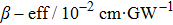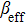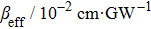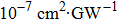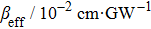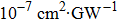† Corresponding author. E-mail:
Two dimethylamino-carbaldehyde derivatives with different π-bridge lengths were prepared, and their transient optical properties and photophysical mechanisms were investigated by transient absorption spectroscopy and Z-scan measurements. Owing to the difference in molecular structures, the two compounds exhibit different populations of locally excited states and, therefore, they also produce different transient absorption spectra. After photoexcitation, both molecular materials exhibit a wide excited state absorption band from 450 nm to 1000 nm. Meanwhile, the excited state lifetimes are dramatically different, 2 ns and 100 ps, for the two molecules. A figure of merit greater than 2 at the wavelength of 1000 nm is obtained. The results show that modulating the population of the locally excited states in this type of molecule can be a promising approach for obtaining optical switching and solar cell materials.
Nonlinear optical (NLO) materials have many potential uses in various photoelectronic devices and photonic applications.[1–3] Much effort has been devoted to the development of NLO devices with a large nonlinear response.[4–6] Accompanied by the advance of materials chemistry, researchers are no longer satisfied with optical nonlinearity at specific wavelengths, and broadband response becomes a desired property in developing nonlinear optical materials. Donor-π-acceptor (D-π-A) molecular materials have recently attracted considerable academic and technological research attention for their applications in nonlinear optics.[7–9] Their NLO properties can be adjusted by the appropriate manipulation of their chemical structures such as the different connectors linking the electron-donor/electron-acceptor (D/A) building blocks, the distance between the D/A units, and the symmetry of the molecular structure.[10–12] Many types of functional groups with strong electron withdrawing or electron donating properties, linked to a connector, have been selected by researchers for study. Although this is a good approach for synthesizing materials with large NLO properties and broadband response, the results have not always been satisfactory.
Transient absorption spectroscopy is widely used to follow photophysical dynamics in molecular systems.[13–15] In the field of nonlinear optics, transient absorption spectra are widely used in studying the NLO properties of materials.[16–18] Modulating the transient absorption spectra of materials can lead to broadband excited state absorption and enhanced NLO properties. For many fluorescent materials with D-π-A structure, two excited states, namely, locally excited (LE) states and intramolecular charge-transfer (ICT) states, are accessible upon photoexcitation. The LE states are proposed to be emissive, but the ICT states are usually not.[19–21] Suppressing transition from the LE states to the ICT states results in increased population of the LE states, and thus, the fluorescence emission intensity will be enhanced.[22,23] However, the transient absorption spectra of these materials can also exhibit saturated absorption (SA) due to the influence of the stimulated emission. Our interest is in modulating the transient absorption spectra of D-π-A molecular materials by changing the population of the LE states. This approach has been used to obtain improved fluorescent materials, but there are few reports on modulating the transient absorption spectra of materials in this way.
Herein, we report investigations into two dimethylamino-carbaldehyde derivatives 4-{2-[4-(N,N-dimethylamino)phenyl]ethynyl} benzenecarbaldehyde (DPC) and 4’-(dimethylamino)-[1,1’-biphenyl]-4-carbaldehyde (DBC). Owing to the difference of molecular structures, upon photoexcitation, tilted molecules exhibit different population of locally excited (LE) states. In order to study the influence of tuning the population of the LE states on transient absorption spectra and NLO properties, we systematically investigate the broadband nonlinear optical properties of DPC and DBC by performing broadband transient absorption spectroscopy and Z-scan measurements with 190 fs laser pulses. Our results demonstrate that tuning the population of the LE states of this type of material can enhance its NLO properties, and we conclude that such methods may be considered as a promising candidate for modulating their transient absorption spectra.
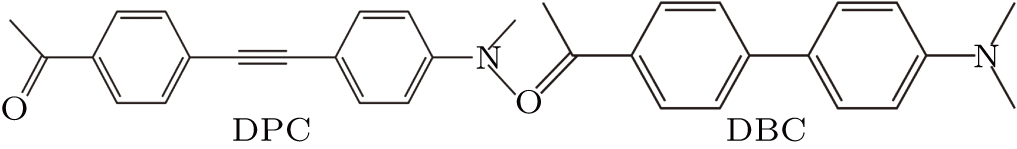
The first compound, DPC, is a diphenylacetylene derivative with a dimethylamino group and a carbaldehyde group, and the second, DBC, is a biphenyl derivative with the same groups (Fig.
