† Corresponding author. E-mail:
Thermoelectric (TE) materials have been considered as a strong candidate for recovering the waste heat from industry and vehicles due to the ability to convert heat directly into electricity. Recently, multinary diamond-like chalcogenides (MDLCs), such as CuInTe2, Cu2SnSe3, Cu3SbSe4, Cu2ZnSnSe4, etc., are eco-friendly Pb-free TE materials with relatively large Seebeck coefficient and low thermal conductivity and have aroused intensive research as a popular theme in the TE field. In this review, we summarize the TE performance and device development of MDLCs. The features of crystalline and electronic structure are first analyzed, and then the strategies that have emerged to enhance the TE figure of merits of these materials are illustrated in detail. The final part of this review describes the advance in TE device research for MDLCs. In the outlook, the challenges and future directions are also discussed to promote the further development of MDLCs TE materials.
The ever-growing concern around the energy crisis and environmental pollution have motivated scientists to study and design alternative energy technologies.[1,2] Thermoelectric (TE) materials, which can directly convert heat into electricity or vice versa, have received a great deal of attention as a robust candidate for the waste heat recovery and solid state refrigeration. Its efficiency depends on the figure of merit, 


Multinary diamond-like chalcogenide (MDLC), such as ternary I–III–VI2, I2–IV–VI3, I3–V–VI4 compounds, and quaternary I2–II–IV–VI4 compounds (Fig.
 | Fig. 1. (color online) (a) Different crystal structures of multinary diamond-like chalcogenides (MDLCs) derived from cubic zincblende structure; (b) timeline for thermoelectric bulk materials with diamond-like structures, showing data from Cu(In, Ga)Te2,[21–32] AgGaTe2,[33] AgInSe2,[34,35] CuFeS2,[36–44] Cu2SnS3,[45,46] Cu2GeSe3,[47] Cu2SnSe3,[48,49] Cu3SbS4,[50] Cu3SbSe4,[51–60] Cu2ZnSnS4,[61] Cu2ZnSnSe4,[19,20,62,63] Cu2ZnGeSe4,[64] Cu2CdSnSe4,[65] Cu2FeSnSe4,[66] and Cu2CoSnS4.[67] |
In this review, we first analyze the crystalline and electronic structure properties of MDLCs briefly, and then the strategies for improving the TE properties of MDLCs are described in detail, including carrier concentration regulation, band structure engineering, the decrease of κL and synergistic regulation. Finally, challenges and directions for this type of semiconductor in future commercial applications are also discussed.
MDLCs and their analogies can be taken as derivatives from cubic zincblende binary II–VI compounds (like ZnSe) through orderly substitution of II atoms with the same number of I + III, I2+IV, I3 +V or I2 + II + IV atoms (Fig. 
 | Fig. 2. (color online) (a) Structure of Cu2ZnSnSe4 with alternative (Cu2Se4) and (ZnSnSe4) layers; (b) the temperature-dependent κL of Cu2ZnSnSe4;[20] [(c)–(e)] the orbital-decomposed band structures of Cu2ZnSnSe4.[73] Reproduced from Ref. [73] with permission from the Royal Society of Chemistry. |
Owing to the similar characteristics in the crystalline structure, MDLCs have some similarities in electronic band structure. It should also be noted that the electronic structure for MDLCs was calculated very early,[68,69] and the results have become more and more accurate recently with the development of density functional theory.[70] For Cu-based ternary and quaternary chalcogenides, the valence band maximum (VBM) mainly contains a hybridization of Cu 3d orbitals and VI p orbitals because Cu has higher d orbital energy than the other cations, whereas the conduction band minimum (CBM) is primarily derived from the II/III/IV/V s orbital and VI p orbitals due to the lower s orbital energy of III/IV/V than the other cations.[14,70–72] For stannite Cu2ZnSnSe4 compound, the (Cu2Se4 tetrahedral slabs serve as the conduction pathway for holes and the insulating (ZnSnSe4) ones can work as a charge reservoir (Fig.
