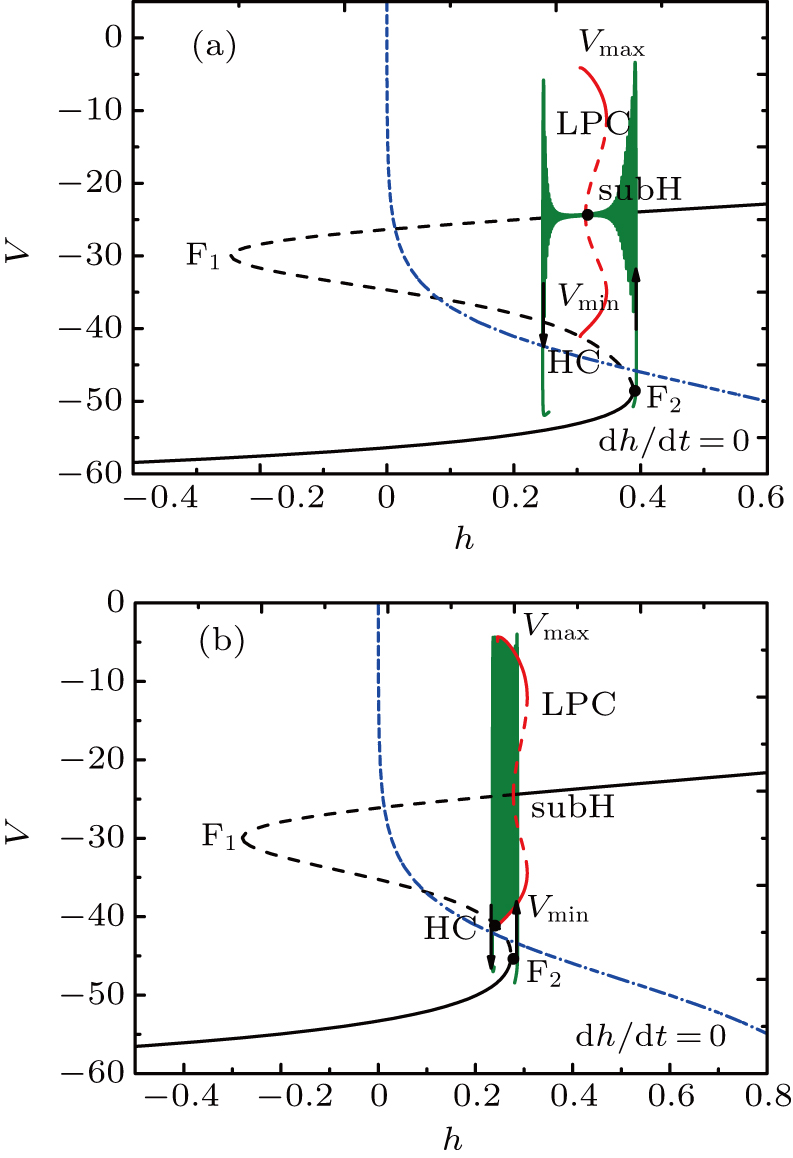
At I = −20 μA, the full system displays MB while the somatic part exhibits spiking activity, as shown in Fig.
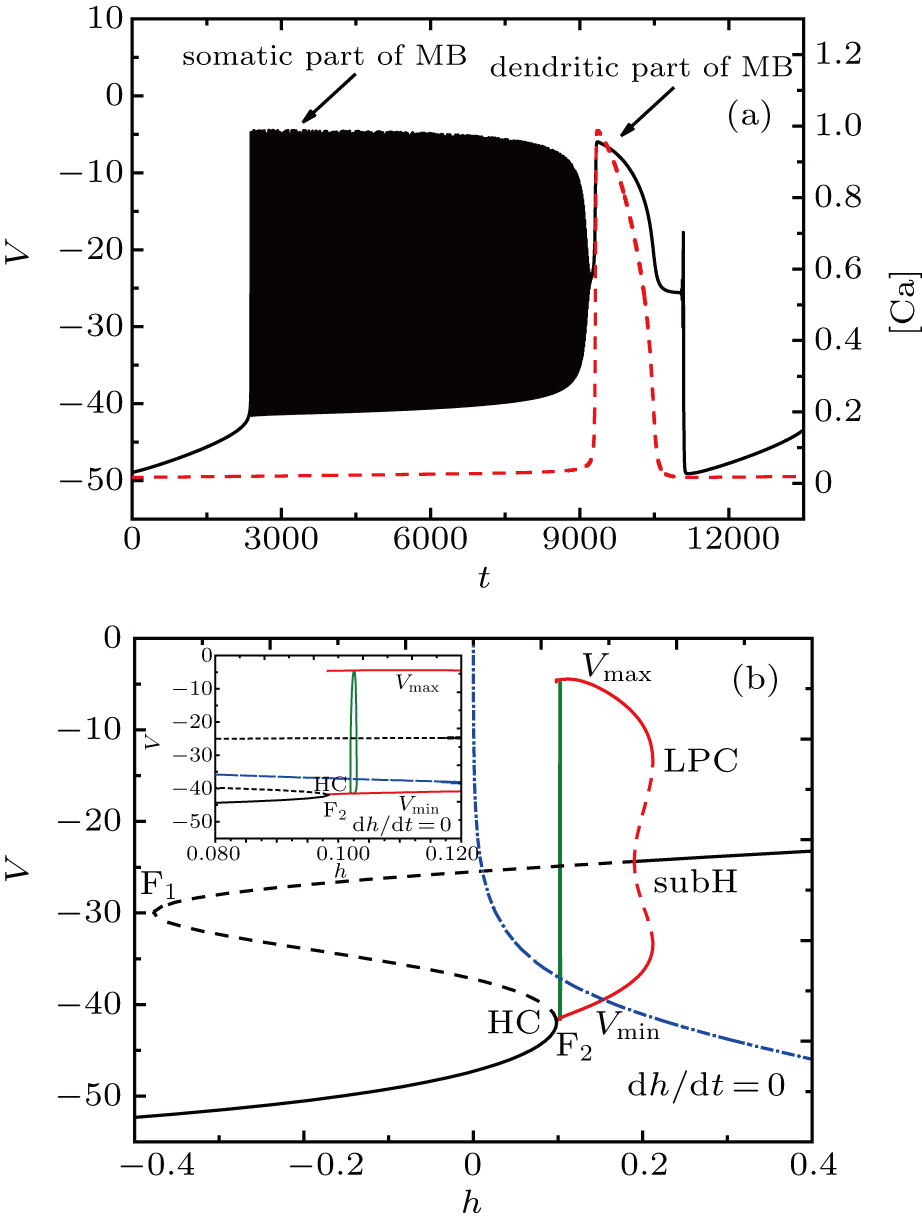 | Fig. 4. (a) MB pattern obtained with I = −20 μA and default parameters. (b) Fast-slow decomposition and bifurcation analysis on somatic part of the MB. The somatic part of MB exhibits spiking. |
