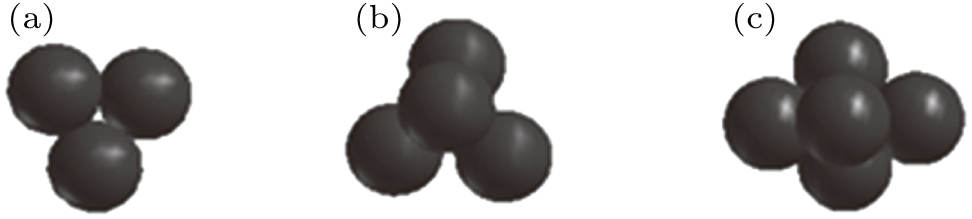Project supported by the National Natural Science Foundation of China (Grant No. 11505120) and the Project of Innovative Talents of North China University of Water Resources and Electric Power, China (Grant No. 70483).
Project supported by the National Natural Science Foundation of China (Grant No. 11505120) and the Project of Innovative Talents of North China University of Water Resources and Electric Power, China (Grant No. 70483).
† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 11505120) and the Project of Innovative Talents of North China University of Water Resources and Electric Power, China (Grant No. 70483).
The helium bubble structure and growth modes near dislocations in titanium are studied using the molecular dynamics method. A helium crystal with an HCP structure in titanium is found to have a lattice constant of 1.977 Å at 0 K. On either side of the slip plane, helium bubbles form in the (001) plane, but they are in different growth modes. On the side of the slip plane with full atomic layers, helium bubbles grow toward the slip plane and easily cross the slip plane. In the growth process, the position of the top surface of the helium bubble remains almost unchanged. On the other side of the slip plane, the helium bubble grows initially toward the dislocation core, but it is difficult to cross the slip plane, which results in growth in the opposite direction upon reaching the slip plane.
Helium atoms are usually introduced into metals through ion implantation, tritium decay, or (n, α) reactions during neutron irradiation.[1–3] It is difficult to dissolve helium atoms in metals, because they easily migrate and accumulate, forming helium bubbles. The pressures inside the bubbles are very large, which can destroy the mechanical properties of the materials.[4–8] The changes in the mechanical properties of the materials reduce the safety of nuclear installations and shorten their service life, thus it becomes important to study the behaviors of helium atoms in metals.
So far, a considerable amount of research has focused on the experimental and theoretical aspects of helium in iron and tungsten,[9–14] while only a few studies have been devoted to the behaviors of helium atoms in titanium, which is often used as a structural material in nuclear power plants. In a fast neutron reactor, titanium is a competitive candidate as a cladding material. In addition, titanium has the ability to absorb hydrogen, providing a useful tritium storage material.[15]
In previous studies, the behavior of helium in perfect titanium crystal was simulated,[16,17] and some useful results were obtained. In this study, we analyze the structures and growth modes of helium bubbles near dislocations in titanium using the molecular dynamics (MD) method. Different growth modes and HCP crystal structures of helium bubbles near the dislocations in titanium are observed, thus providing further insight into the behavior of helium in titanium.