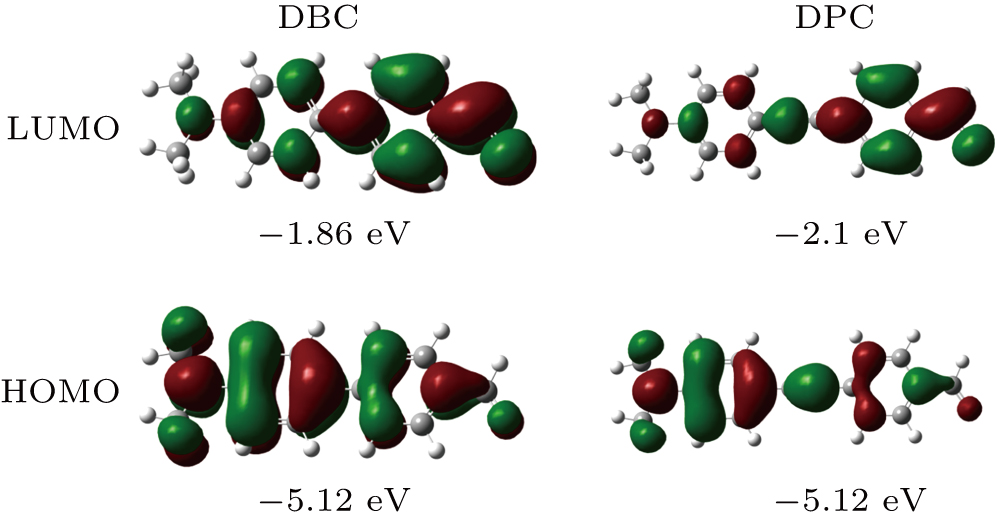
In order to study the relationship between the structures and the properties of the compounds, density functional theory (DFT) calculations were performed using Gaussian 09 software.[28] A B3LYP/6-31G(d,p)//CPCM (DMSO) model was used to optimize the structures of the molecular systems. The electron-cloud distribution of various frontier molecular orbitals was estimated from these calculations, and the results are summarized in Fig.
Broadband transient absorption measurements were performed using femtosecond pulses generated by a regeneratively amplified Yb: KGW fiber laser system (Light Conversion, PHAROSSP) that produces pulses with energies of 1 mJ, centered at 1030 nm, with a repetition rate of 6 kHz, and a full width at half maximum (FWHM) of 190 fs. The main portion of the output was delivered to pump an optical parametric amplifier (OPA, ORPHEUS, Light Conversion). The OPA output was tuned to a wavelength of 365 nm, and this was used as the pump beam for the transient absorption excitation. Before interacting with the sample, this beam was modulated at 137 Hz using a chopper, and attenuated to the desired intensity using a gradual neutral density optical filter for minimizing undesired cross-phase modulation effects. Probe pulses for spectroscopic characterization were produced by passing a small portion of the laser output at 1030 nm wavelength through a computer-controlled optical delay stage (with a maximum delay line of 1830 ps) and focusing it onto a 2-mm-thick Ti:sapphire plate to produce a white-light continuum spanning the 455–1000 nm window. The polarization of the pump and the probe pulses at the sample location was at the magic angle to avoid the anisotropic effect. The angle between the pump and the probe beams was 5° and the beam waists of the pump and probe beams were 1 mm and 
The NLO properties of the two asymmetric dimethylamino-carbaldehyde derivatives were measured using the Z-scan technique. The light source for the femtosecond Z-scan was an OPA (ORPHEUS, 190 fs, 20 Hz, Light Conversion) with a tunable output wavelength ranging from 760 nm to 1000 nm. The experimental apparatus has been described in detail elsewhere.[31] The sample solution was injected into 2-mm-thick cuvettes. The cell was placed on a translation stage manipulated by a computer that moved along the Z-axis of the focal point of a lens with a 400-mm focal length. The laser pulses, after modulation by an attenuator, were separated into two beams by using a beamsplitter; one beam was focused on the sample through a lens with a 400-mm focal length and the other beam served as the reference beam. The two beams were simultaneously measured using two energy detectors (Rjp-765 Energy Probe, Laser Probe, Inc.) linked to an energy meter (Rj-7620 Energy Ratiometer, Laser Probe, Inc.). A computer was used to collect the data from the energy meter through a general-purpose interface bus (GPIB). All of the samples were measured using dimethylsulfoxide (DMSO) as the solvent.
