† Corresponding author. E-mail:
Ab initio molecular dynamics simulations were performed to investigate the effect of similar elements on the short- to medium-range atomic packing features in Ce70Al30and La70Al30glass-forming alloys. 4f electrons of Ce element in Ce70Al30 alloy were properly treated in electronic calculations. The local atomic structures in both alloys are qualitatively similar. However, the local environments of Al atoms in Ce70Al30 alloy show fluctuation with temperature in the cooling process, which could result from 4f electrons of Ce elements. Surprisingly, the medium-range atomic packing features of Al atoms in both MGs are quite different, although Ce and La elements are similar. These findings are useful for understanding the enhanced glass-forming ability by similar element substitution in RE-based MGs from a medium-range structure perspective.
Recently, rare earth (RE)-based metallic glasses (MGs) have attracted strong interest, because of their valuable scientific problems and technological applications, such as superplasticity, thermoplasticity, high glass-forming ability, and ductility.[1–6] It has been found that Ce-based bulk metallic glasses (BMGs) have exceptionally low glass transition temperature, similar to or lower than that of many polymers, which makes Ce-based BMGs show excellent thermoplastic molding and imprinting ability.[2] This demonstrates that Ce-based BMGs exhibit a superior glass-forming ability (GFA) and high resistance to crystallization over a wide temperature range.[2] In addition, the polymorphic transitions and anomalous compression behavior induced by pressure were also observed in Ce-based BMGs, indicative of the importance of 4f electrons in the Ce element.[7–9] On the other hand, the GFA of La-based alloys can be improved by substituting other lanthanide elements such as Ce.[2] The similar element substitution enhanced GFA in RE-based MGs cannot be understood by conventional criteria such as large atomic size mismatch.[10] This implies that the lanthanide elements with similar atomic sizes and various valence electronic structure may have a significant impact on improving GFA in RE-based glass-forming alloys. Ab initio molecular dynamics simulations were performed, which showed that more icosahedral-like clusters and less free volume could be the structural origin of the higher GFA induced by partial substitution of Ce for La in LaCoAl bulk MG.[11] So far, however, the atomic structure features in Ce- and La-based MGs were not fully characterized. How the 4f electrons in the Ce element affect atomic structures has not been addressed yet. It is not clear, either, whether the medium-range atomic packing is also similar in the similar element substitution of Ce and La in RE-based MGs, and whether there is any correlation between medium-range atomic packing and GFA.
In this work, we performed ab initio molecular dynamics (MD) simulations to systematically investigate the short- and medium-range structures in Ce70Al30 metallic liquids and glasses. Here binary alloy with Ce rich was chosen for simplicity in both simulations and structural analysis. In our simulations, 4f electrons in the Ce element at ambient pressures were modeled by adopting LDA+U calculations. Meanwhile, we also performed ab initio MD simulations for La70Al30 metallic liquids and glasses in which La has similar valence and ionic radii but without 4f electron. Therefore, the structure features in both Ce70Al30 and La70Al30 glass-forming alloys can be fully characterized for understanding the effect of similar elements on short- to medium-range atomic structures and thereby GFA.