With the decrease of I, the somatic part of MB displays different patterns of bursting. When fixing I = −37 μA, the full system exhibits the MB pattern as shown in Fig.
The two-parameter bifurcation diagram is shown in Fig.
The bifurcation diagram of the fast subsystem in Eqs. (
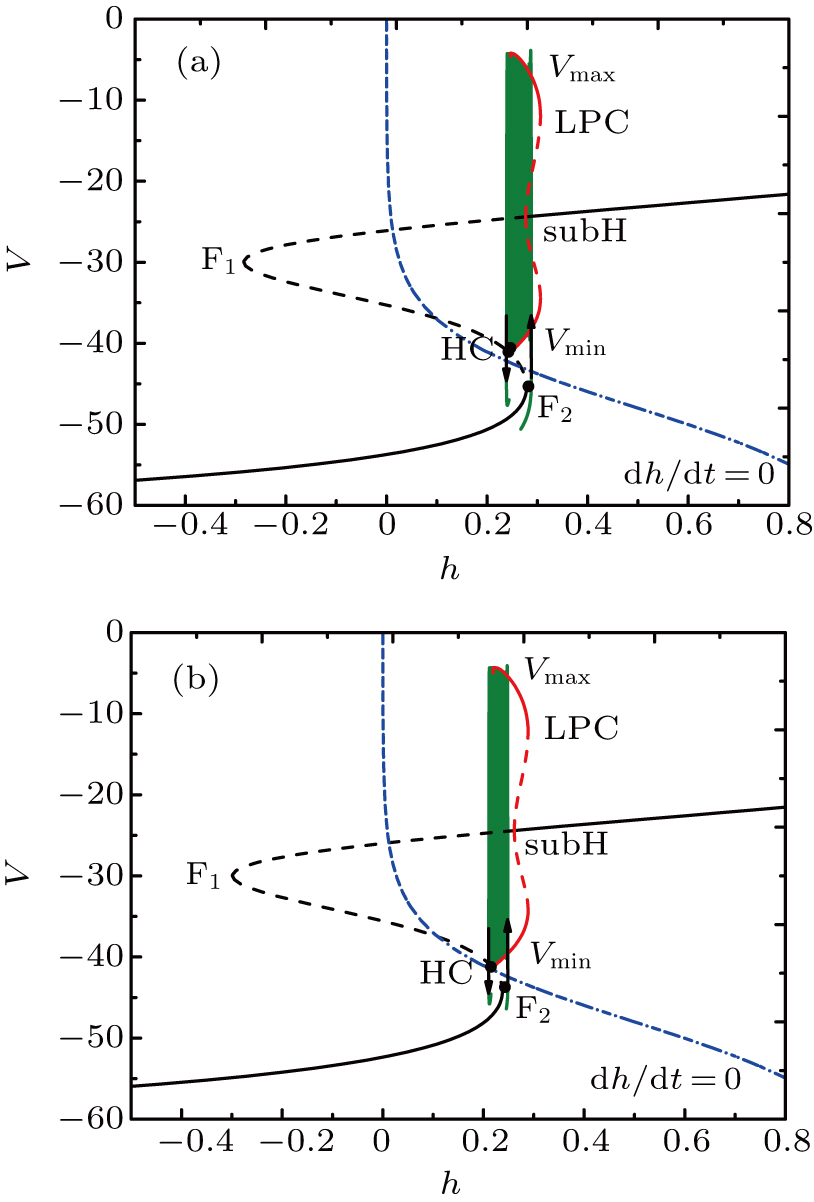
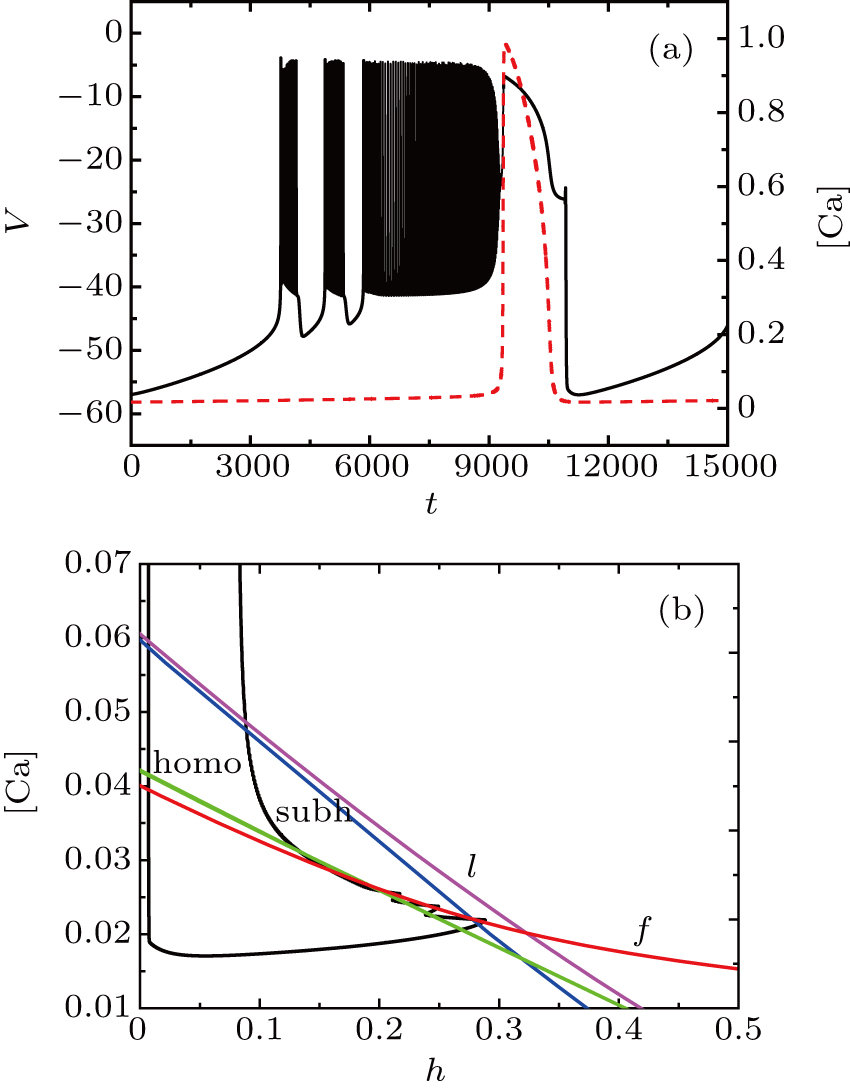
Unlike the earlier case, the somatic part of MB contains spiking and two burstings when I = −42 μA, as shown in Fig.
 | Fig. 7. (a) MB pattern obtained with I = −42 μA and default parameters. (b) The two-parameter bifurcation diagram on the ([Ca], h) parameter space. |
The two bursts in somatic part correspond to [Ca] = 0.022 and [Ca] = 0.024, respectively. Figure
If setting I = −48 μA, the full system exhibits the MB pattern as shown in Fig.