The intrinsic zT values for most of MDLCs are still very low primarily because their carrier concentrations are considerably lower than their corresponding optimal carrier concentrations, and thus resulting in the poor electrical transport properties. Thus, the common method of elemental doping is widely employed to increase carrier concentrations in MDLCs for achieving a high zT.[75] Importantly, the choice of doping site does lead to a difference in carrier mobility for heavily doped semiconductor, and high mobility is expected to be obtained when dopants are on the sub-lattice which makes less contributions to the formation of the charge-conducting band in principle, which is termed ‘modulation doping on the atomic level (MDAL)’ by Wang in the TE field.[76] High mobility is experimentally achieved from the anion-doped sample while the cation dopant is distinctly inferior in PbSe. We should point out that the enhanced mobility by this method can be observed when the band structure does not change (at least not significantly) with dopant. According to this concept, II/III/IV/V sites are reasonable doping places to increase the carrier concentration in p-type MDLC due to the above band structure features that the I–IV bonds dominate the p-type electrical transport while other elements (i.e., II/III/IV/V) exhibit weak contributions to the conductive framework. In experiment, substituting Sn with Indium was used as an effective guide to improve the carrier concentration in diamond-like Cu2SnSe3 compound and the generated hole would smoothly transport through three-dimensional (3D) Cu–Se conductive framework, thereby greatly enhancing the electrical properties.[48] Meanwhile, the copper deficiencies in Cu2GeSe3 are not effective in increasing the electrical properties.[77] Other investigations of GeSb/SnSb in Cu3SbSe4,[51,52,55–57] ZnM in CuMTe2 (M=Ga, In),[78–80] CuZn/InSn in Cu2ZnSnSe4,[19,20] etc. also demonstrate the effectiveness of doping at II/III/IV/V sites in the enhancement of electrical properties and thus highly improving zT s. In addition, the possible presence of donor–acceptor defect pairs in MDLCs, such as (




Previous studies indicated that the high band degeneracy (i.e., high 
(i) Pseudocubic approach
A high Nv number, which is normally observed in a crystal structure with high symmetry, is desired for gaining both a large Seebeck coefficient and high electrical conductivity. Snyder et al. successfully implemented the band-convergence scheme in Se-alloyed PbTe for the first time and attained an excellent zT value of ∼1.8.[7] This scheme has also been successively applied in other highly symmetric semiconductors (e.g., Mg2Si, SnTe, half–Heusler, and CoSb3).[82–85] In 2014, Zhang et al. expanded it to chalcopyrite with low symmetry, and devised a ‘pseudocubic approach’ (Fig. 
 | Fig. 3. (color online) Schematic diagrams illustrating the pseudocubic band-convergence, showing (a) crystal structure and schematic electronic bands of cubic zinc blende structure, and (b) crystal structure and schematic electronic bands of ternary chalcopyrites. The c and a are the lattice constants. 




|
(ii) Highly-efficient-doping (HED)
As for TE materials possessing intrinsically high band degeneracy, heavy doping by guest atoms to reach the optimal carrier concentration will more or less cause the degenerate bands to destruct, leading to both the Seebeck coefficient and the power factor decreasing. Thus, selecting highly efficient dopants which can provide optimal carrier concentration at relatively low doping content, should be a feasible attempt to improve the electrical conductivity while maintaining the large Seebeck coefficient with little divergence of degenerate bands.[60] As illustrated in Fig.
 | Fig. 4. (color online) (a) Schematic diagram illustrating the maintenance of highly degenerate electronic bands of the matrix by highly efficient doping (HED), and (b) curve of S versus carrier concentration showing that the nearly non-changed band structure can be obtained via HED (i.e., Al, Ga, In doping)[60] in Cu3SbSe4 with high band degeneracy. Reproduced from Ref. [6] with permission from the American Chemical Society. |
It was suggested that the accompanying extra entropy in magnetic ions with degeneracy of electronic spin configuration in real space, such as transition metal ions with unpaired 3d electrons, should play a key role in obtaining large S at high σ in cobalt oxide.[61,87,88] This striking fact has encouraged many researchers to explore the possibility of substitution using magnetic ions to improve the TE performances of the other materials. Yao et al. firstly investigated the TE properties of magnetic-ion-substituted chalcopyrite in 2011 and observed the simultaneous optimizing of all three TE parameters in Mn-substituted CuInSe2.[89] The Mn-doping moved the Fermi level (Ef) downward into the region of the valence band, and the Mn 3d level hybridization with the Se 4p level resulted in the finite DOS in the proximity of the Ef, enabling the considerable enhancing of S and σ for CuInSe2. The introduced Mn–Se networks could provide an additional conduction pathway for charge carriers. The increased local disorder in the crystal lattice by Mn substitution could reduce κL, which was also verified by Raman spectroscopy. However, the contribution of the spin entropy has been neglected in the work of Yao et al. In 2013, Xiao et al. reported that magnetic ions could also decouple the strongly interrelated three TE parameters in Cu2ZnSnS4 (Fig.