Ab initio MD simulations in the canonical NVT (constant atom number, volume, and temperature) ensemble were performed using the Vienna ab initio simulation package (VASP).[12] Projected augmented plane waves (PAW) with the PBE exchange-correlation potential[13] were adopted to describe electron–ion interaction. The simulations were performed only at the Γ point. Cubic supercells containing 200 atoms for Ce70Al30 and La70Al30 systems were initially constructed with periodic boundary conditions applied in three directions. A stepwise quenching process with a cooling rate of 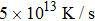
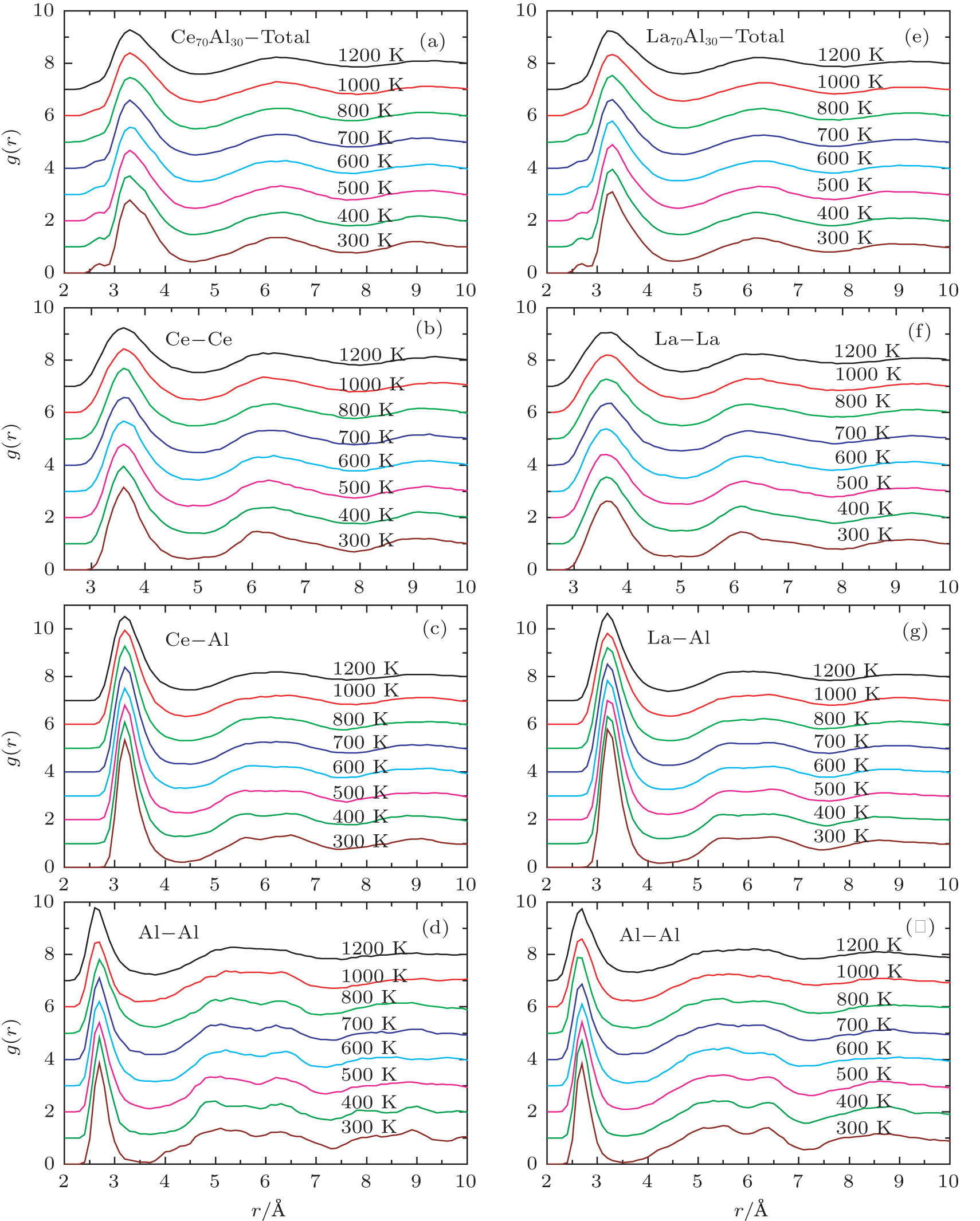
Figure
To investigate the local atomic structures beyond the PCFs in both Ce70Al30 and La70Al30 systems, the Voronoi tessellation method was employed, which divides space into close-packed polyhedra around atoms by construction of bisecting planes along the lines joining the central atom and all its neighbors.[16–18] Each Voronoi polyhedron can be identified with the Voronoi index 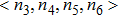
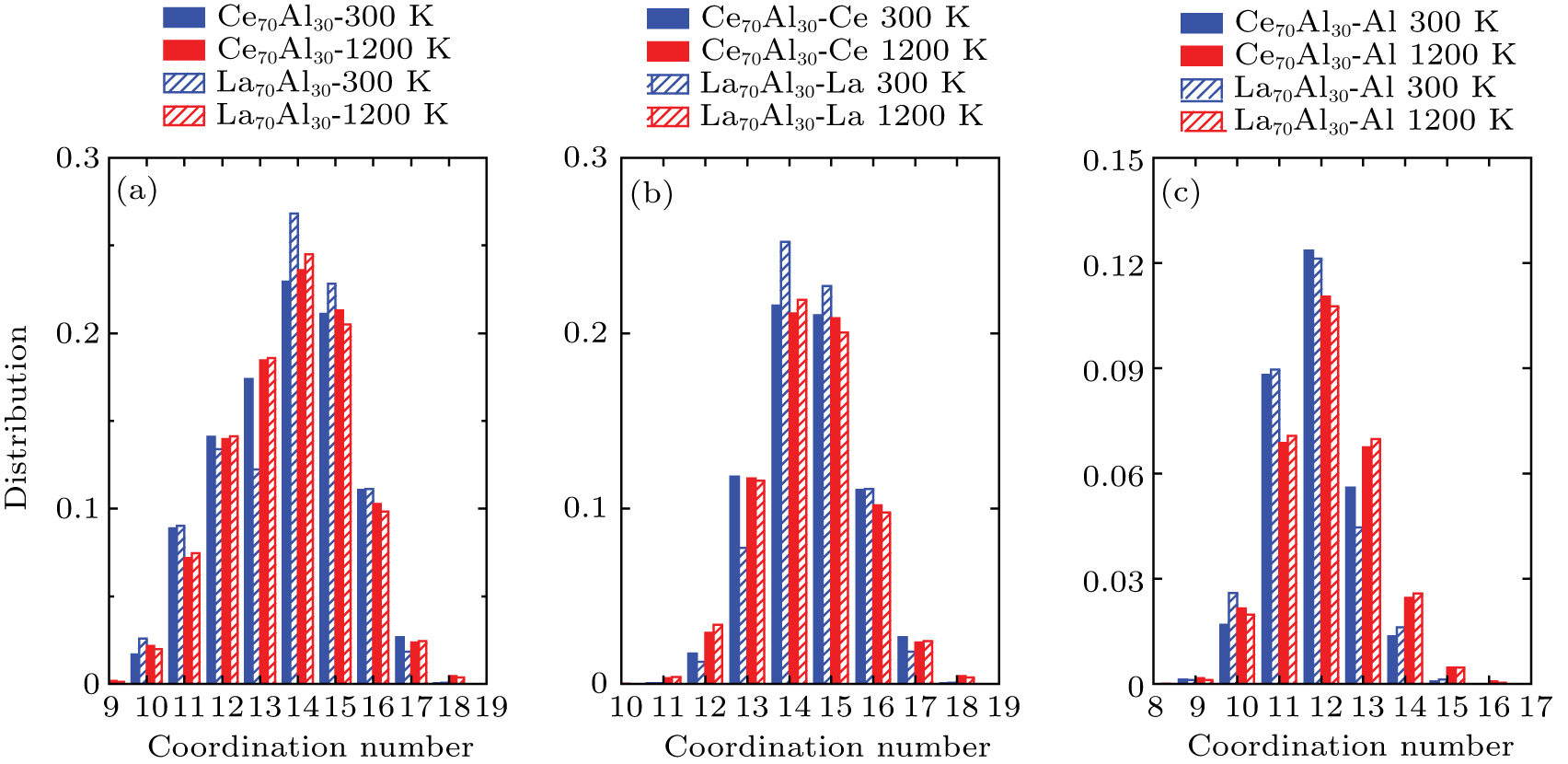 | Fig. 2. (color online) Total (a) and partial ((b), (c)) coordination numbers in Ce70Al30 and La70Al30 glass-forming alloys at 1200 K and 300 K, respectively. |