An MD box (24a0 × 24b0 × 22c0) is used to simulate the growth of a helium bubble near a dislocation in bulk titanium, where a0, b0, and c0 are the lattice constants of titanium. Periodic boundary conditions are applied to the direction of the Boggs vector and also to the direction of the dislocation. The interaction between the Ti–Ti atoms is described by the interatomic potentials of Fabrizio et al.[18] The He–He interaction is described by the well-known Lennard–Jones potential.[19] The Ti–He potential is constructed by fitting the pair potential to ab initio data.[17] This set of potentials has been used in our recent studies, providing a good description of the bulk properties and defect properties of the Ti–He system.[16,17]
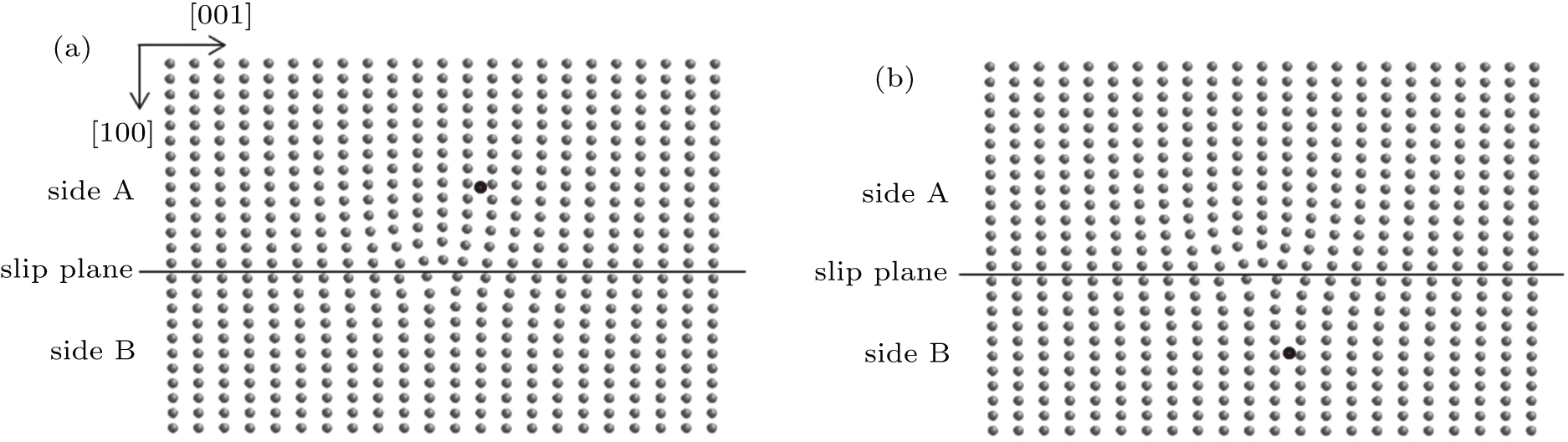