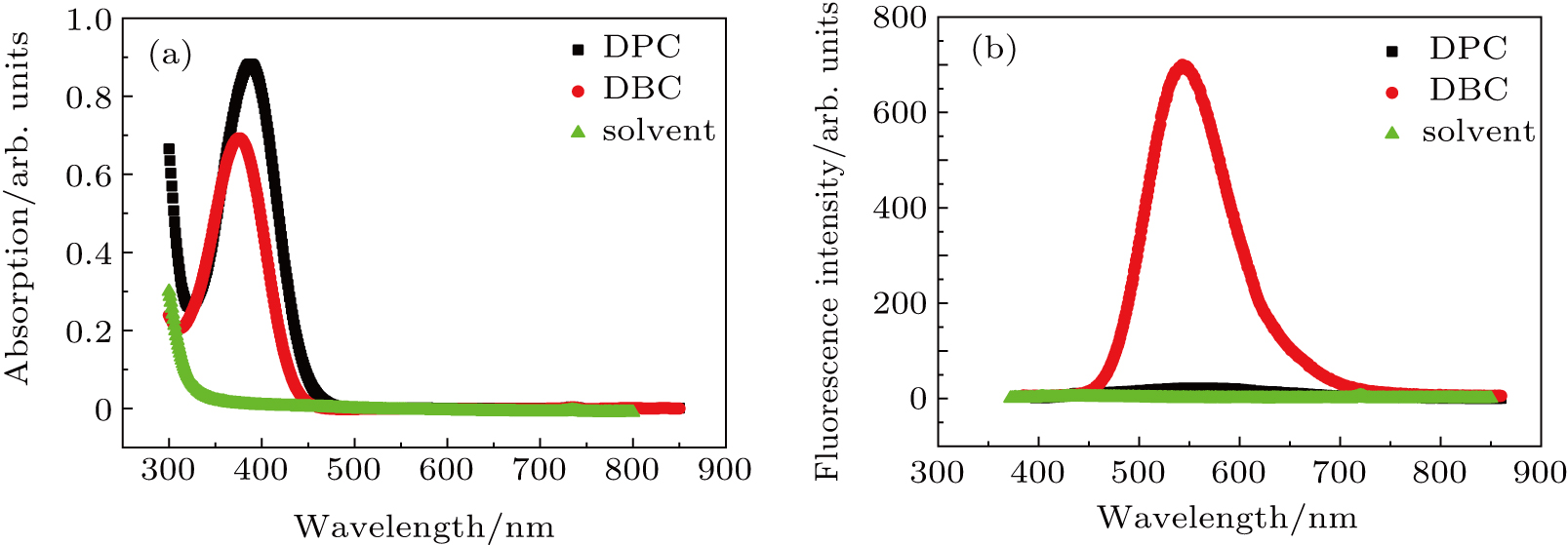
The ultraviolet–visible (UV–Vis) absorption spectra of the compounds DBC and DPC were recorded in a diluted DMSO solution (1×10−5 mol/L) at room temperature, and the results are presented in Fig.
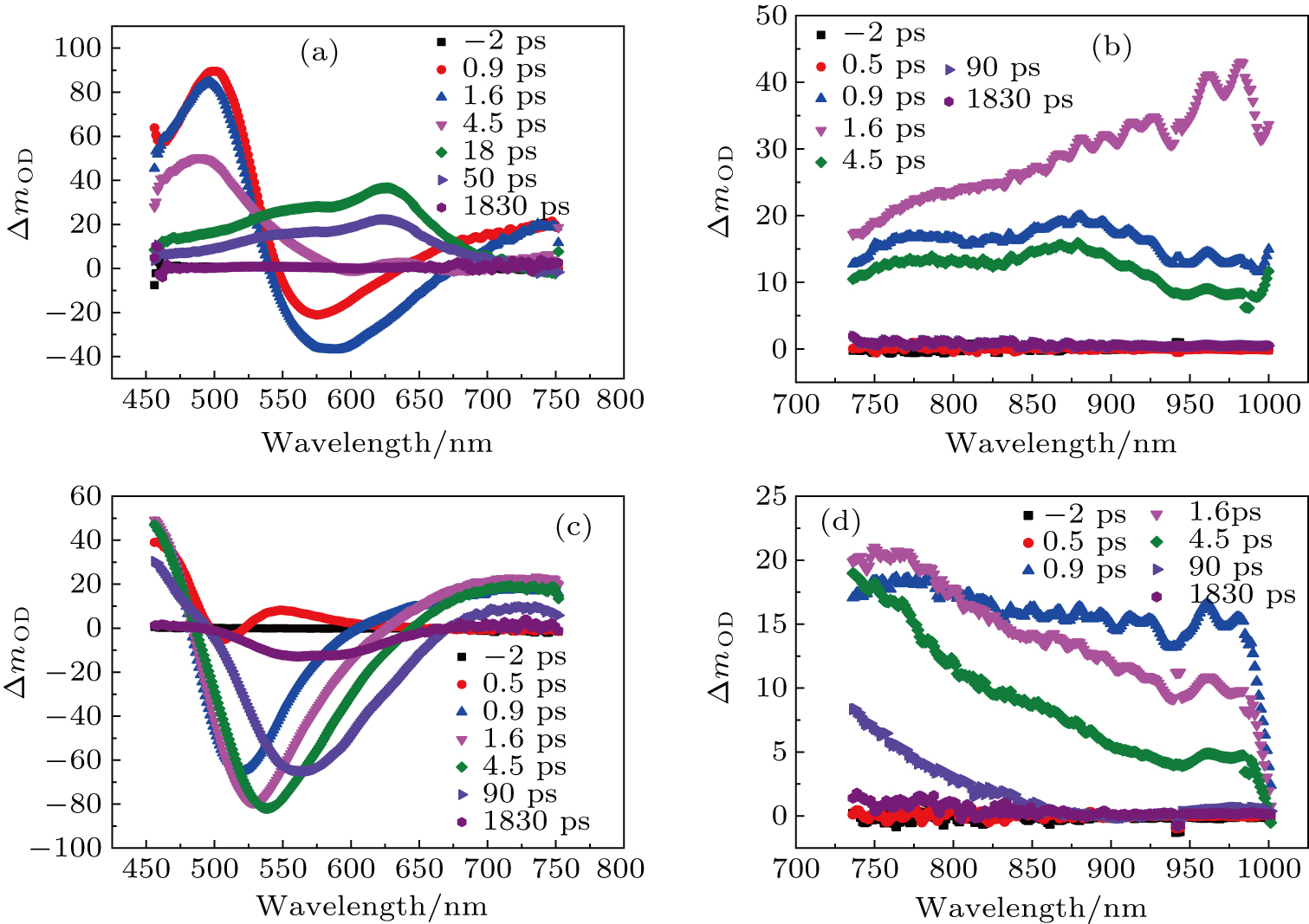