To get more detailed structural information for the local atomic arrangement, we analyzed the bond–angle distribution, the so-called three-body correlation,[18] which characterizes the angles between two bonds connecting the central atom to any two neighboring atoms. Figure
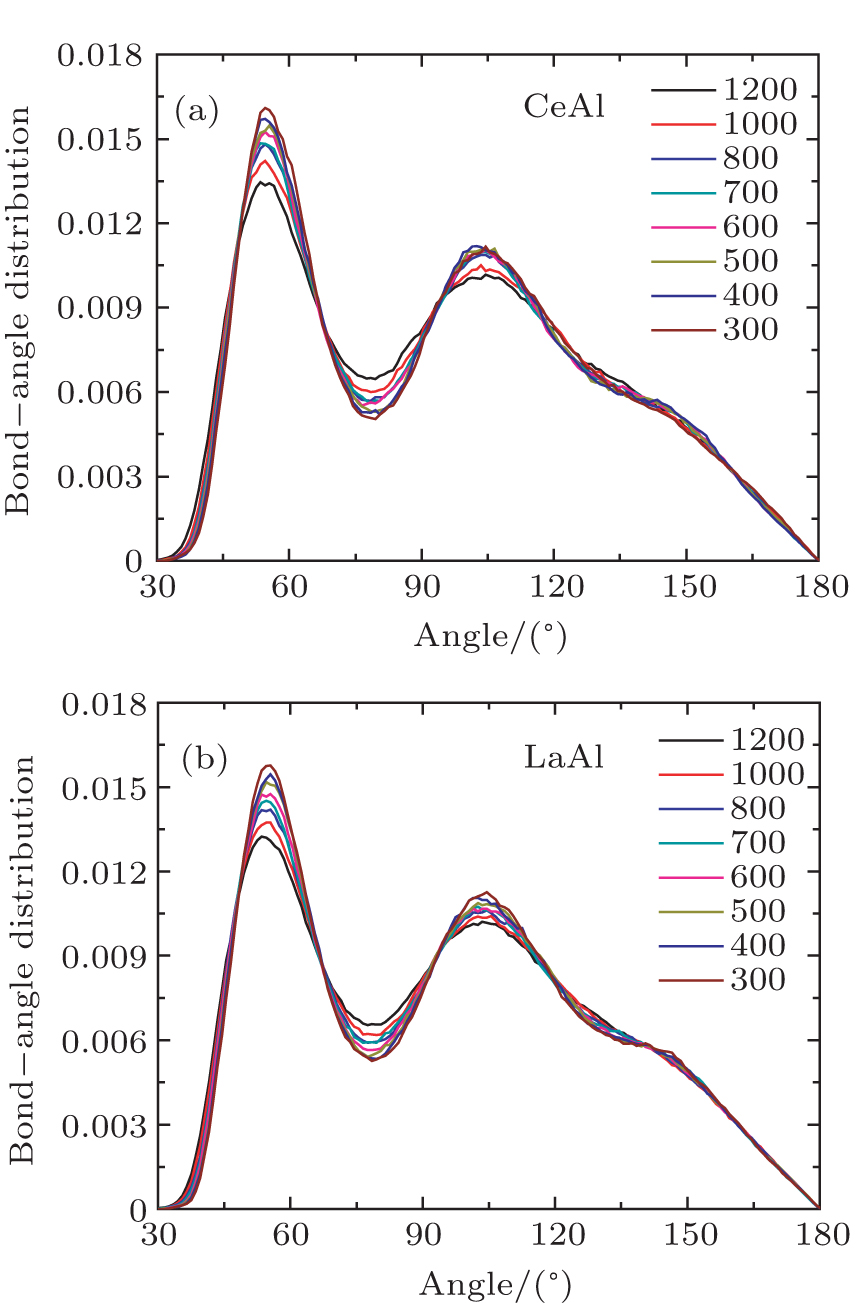 | Fig. 3. (color online) Total bond-angle distributions in Ce70Al30 (a) and La70Al30 (b) glass-forming alloys at various temperatures, respectively. |
We also investigate the partial bond–angle distributions at various temperatures in Ce70Al30 and La70Al30 glass-forming alloys, respectively. As shown in Fig.