The periodic atomic arrays of dislocations are constructed based on the method proposed by Daw and Baskes, which has proved to be very successful in studying edge dislocation.[20–22] The titanium crystal is divided into two parts along the slip plane. One part is made of N atomic layers, and the other part is composed of N – 1 atomic layers, with the total width of the two parts being the same so that the interface of the two parts has a specific dislocation. The system is relaxed at 0 K to attain the lowest energy state, and a titanium substrate containing edge dislocation is formed. For convenience, the side of the substrate containing N atomic layers is named side A, and the other side containing N – 1 atomic layers is called side B.
In the following simulations, the first helium atom is placed at an interstitial site on sides A and B as shown in Figs.
On side A of the slip plane, the positions of the helium bubble are shown in Fig.
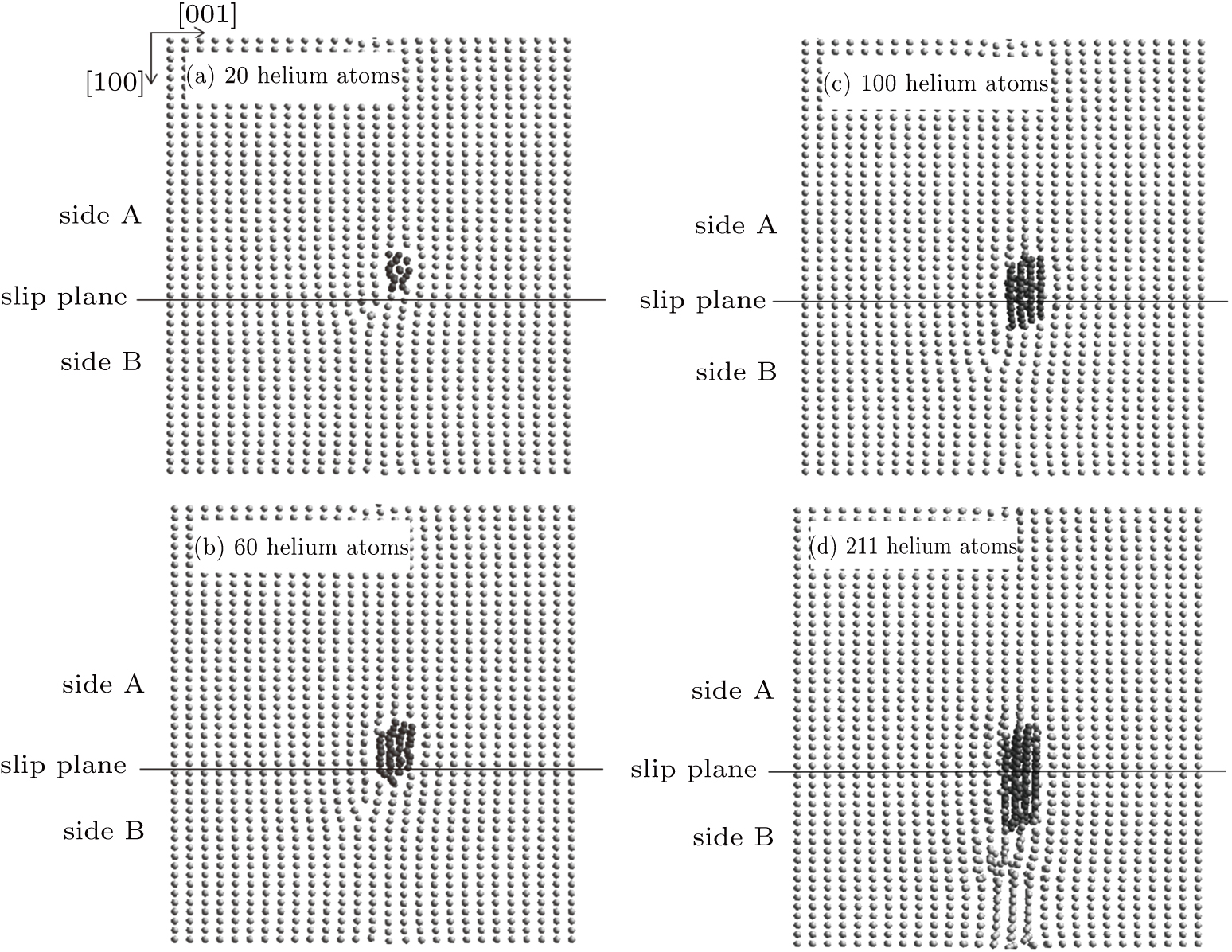 | Fig. 2. Growth of the helium bubble nucleated on side A of the slip plane in the cases of (a) 20, (b) 60, (c) 100, and (d) 211 helium atoms. |
Figure
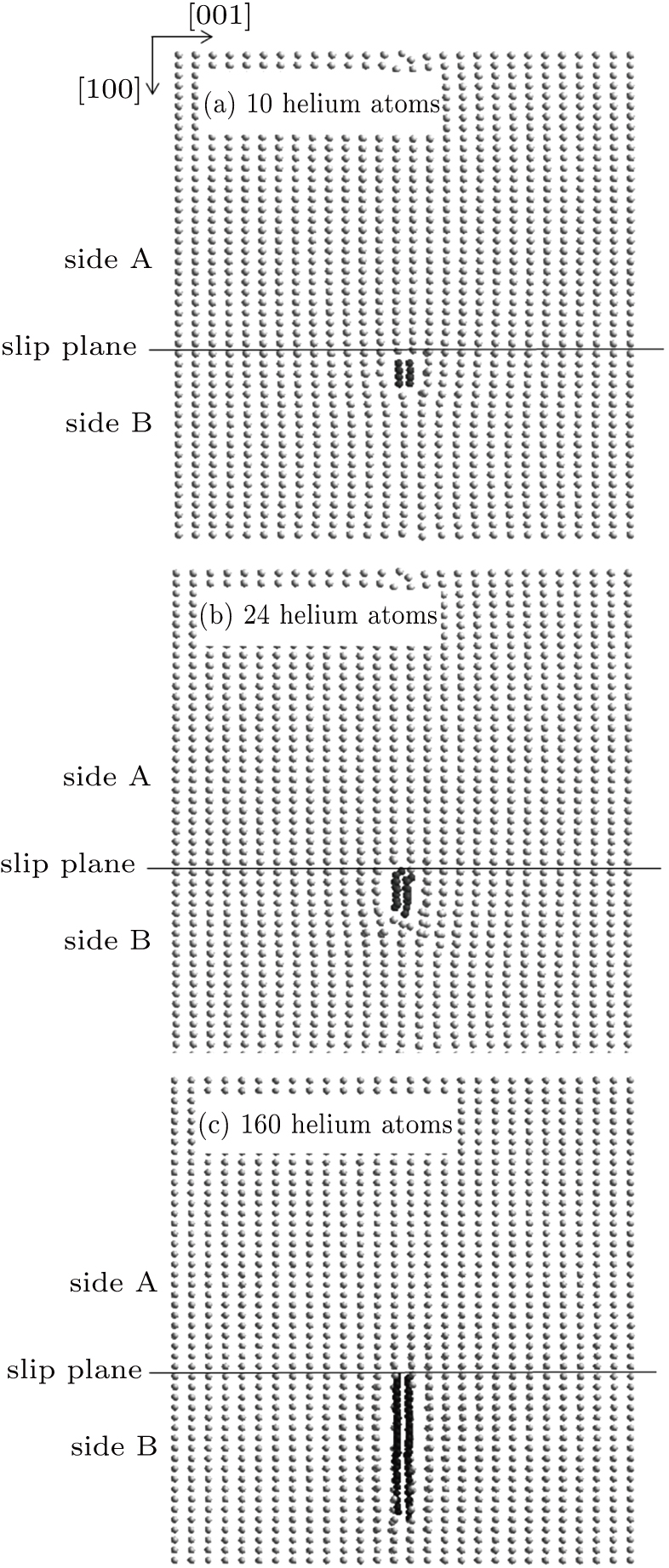 | Fig. 4. Growth of the helium bubble nucleated on side B of the slip plane, in the cases of (a) 10, (b) 24, and (c) 160 helium atoms. |
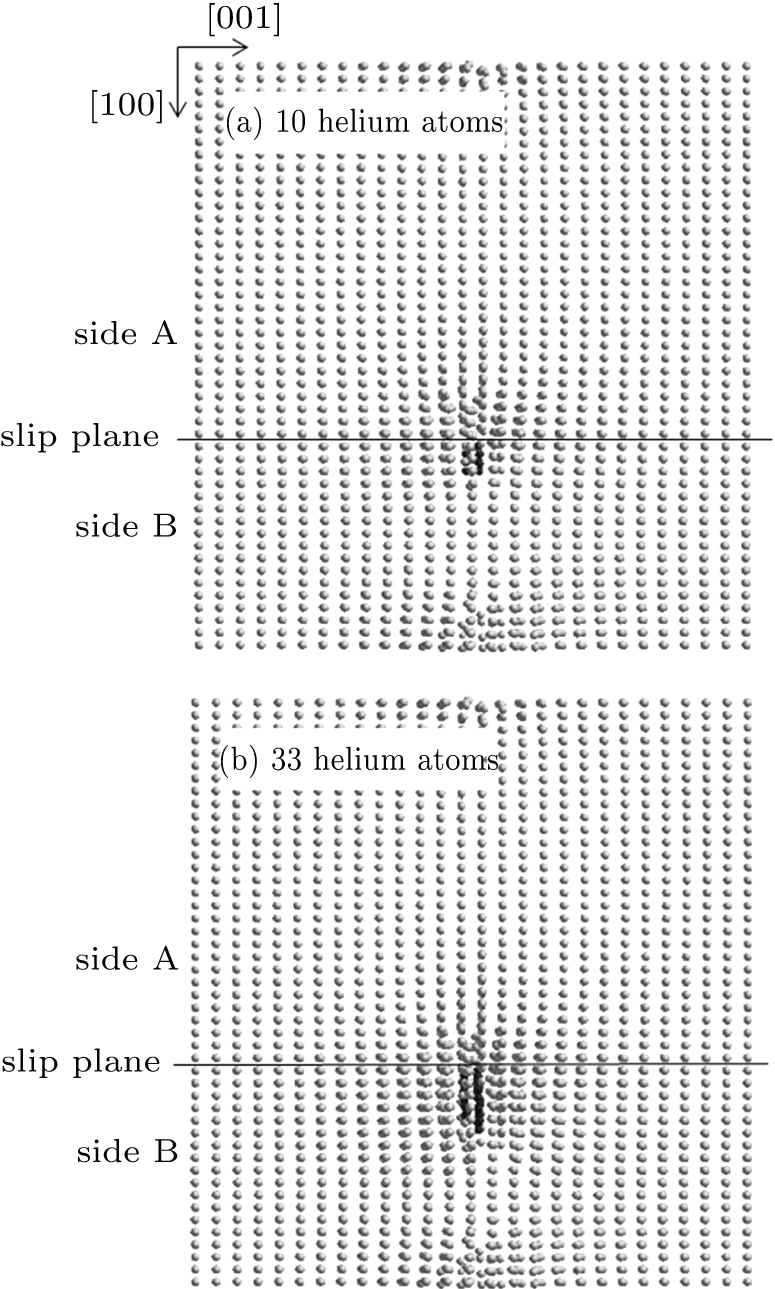 | Fig. 5. Defects induced by the helium bubble growth nucleated on side B in the cases of (a) 10 and (b) 33 helium atoms. |
Then the size of substrate is changed and relaxed at 0 K. The above simulations are repeated and the same phenomena are observed.
The helium bubbles have the same structure on both sides of the slip plane, except those with different growth modes. Figure