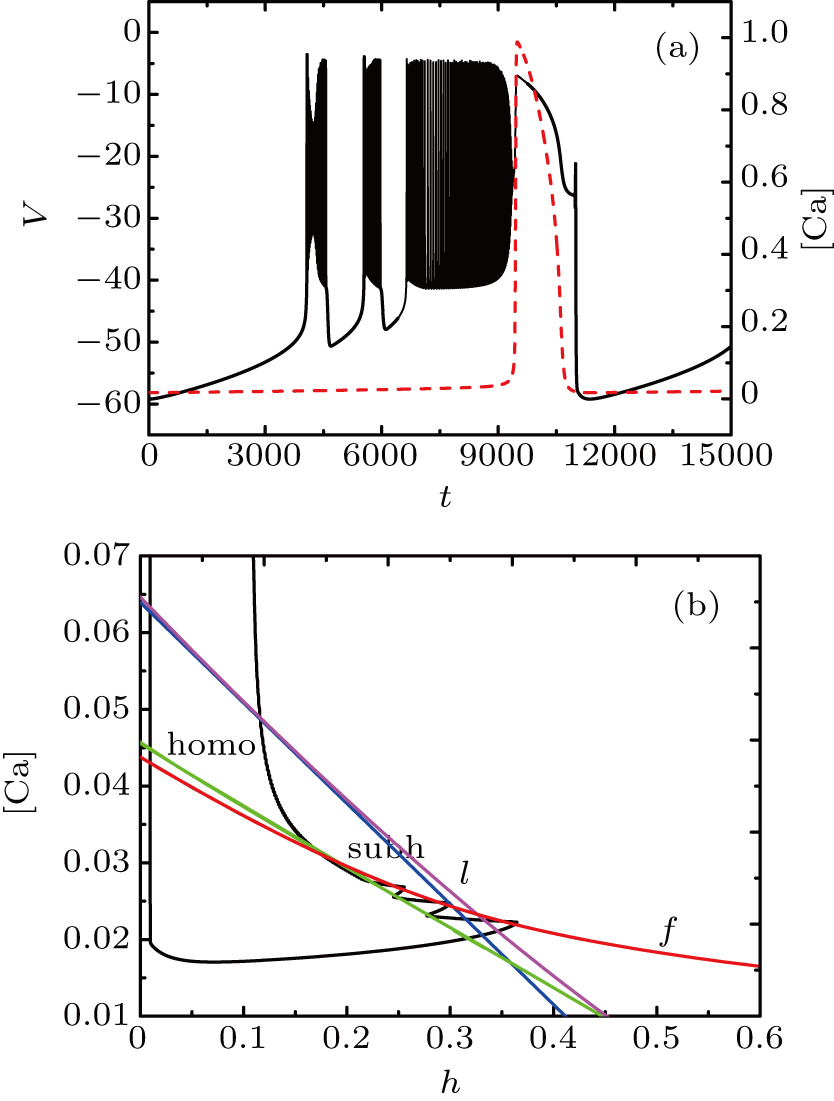 | Fig. 9. (a) MB pattern obtained with I = −48 μA and default parameters. (b) The two-parameter bifurcation diagram on the [Ca], h) parameter space. |
Although the bifurcation curves in the parameter region are the same as before, they have changed the relative position with the trajectory of the full system. It seems that the bursts in somatic part are related to the fold bifurcation, subhopf bifurcation, fold bifurcation of limit cycle, and the homoclinic bifurcation, while the spiking is near the homoclinic bifurcation. To obtain more information about the somatic part of MB, we consider the one-parameter bifurcation analysis.