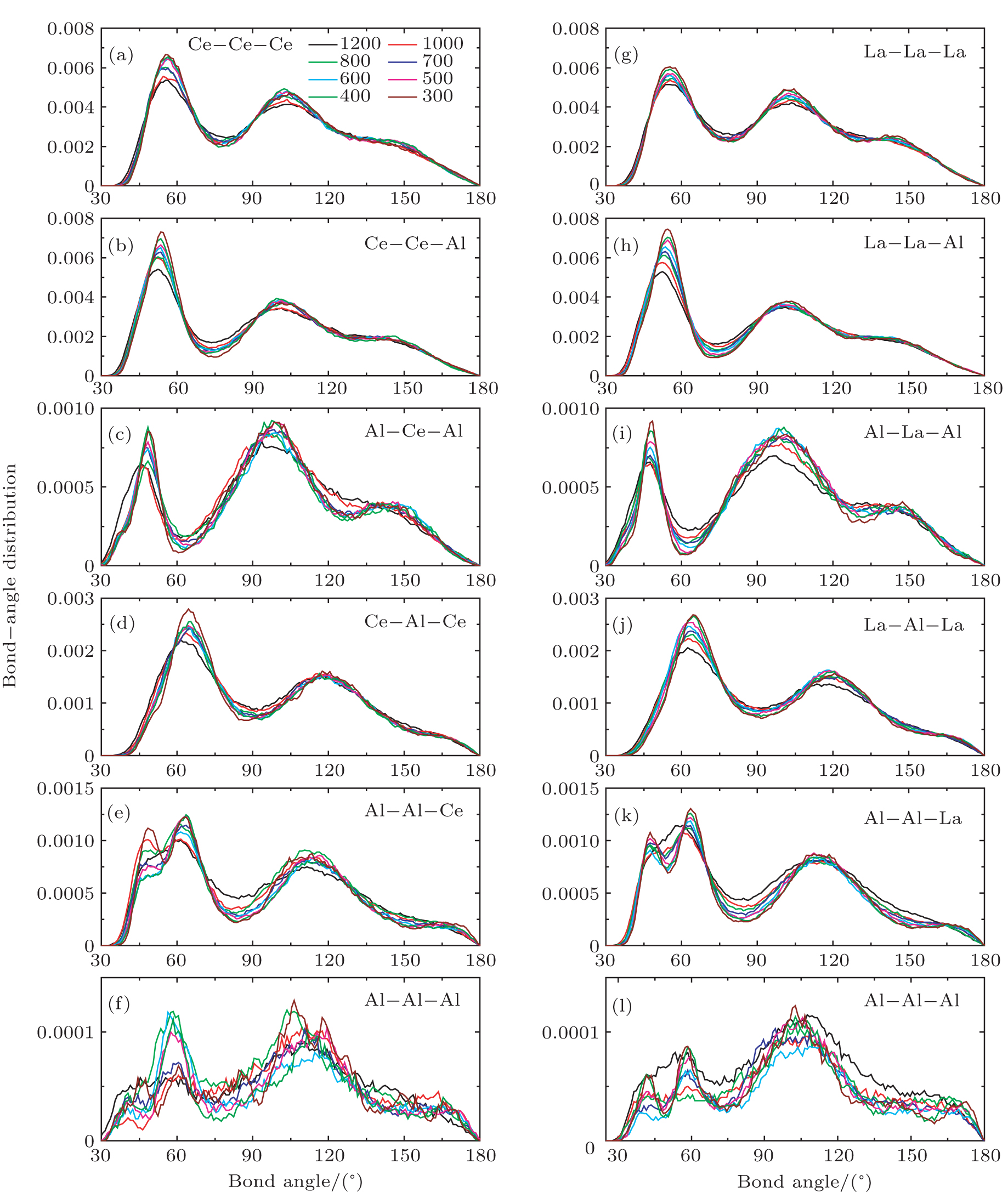 | Fig. 4. (color online) Partial bond–angle distributions in Ce70Al30 ((a)–(f)) and La70Al30 ((g)–(l)) glass-forming alloys at various temperatures, respectively. |
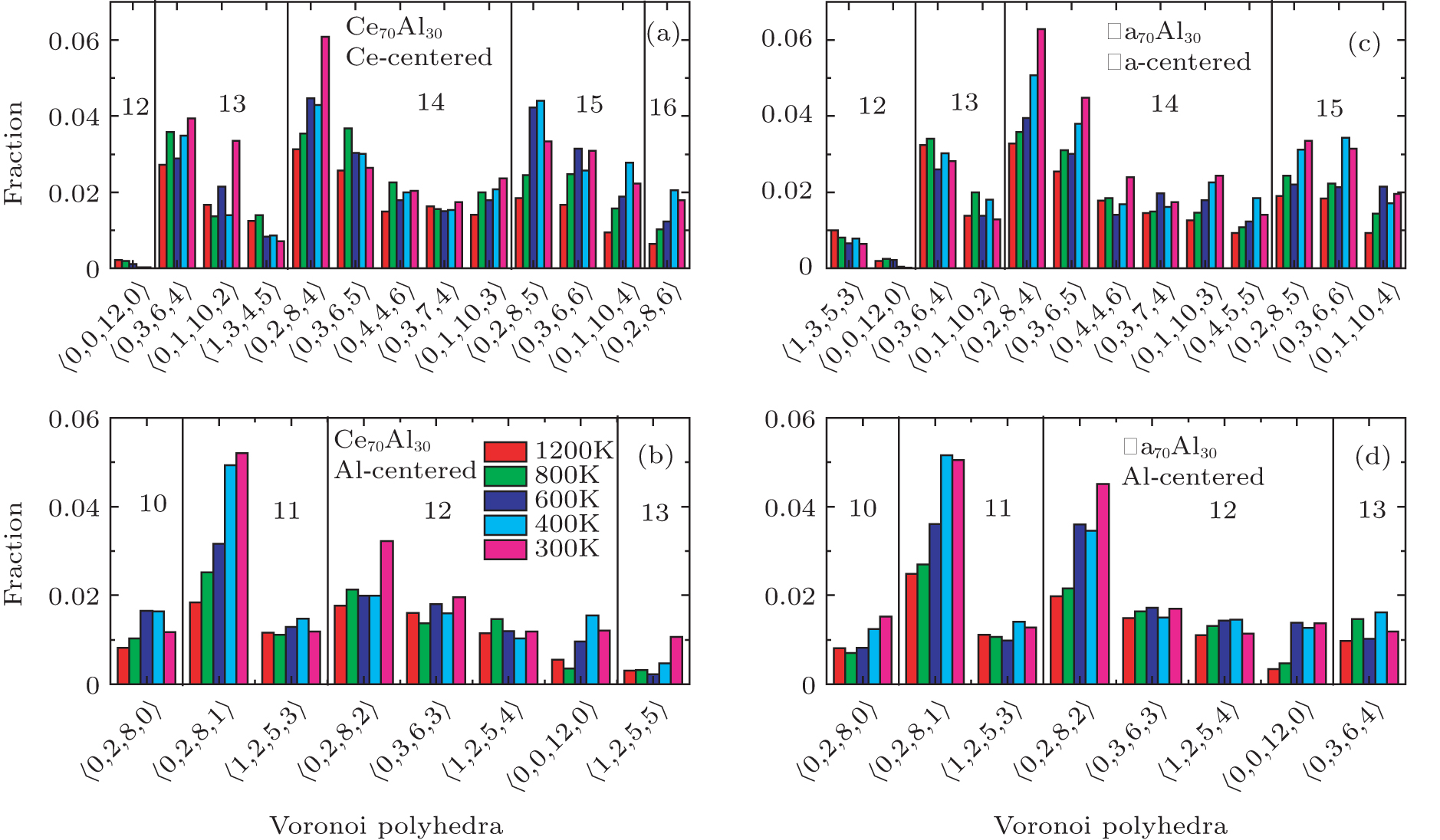
According to the above analysis, the local atomic arrangements related to Al atoms change significantly as temperature decreases. In addition, the local structures around Al atoms are also different in Ce70Al30 and La70Al30 glass-forming alloys. To get a deeper insight into the local atomic structure features, we analyzed the distributions of the major populated atomic clusters in Ce70Al30 and La70Al30 glass-forming alloys at various temperatures. It is found that Ce (La) and Al atoms in Ce70Al30(La70Al30) alloy rarely share the same types of atomic clusters, so that only the major populated Ce (La)-centered and Al-centered atomic clusters were presented, respectively, as shown in Fig. 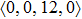
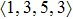
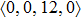
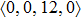
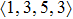
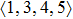
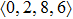
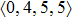
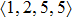
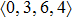
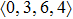

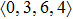