The femtosecond transient absorption spectra were recorded separately in the visible light region and the near-infrared band with 190 fs laser pulses as the pump at the wavelength of 365 nm; the spectra are plotted in Fig.
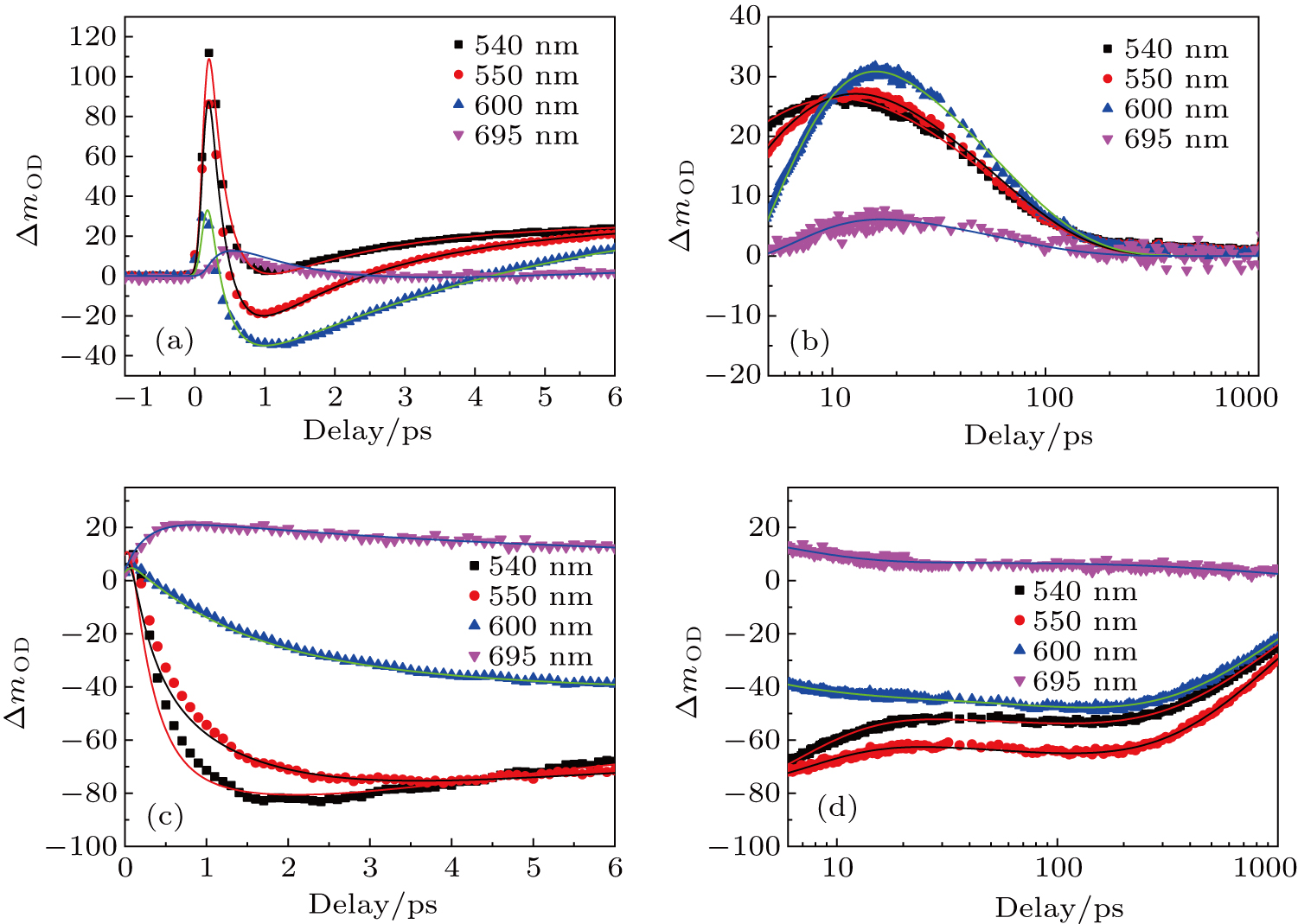
In order to study the dynamic process of the two compounds, the