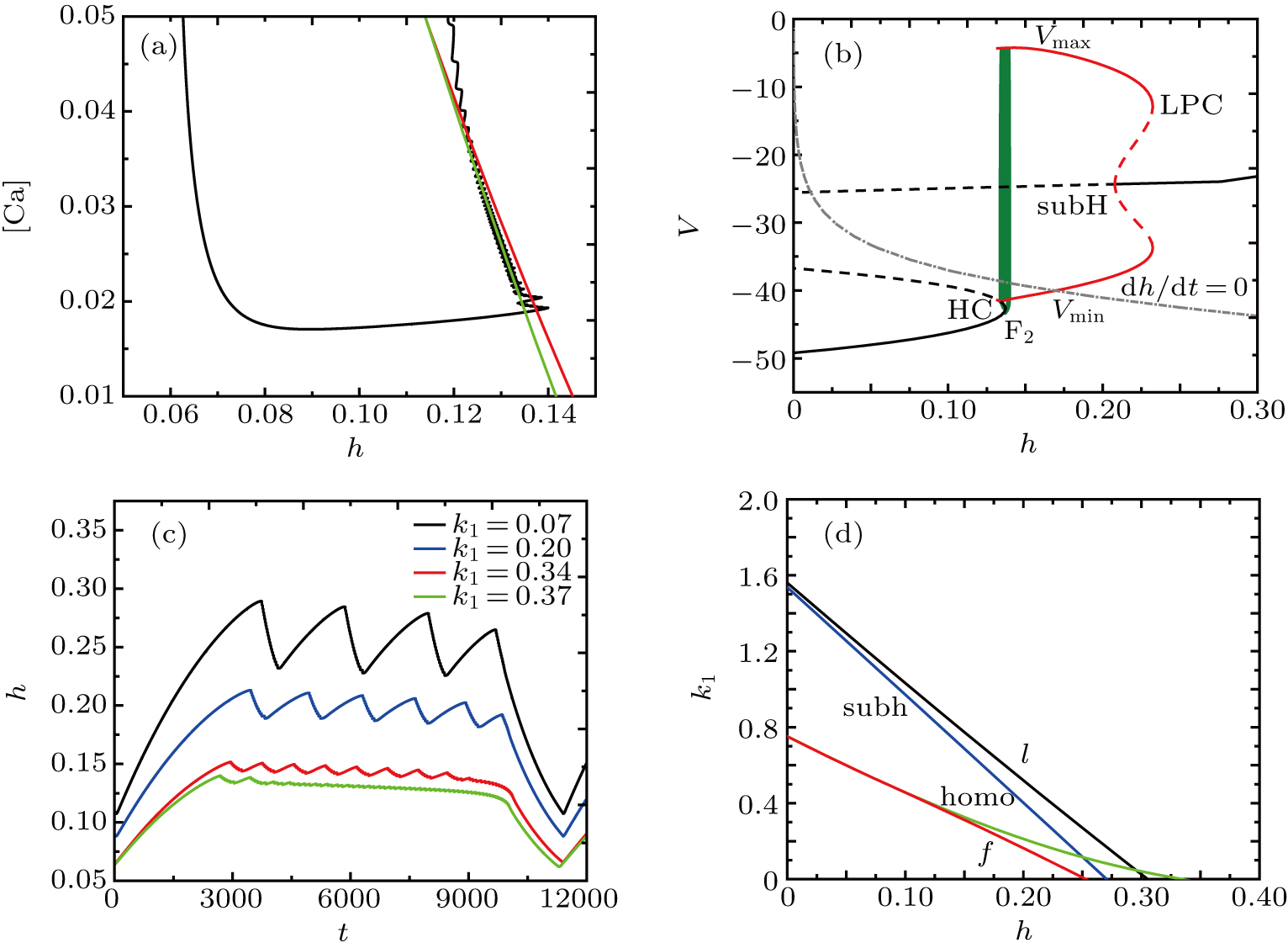
Figure
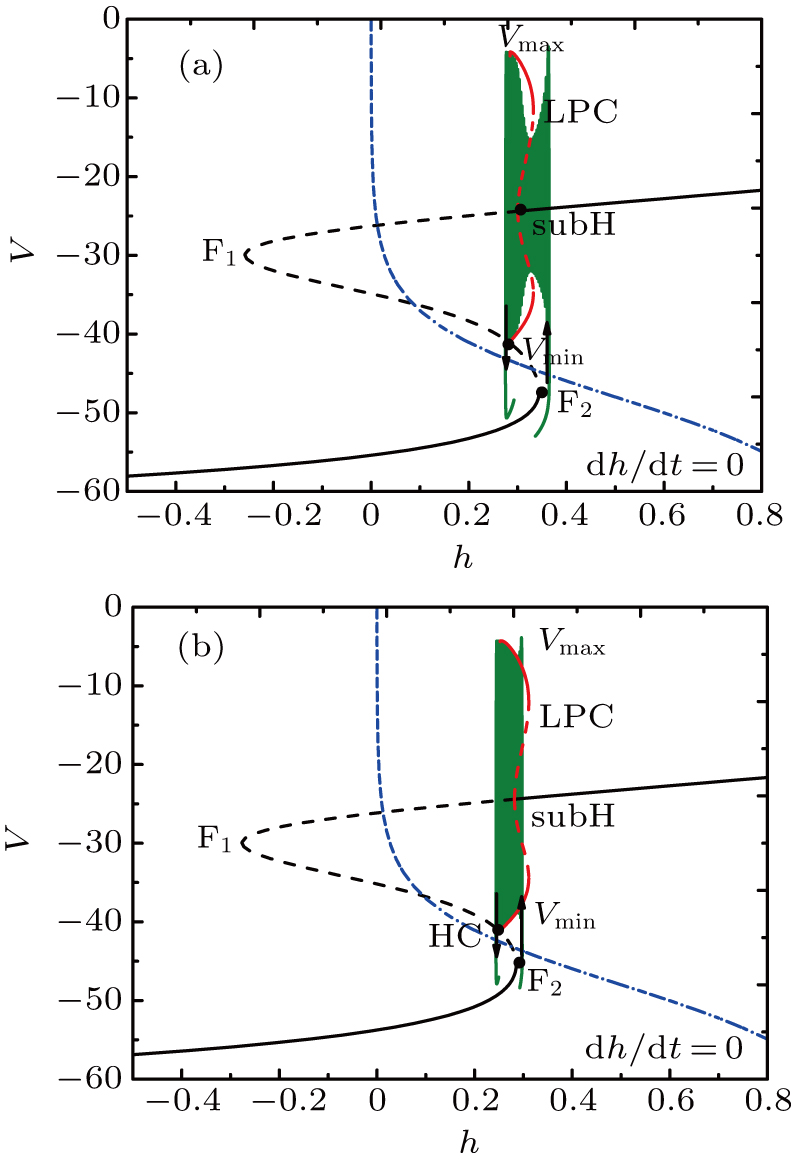
For fixed I = −50 μA, the full system exhibits the MB pattern as shown in Fig.
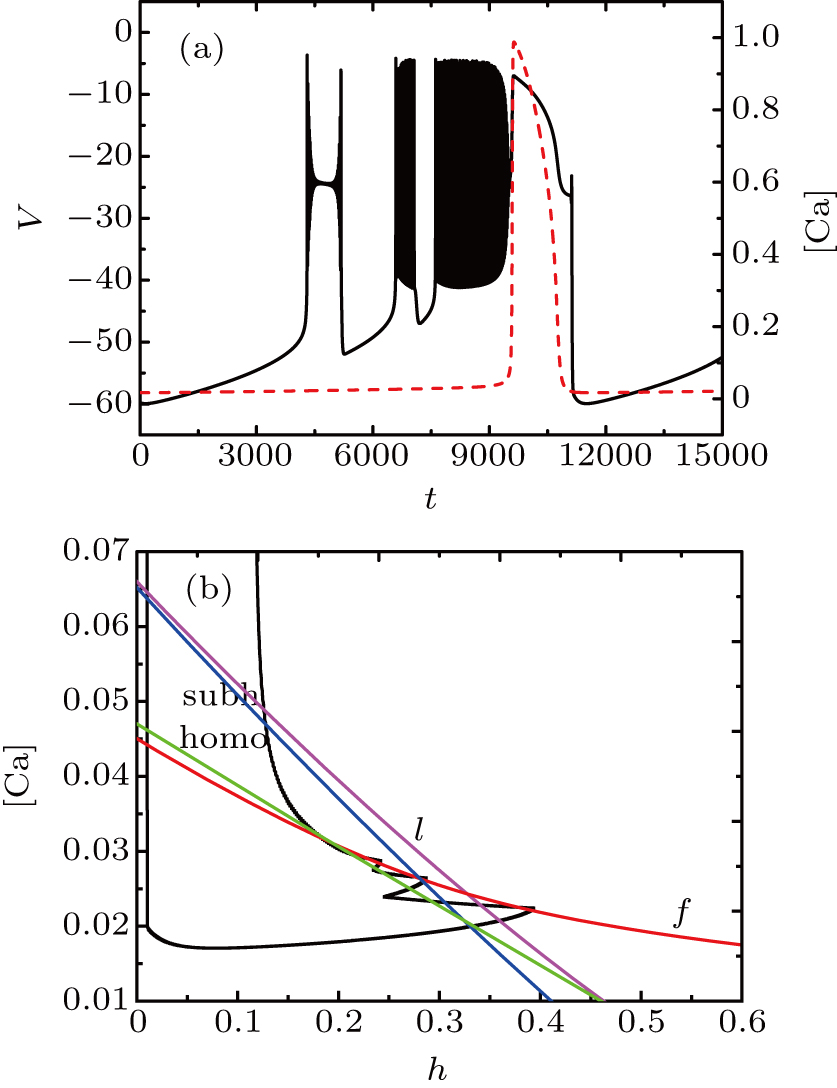 | Fig. 11. (a) MB pattern obtained with I = −50 μA and default parameters. (b) The two-parameter bifurcation diagram on the ([Ca], h) parameter space. |
While the bifurcation diagram is so similar to that at I = −48 μA, the homoclinic bifurcation curve intersects the first bursting trajectory of the full system at a more right position contrasted with the previous case.