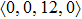
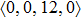
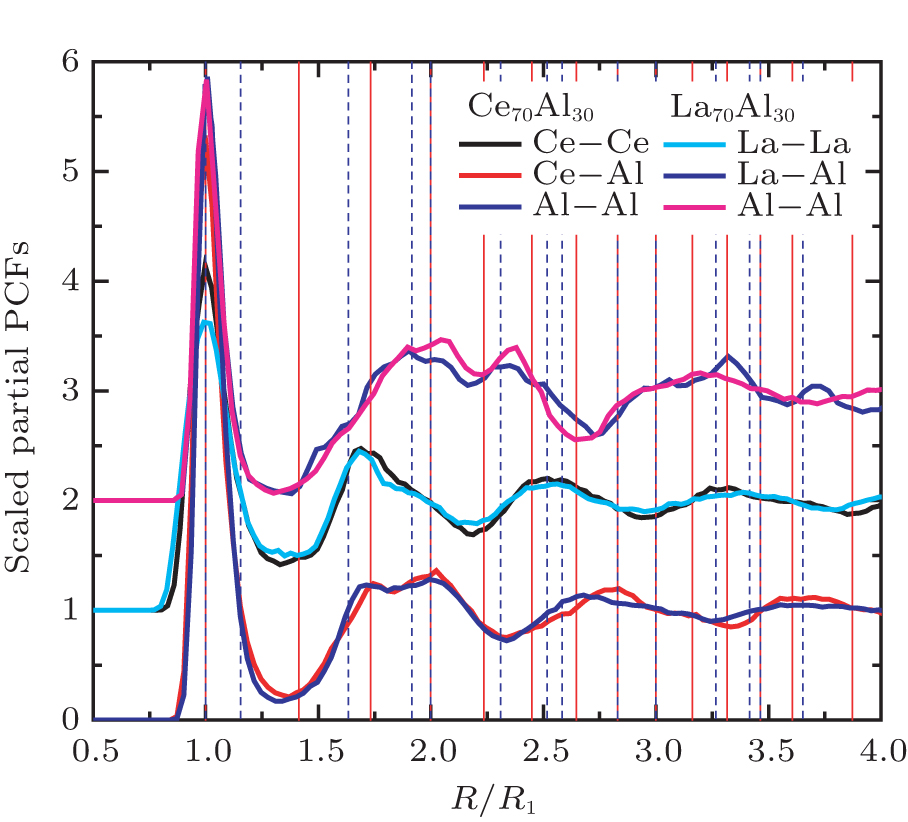
As mentioned above, in partial PCFs of the Al–Al pair in Ce70Al30 and La70Al30 alloys, the peak positions show a slight difference, indicative of different atomic packing in medium-range structures in two MGs. To further characterize the medium-range atomic packing features in two MGs, we analyzed the partial PCFs with the distance scaled by the first peak position R1. Previous studies show that the oscillation in the PCFs is an indication that a certain order does exist in amorphous solids, and the values of 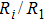
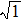
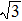
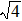
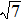

Figure 7 shows the scaled partial PCFs for Ce–Ce, Ce–Al, and Al–Al in Ce70Al30 MG and La–La, La–Al, and Al–Al in La70Al30 MG, respectively. It can be seen that the scaled PCFs of Ce–Ce and La–La are quite similar. The peak positions are almost the same. However, the scaled PCFs of Ce–Al and La–Al, Al–Al (Ce70Al30) and Al–Al (La70Al30) are significantly different. For Ce–Al and La–Al pairs, the second and third peak positions are almost the same. However, the fourth peak positions are quite different, located at 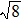
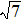
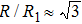
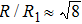
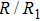
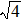

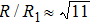
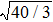
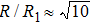

Recently, similar element substitution enhanced GFA was observed by substitution of Ce for La in (La0.7Ce0.3)65Co25Al10 bulk MGs.[3] It was found that more icosahedral-like clusters may be one of the structural origins of the higher GFA enhanced by similar element substitution.[9] This is consistent with the above local structure analysis in terms of the Voronoi tessellation method. The substitution of Ce for La will introduce more 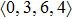
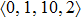
In summary, we performed systematic ab initio MD simulations for Ce70Al30 and La70Al30 glass-forming alloys. The short-range local atomic structures exhibit a little difference, especially for the local structures of Al atoms, which could result from the 4f electrons in the Ce element. More importantly, the medium-range atomic packing between Al atoms in both MGs is significantly different, although Ce and La are similar elements, which may be the key underlying structural basis for the enhanced GFA by similar element substitution in RE-based MGs.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] |