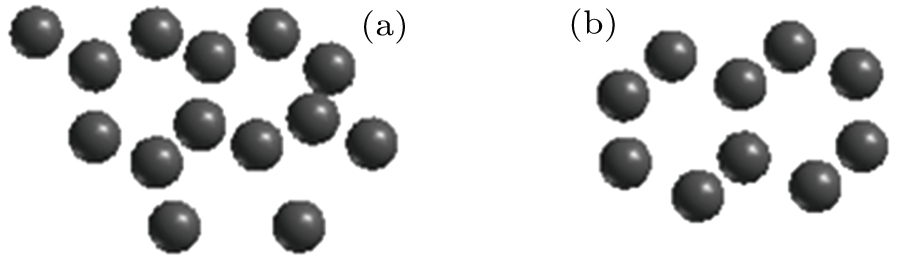 | Fig. 7. Arrangements of (a) 14 helium atoms on side A and (b)10 helium atoms on side B of the slip plane. |
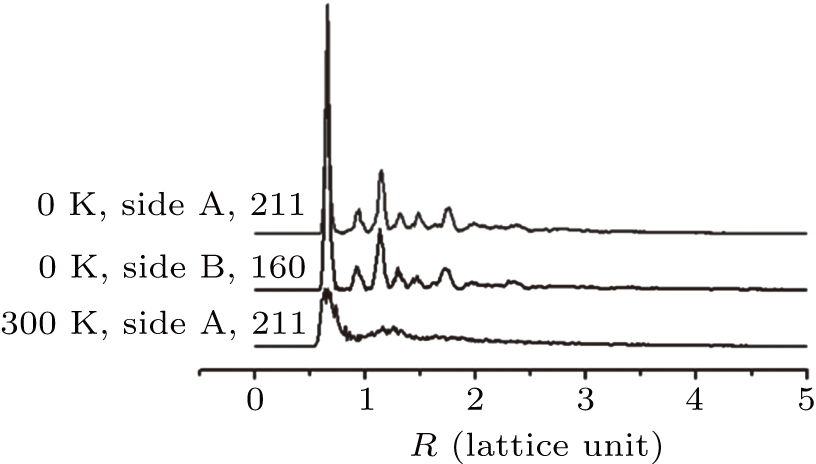
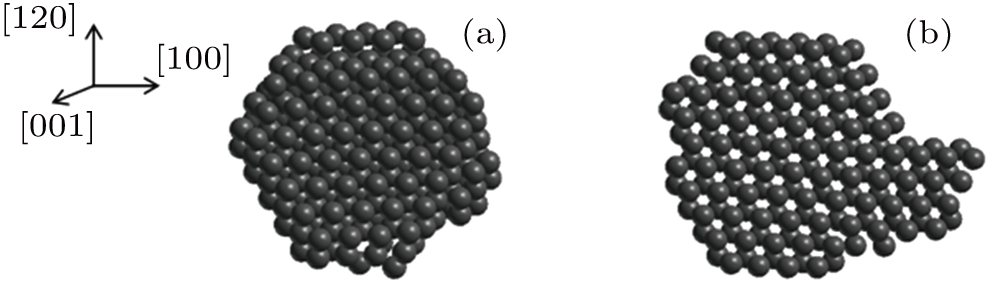 | Fig. 8. Arrangements of (a) 211 helium atoms within the bubble nucleated on side A and (b) 160 helium atoms within the bubble nucleated on side B of the slip plane. |
The pair correlation function is used to analyze the arrangement of the helium atoms as shown in Fig.
The helium crystal in titanium has been speculated previously in our study on the phase transition of high-density helium.[23] Here, the helium crystal with an HCP structure near the dislocation in titanium is clearly observed. Maji et al.’s study on solid helium bubbles in aluminum analyzed the bubbles using positron lifetime measurements.