time-resolved transient absorptions of DBC and DPC are displayed in Figs.
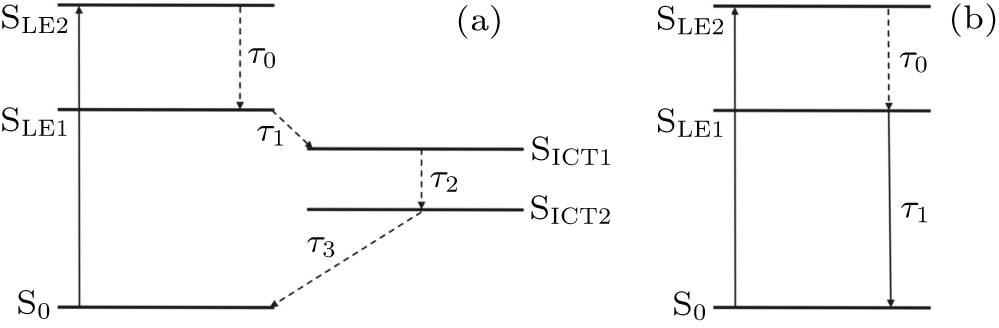 | Fig. 6. Simplified energy-level model of (a) DPC and (b) DBC. The dotted lines represent non-radiative transitions, and the solid line represent radiative transitions. |
| Table 1.