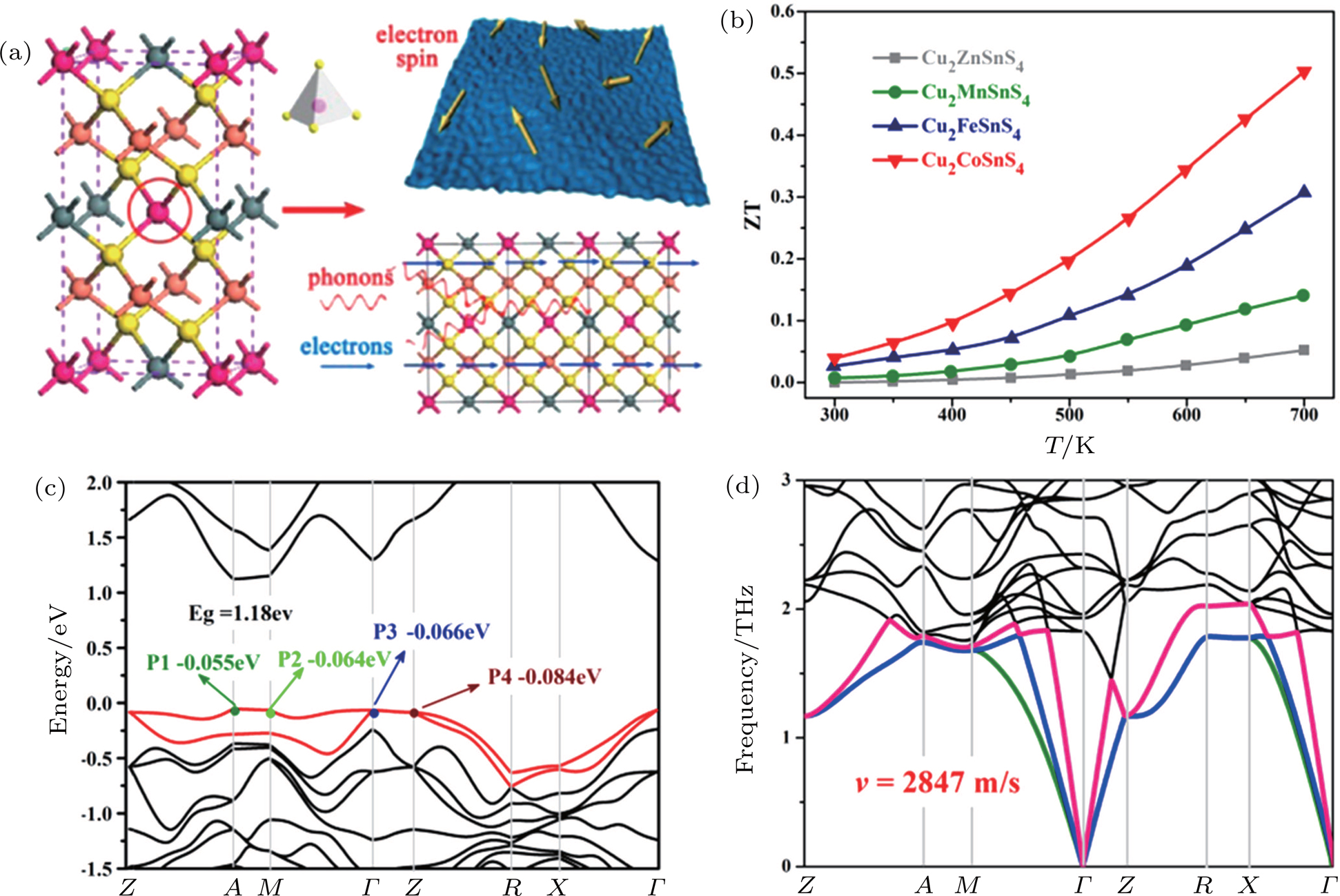 | Fig. 5. (color online) (a) Schematic diagrams illustrating various electron spin and phonon scatterings in quaternary magnetic nanocrystals and (b) comparison of the zT among Cu2XSnS4 nanocrystals.[61] Reproduced from Ref. [61] with permission from the Royal Society of Chemistry, (c) electronic and (d) phonon band structure for Cu2CoSnS4.[67] Reproduced from Ref. [67] with permission from Elsevier. |
Other researches for improving TE performances of Ni-doped Cu3SbS4, Mn-doped CuGaTe2 and cobalt-doped Cu2SnS3,[46,90,91] also proved the effectiveness and good-transplanting of magnetic ion doping strategy in MDLCs. However, most of researches usually focused on explaining the enhancement in S by an additional spin entropy effect or the participation of magnetic-ion-3d states near the Ef in the electronic transport.[46,61,89–92] The above two factors, which may interconnect with each other by magnetic-ion, should be considered together and also clarify the primary cause for enhancing the value of S at different magnetic ion content (e.g., in 2.6% Ni-doped Cu2ZnSnS4, Cu2CoSnS4, and CuFeS2.[36,61,67] Additionally, as the spin-disorder scattering in magnetic ion doping more or less hampers the mobility of carriers,[93] its influence should also be taken into consideration, especially at low temperature. If the reduction in carrier mobility overweighs the increase in S, no enhancement of power factor may appear. Therefore, the relationship between magnetism and TE property still needs further exploring.