The fast-slow decomposition and bifurcation analysis on the somatic part of MB imply that the first bursting is “subHopf/subHopf” bursting via “fold/homoclinic” hysteresis loop, while the the second bursting is “fold/homoclinic” bursting (see Fig.
The spiking in somatic part has no difference with the previous cases. When I decreases to −55 μA, it is interesting that the somatic part of MB contains only one burst again, as shown in Fig.
Let us now turn to discuss the influence of magnetic flow effect on the membrane potential of pre-BötC neuron by varying the parameter k1. In this section, we set gCAN = 0.7 nS, I = 0.
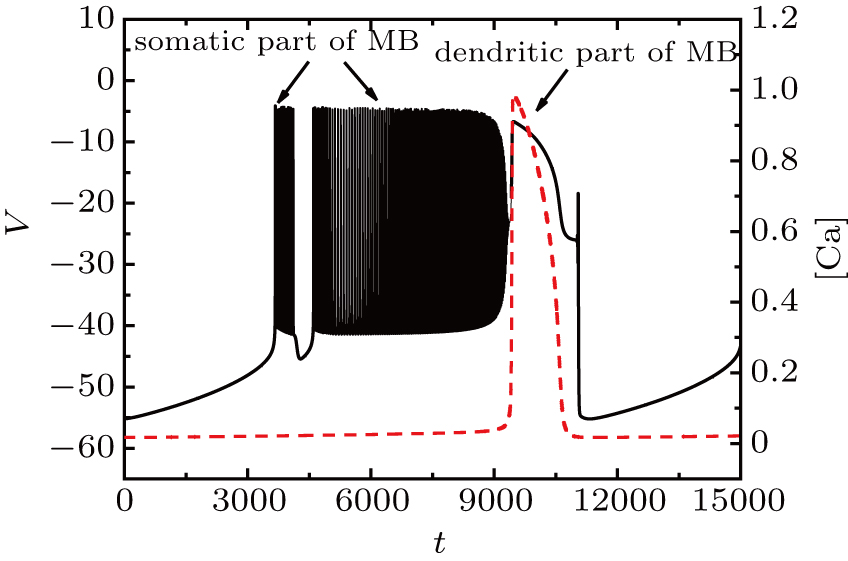
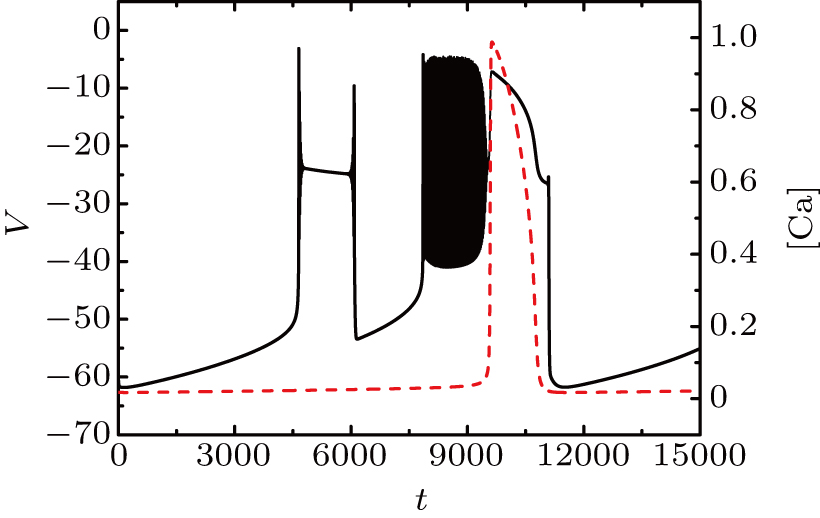
Figure
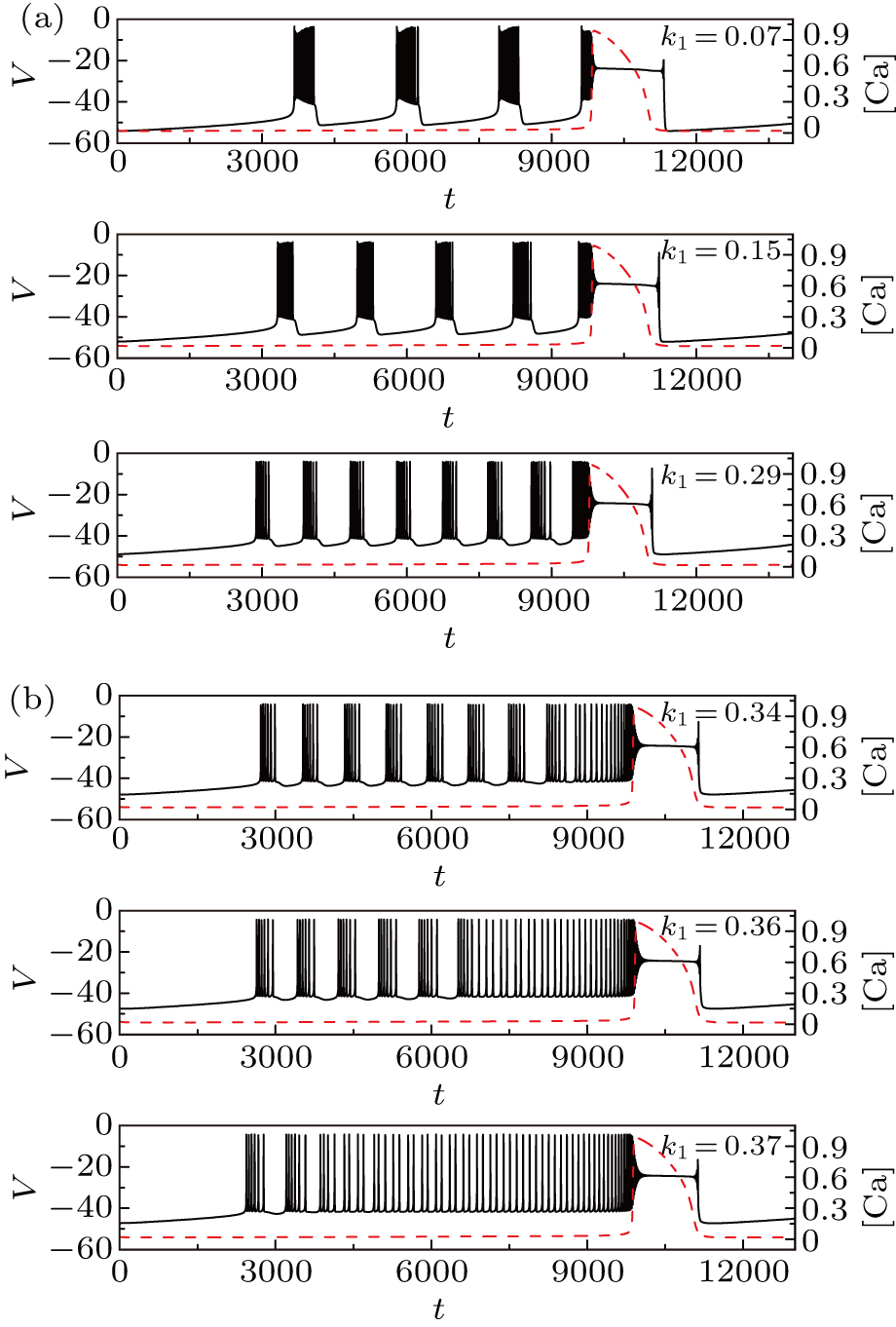 | Fig. 15. Time series of the membrane potential of the modified model with different values of k1. The red dashed curve represents the time series of intracellular calcium concentration [Ca]. |
We take the case of k1 = 0.37 as an example to discuss the mechanism of bursting in the somatic part. Figure
To explore the mechanism why the frequency (number) of bursting in somatic part increases as k1 increases, we show the two-parameter bifurcation diagram of the somatic subsystem in Fig.
The effect of magnetic flux and external current on dynamical behaviors of the pre-BötC neuron model has not been considered previously. In this paper, by adding the terms of magnetic flux and direct currents to a single-compartment pre-BötC neuron model, we aim to assess how the two factors influence the mixed bursting of the system. Numerical simulations have revealed that under nonzero magnetic flux, the variation of the direct current I leads to the conversion of mixed bursting from one type to another. On the other hand, if setting I = 0, the change of feedback coefficient k1 related to magnetic flux leads to the change of the frequency of somatic part of mixed bursting. We have also explored the dynamical mechanism of different mixed burstings. The fast-slow decomposition and two-parameter bifurcation analysis are the main methods we have adopted. Our results help to better understand the dynamic properties of pre-BötC system containing magnetic flux and external current terms. Also, the analysis may broaden our understanding of the mixed bursting.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] |