Project supported by the National Natural Science Foundation of China (Grant Nos. 11574276, U173112, and 41401384), the Project of Shandong Provincial Higher Educational Science and Technology Program, China (Grant No. J17KB127), the Science and Technology Development Plans of Binzhou City, China (Grant Nos. 2014ZC0307 and 2015ZC0210), and Binzhou University Research Fund Project, China (Grant Nos. BZXYG1513 and BZXYG1706).
Project supported by the National Natural Science Foundation of China (Grant Nos. 11574276, U173112, and 41401384), the Project of Shandong Provincial Higher Educational Science and Technology Program, China (Grant No. J17KB127), the Science and Technology Development Plans of Binzhou City, China (Grant Nos. 2014ZC0307 and 2015ZC0210), and Binzhou University Research Fund Project, China (Grant Nos. BZXYG1513 and BZXYG1706).
† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11574276, U173112, and 41401384), the Project of Shandong Provincial Higher Educational Science and Technology Program, China (Grant No. J17KB127), the Science and Technology Development Plans of Binzhou City, China (Grant Nos. 2014ZC0307 and 2015ZC0210), and Binzhou University Research Fund Project, China (Grant Nos. BZXYG1513 and BZXYG1706).
The Zr0.5Hf0.5VPO7 is successfully synthesized by the solid-state method with near-zero thermal expansion. Powder x-ray diffraction (XRD), Raman spectroscopy, thermal dilatometry, and scanning electron microscopy (SEM) are used to investigate the structure, the phase transition, and the coefficient of thermal expansion (CTE) of Zr0.5Hf0.5VPO7. The investigation results show that the samples are of the single cubic type with a space group of 
Negative thermal expansion (NTE) materials have the characteristics of heat shrinkage and cold rise, which can effectively control the expansion coefficient of material. The NTE material means it is possible to prepare composite materials each with an arbitrary expansion coefficient. The stress caused by thermal expansion and contraction is often the main cause of fatigue, performance decline, failures, and even breaking and falling off of the device. The NTE materials have extensive applications in many fields, such as optics, electronics, optical fiber communications, medicine, daily life, and many other fields. The NTE materials provide an effective way to control the thermal expansion coefficient and temperature range.[1–9] A series of NTE materials such as AM2O8 (A = Zr, Hf; M = W, Mo),[1–6] AM2O7 (A = Zr, Hf; M = V, P),[7–12] and A2M3O12 (A = Y, Sc, Fe; M = W, Mo) has been reported.[13–19] Recently researchers investigated the materials with near-ZTE, which include Mn3Cu0.5A0.5N (A = Ni, Sn)/Cu composites,[3] Mn3Ga0.5Ge0.5N,[4] ZrW2O8/A (A = ZrO2, Al2O3, etc.) composites,[5–7] Zn4B6O13,[20] ZrFeMo2VO12,[21] and HfMgMo3O12.[22] The materials of MV2O7 (M = Zr, Hf) have been extensively explored to reduce the phase transition temperature.[1,7] The AM2O7 family within the cubic 
In this paper, a near-zero thermal expansion material of Zr0.5Hf0.5VPO7 is synthesized by the solid state reaction method. Thermal dilatometry, XRD, and Raman spectra are used to investigate the CTE, anisotropy, and phase transition of Zr0.5Hf0.5VPO7. It is found that the materials have the stable near-zero thermal expansion in a wide temperature range.
Zr0.5Hf0.5VPO7 samples were synthesized by the solid-state reaction method. The stoichiometric quantities of commercial chemicals of ZrO2 (99%), HfO2 (99%), (NH4)H2PO4 (99%), and V2O5 (99%) were ground together thoroughly with an agate mortar for 2 h. The mixed powder was heated from room temperature (RT) to 1023 K in a furnace at a rate of 5 K/min, maintained at 1023 K for 5 h, and cooled down slowly in the furnace. The sintered powder was reground, pressed into pellets with a thickness of about 10 mm and a diameter of 10 mm, and then sintered again at 1023 K for 5 h. These processes were repeated twice to implement the complete reaction. The obtained compact pellets were kept for measuring their linear CTEs.
The samples were analyzed by XRD with a PANalytical X’Pert PRO x-ray diffractometer to identify the crystalline phase. Variable-temperature x-ray powder data were collected on a Rigaku (Japan, SmartLab 3 KW) diffractometer with Cu Kα (λ = 0.15405 nm) radiation. Unit cell dimensions above the phase transition temperature were determined with software of Powder X. The relative length changes at low temperature were measured with a LINSEIS DIL L75 dilatometer at heating and cooling rates of 5 K/min. The Raman spectra of the samples were tested by a LabRAM HR Evolution Raman spectrometer. The microstructures and energy dispersive spectra of the samples were examined with a scanning electron microscope (SEM, Model Quanta 250).