[24] The lifetime of the positron changed suddenly when the temperature decreased to 190 K, suggesting a change in the helium bubbles from a liquid to a solid state caused by the redistribution of electron density.
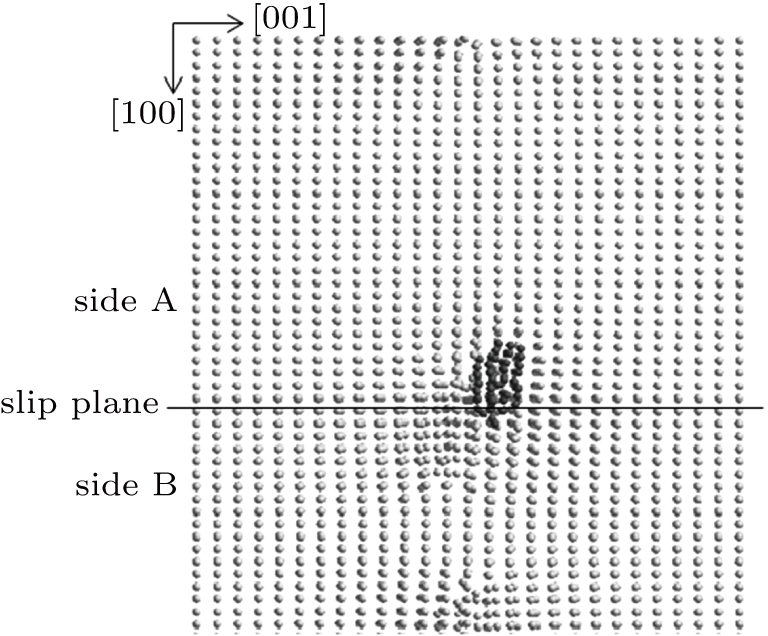
Next, the Ti–He system containing the helium bubble nucleated on side A is increased to 300 K by electron-phonon coupling. In the electron–phonon coupling model, atoms exchange energy with the electron gas in the substrate according to electron–phonon interaction dynamics.[14,25] Due to the high thermal conductivity of the electron gas in metal, the energy transferred to the electron gas is quickly dispersed, and the electron gas is considered to maintain a constant temperature. At 300 K, the helium bubble continuously expands along the [100] direction in the (001) plane as shown in Fig.