Excited state lifetime of DPC and DBC. . |
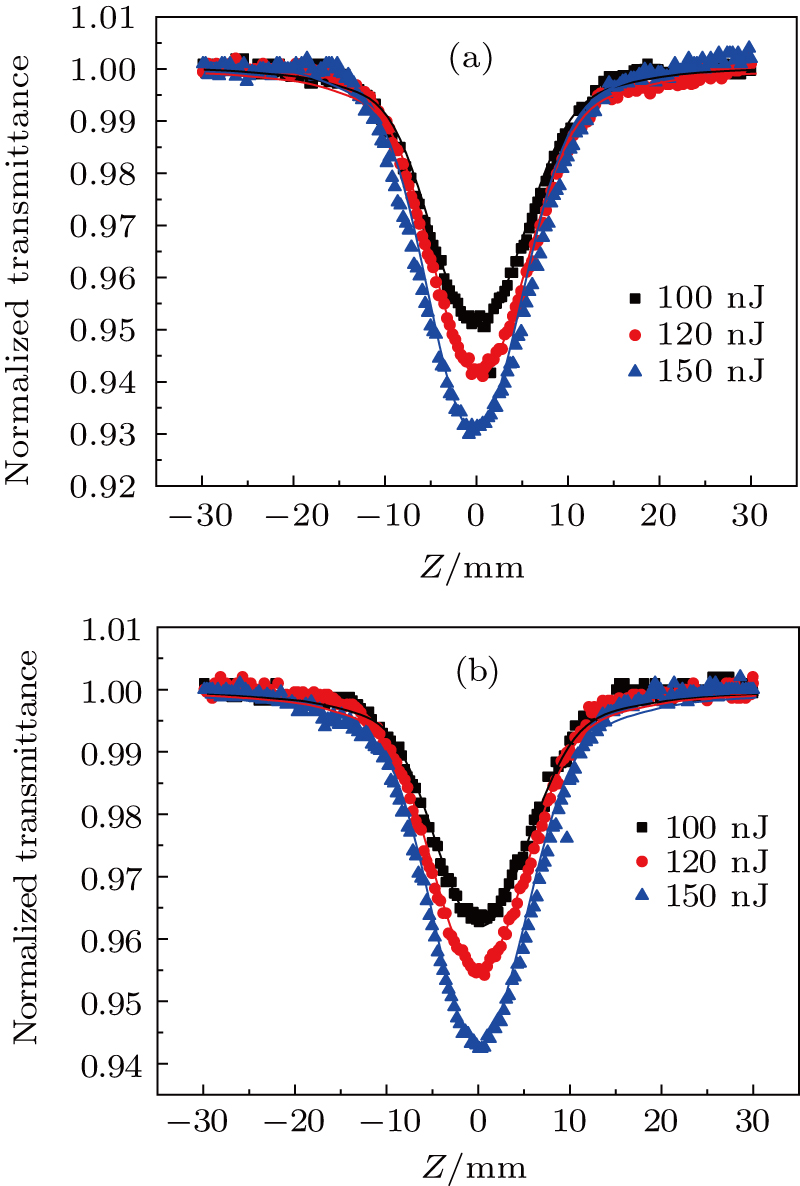
The transient absorption spectroscopy results demonstrate that the modulation can enhance RSA in the near-infrared band. With the aim of evaluating the influence of our modulation on nonlinear coefficients in the near-infrared band, open-aperture and closed-aperture Z-scan measurements with femtosecond laser pulses were conducted at different wavelengths. DBC and DPC were dissolved in DMSO using the same concentrations as in the transient absorption spectroscopy. The solvent itself did not exhibit any nonlinear absorption under our experimental conditions. We presumed that two-photon absorption (TPA) dominated the nonlinear mechanism on the femtosecond time scale because the molecules we were studying did not show any linear absorption in the near-infrared region (Fig.
| Table 2.
TPA coefficients βeff for DPC and DBC at different levels of light intensity I at 760 nm. . |
Furthermore, we observed TPA and the third-order Kerr refraction nonlinearities caused by TPA using Z-scan experiments at wavelengths ranging from 760 nm to 1000 nm (Fig. S1, supporting information). The DMSO solvent has a nonlinear refraction. To avoid the influence of the solvent, the nonlinear refraction of the solvent was eliminated and thereby we obtained the nonlinear refraction of the pure sample. Through theoretical fitting, the values of the effective nonlinear absorption coefficient (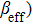
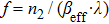
| Table 3.
TPA coefficients ( |
Table
In summary, two materials with a D-π-A structure based on the diphenylacetylene (D) group and the carbaldehyde (A) group were prepared. The response of transient absorption spectra can be modulated by tuning the population of the LE states. Moreover, the RSA and NLO coefficients can also be enhanced under the modulation. Our experimental results show that DPC with a lower population of LE states, due to the extended π-conjugated system, exhibits a wide RSA from visible to near-infrared in transient absorption spectra. Our research on modulating the transient absorption spectra is aimed at guiding the synthesis of broadband NLO materials. The modulation of transient absorption spectra within these types of molecular materials results in fascinating properties, which makes the materials promising candidates in the field of NLO, optical switching, and solar cells.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] |