Figure
 | Fig. 1. (color online) (a) Raman spectra of (curve A) ZrV2O7, (curve B) Zr0.5Hf0.5VPO7, and (curve C) Zr0.5Hf0.5 V2O7. (b) Temperature-dependent Raman spectra of Zr0.5Hf0.5VPO7. |
The most obvious change of the Raman spectrum of Zr0.5Hf0.5VPO7 is the splitting of the symmetric stretching and asymmetric stretching modes of VO4 tetrahedron around 1101 cm−1–700 cm−1 in which there are strong peaks at 1101, 1061, and 972 cm−1 belonging to the symmetric stretching vibrations (ν1 modes) of VO4. The peaks at 887, 806, and 700 cm−1 belong to asymmetric stretching (ν3 modes) of the VO4. The peaks at 524 cm−1 and 474 cm−1 are ascribed to asymmetric bending (ν4 modes). While those peaks at 403, 367, 344, 330, and 301 cm−1 originate from symmetric bending (ν2) of the VO4. The peaks at 282, 99, and 55 cm−1 originate from lattice modes, which arise from Zr atom motion, translational and librational modes of VO4.[19]
Figure
Figure 
In order to confirm the crystal structure, the diffraction data are analyzed by the Rietveld and Le Bail methods in FullProf. The XRD data are refined in 
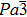

 | Fig. 3. (color online) Results of the Rietveld analysis of the XRD pattern for Zr0.5Hf0.5VPO7 at room temperature (Rp = 14.1, Rwp = 13.7, Rexp = 5.48, x2 = Rwp/Rexp = 2.5). |
| Table 1. Detailed XRD refinement results of Zr0.5Hf0.5VPO7. . |
The near-ZTE property of Zr0.5Hf0.5VPO7 is investigated with a dilatometer. Figure
 | Fig. 4. (color online) Relative length changes of (curve a) ZrV2O7 and (curve b) Zr0.5Hf0.5VPO7 with temperature increasing from 200 K to 673 K and temperature decreasing from 673 K to 310 K. |
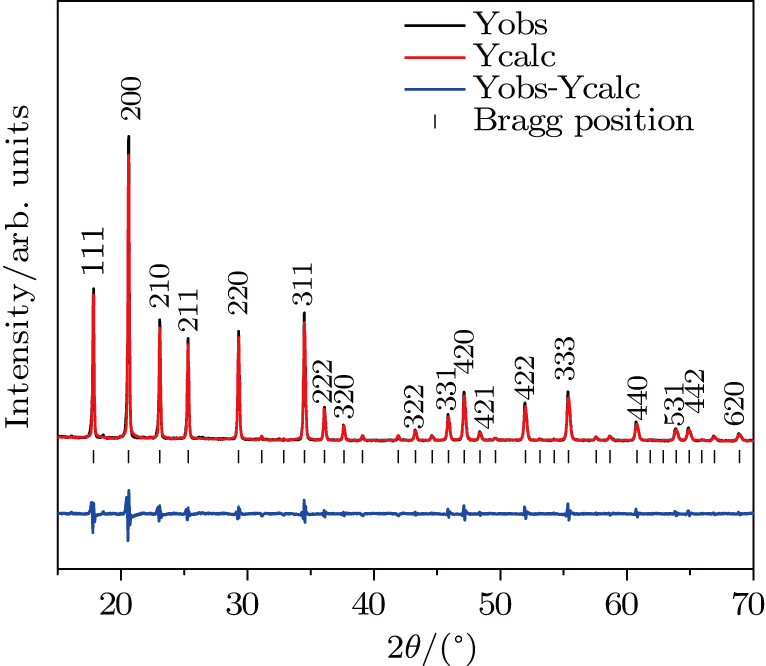
Figure 
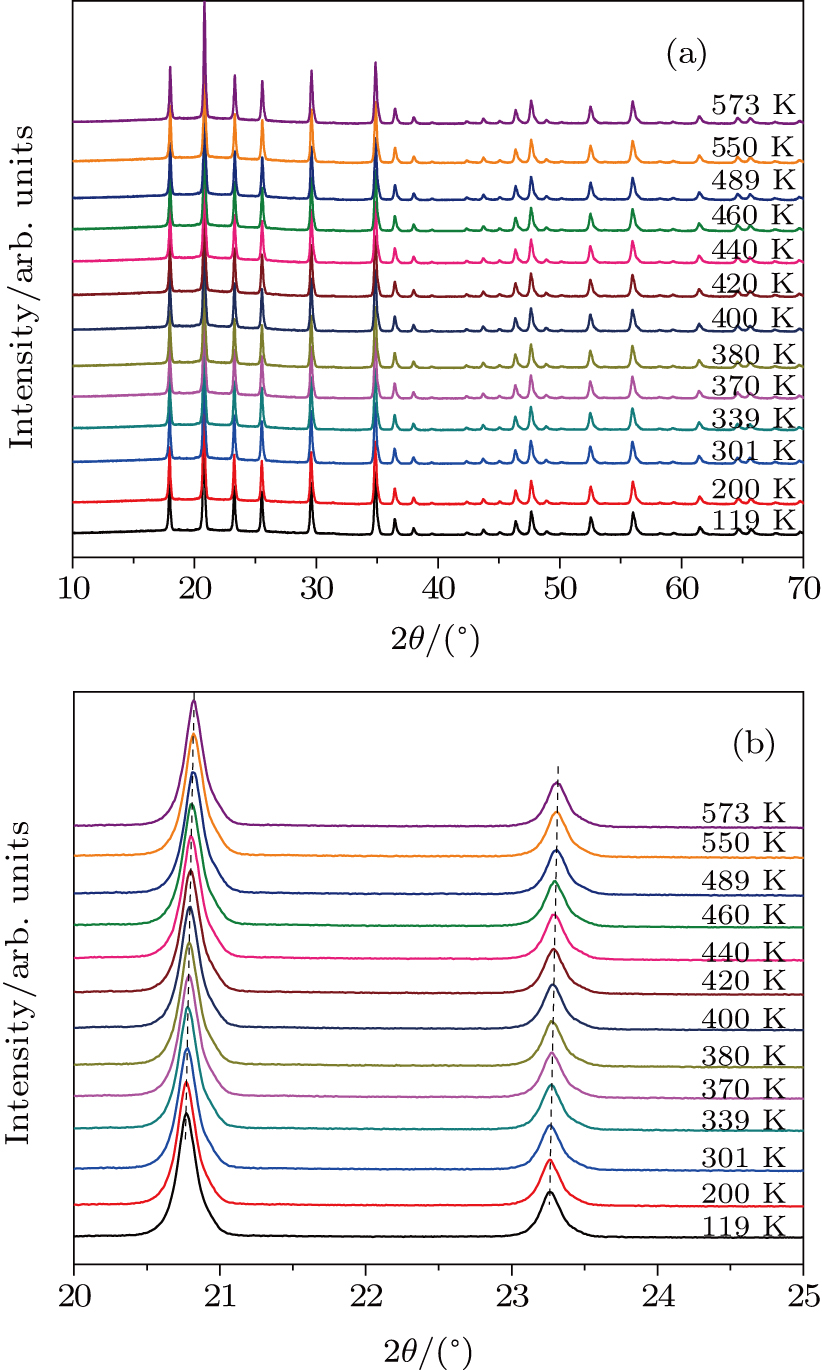 | Fig. 5. (color online) (a) XRD patterns of Zr0.5Hf0.5VPO7 at different temperatures. (b) Magnified part of patterns in panel (a). |
Figure 