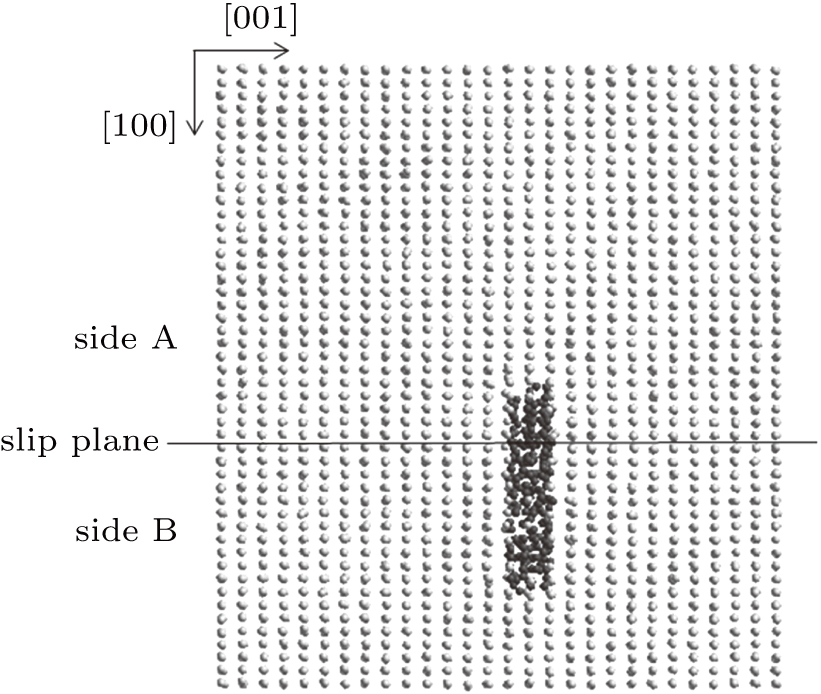 | Fig. 10. Expansion of the helium bubble nucleated on side A of the slip plane along [100] direction at 300 K. |
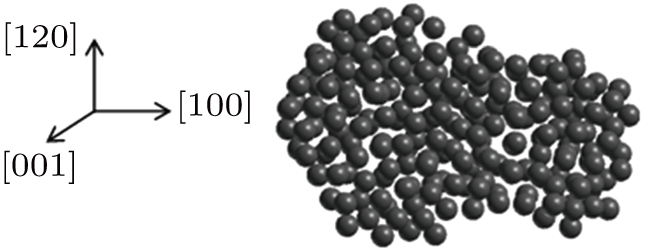 | Fig. 11. Arrangements of 211 helium atoms within the bubble nucleated on side A of the slip plane at 300 K. |
In the following simulations, the temperature of the substrate is set to be 300 K. Helium atoms are added near dislocations to make a comparison with the simulation results of 0 K. Similar phenomena are observed as follows. On the side of the slip plane with full atomic layers, a helium bubble grows toward the slip plane and easily crosses the slip plane. On the other side of the slip plane, the helium bubble has difficulty in crossing the slip plane. It grows initially toward the dislocation core and then grows in the opposite direction. However, the arrangements of the helium atoms are disordered, i.e., helium bubbles do not exhibit a crystal structure at 300 K.
Helium bubble structure and growth near dislocations in titanium have been analyzed using the molecular dynamics method. A helium bubble is shown to form in the (001) plane on either side of the slip plane, but with different growth modes. On the side of the slip plane with full atomic layers, the helium bubble grows toward the slip plane, and then crosses the slip plane easily. In the growth process, the top surface position of the helium bubble remains almost unchanged. On the other side of the slip plane, the helium bubble grows toward the dislocation core initially, but experiences difficulty in crossing the slip plane, and grows toward the opposite direction after reaching the slip plane. Generally, the helium bubble tends to grow toward the dislocation core, or alternately towards the side of the slip plane that lacks an atomic layer. It is interesting to note that the helium bubble demonstrates an HCP crystal structure with a lattice constant of about 1.977 Å at 0 K near the dislocation in titanium, but it is liquid at 300 K.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] |