The electronegativity of the cation and the M–O bond strength can be used to analyze the NTE of Zr0.5Hf0.5VPO7. The electronegativity value of P5+ (2.19) is larger than that of V5+ (1.63). The bridging oxygen connected with V5+ or P5+ gains less negative charges with the content of P5+ increasing, which reduces the effective negative charge due to the oxygen-oxygen repulsion force and makes the transition harder from superstructure to ideal crystal structure. The partial substitution of V in ZrV2O7 by P reduces the symmetry and influences the transverse thermal vibration of V–O–V. The bond angle of V (P)–O–V in ZrV2O7 is close to 180° because the transverse motion of bridge O in V–O–V and P–O–V is responsible for the negative thermal expansion of ZrV2O7.[1,17]
Dilatometric results are slightly different from the results from the variable-temperature XRD and variable-temperature Raman spectra. The microstructure effects might be responsible for the cause of such a difference. Raman spectra and XRD reveal the internal molecular structure and vibration mode of the unit cell. The Raman peak will change when the superstructure is disintegrated. The CTE from the dilatometer reflects the thermal expansion of bulk material, in which microcrack and poles are inevitable and may affect the measured results. The differences between intrinsic (XRD) and extrinsic (dilatometric) thermal expansion can be found in Zr1−xFexV2−xMoxO7,[7] ZrFeMo2VO12,[17] etc.
Figure
A solid solution of Zr0.5Hf0.5VPO7 with near-ZTE has successfully been synthesized by solid state reaction. Thermal dilatometry, XRD, and Raman spectra are used to investigate the compactness, the coefficient of thermal expansion, isotropy, and phase transition of Zr0.5Hf0.5VPO7. The results show that stable near-zero thermal expansion of the prepared material goes over a wide temperature range. The CTE for Zr0.5Hf0.5VPO7 is measured to be 0.59 × 10−6 K−1 in a temperature range of 310 K–673 K by dilatometry while it is calculated to be 0.259 × 10−6 K−1 (400 K–573 K) by XRD. The results from the dilatometer are slightly different from those from the variable-temperature XRD. The differences between the two methods are attributed to the effects of microstructure.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] |