It is known that the introduction of point defects is an effective approach to reducing the value of κL by enhancing the phonon scattering due to the mass contrast and strain field fluctuation between the host and guest atoms. According to the Debye–Callaway–Klemens model, the high concentration of defects possessing large mass difference and strain fluctuations can maximize the scattering parameter (Γ) and thus achieve the lowest κL.[75] Pei et al. observed that AgCu substitutional defects reduced the κL of CuGaTe2 more obviously than ZnGa/InGa defects at the same concentration, which is due to the larger mass contrast.[94] However, the maximum mass contrast (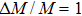
 | Fig. 6. (color online) (a) Room temperature cation defect concentration (nCD)-dependent normalized lattice thermal conductivity (κL) for CuGaTe2 solid solutions with (red) and without (black) vacancies.[95] Reproduced from Ref. [95] with permission from the Royal Society of Chemistry. (b) Plots of room-temperature κL and modeled κL against the molar fraction of selenium of the complete solid solution series Cu2ZnGeSe4−xSx with x = 0–4.[96] Reproduced from Ref. [96] with permission from the American Chemical Society. |
(I) Nanocomposite
The proposed concept of low-dimensional TE materials (e.g. quantum dots, nanowires and superlattices systems) in the 1990s has revealed that the simultaneous optimization of the power factor and κL is possible in nanostructures.[97,98] After that, many experimental studies on nanostructured bulk materials, such as (Bi,Sb)2Te3, PbTe, Si80Ge20,[99–101] have also proved that the nanocomposite approach (i.e., reducing the grain size to nanometers or using nanoparticles) is indeed effective in increasing zT of 3D bulk material due to the reduction in the κL by the selective scattering of phonons or the enhancement in the Seebeck coefficient via preferential scattering of low-energy electrons (i.e., energy filtering).[12] Consequently, the nanocomposite approach is also widely adopted to achieve the expected high zT in MDLC, although it probably causes the electrical conductivity to decrease by the deterioration of carrier mobility. Liu et al. reported a solution-based scalable synthesis approach to produce several grams of monodisperse Cu3SbSe4 nanocrystals (Fig.
 | Fig. 7. (color online) (a) Representative TEM micrographs of Cu3SbSe4 nanocrystallines, and the corresponding HRTEM images, electron diffraction patterns and EELS analysis results.[59] Reproduced from Ref. [59] with permission from the Royal Society of Chemistry. [(b) and (c)] Temperature-dependent κ and zT values for micro- and nano-crystalline Cu3SbSe4.[56,59] |
(II) Nanoinclusion
Nanoinclusion strategies by introducing a nano-sized second phase are effectively utilized to obtain high performance TE materials (e.g., CoSb3, PbTe, SnTe)[104–106] due to the depressed κL by additional phonon scattering or the increased S through energy filtering. Most of the research of nanoinclusion about MDLCs focuses on the CuGaTe2/CuInTe2 type materials currently. In the case of CuGaTe2, the incorporation of graphite nanosheets could enhance the S and power factor due to the energy filtering effect, and also cause the value of κL to decrease below 750 K because of the increased interface scattering. The large zT value increased up to 0.93 at 873 K in CuGaTe2 composite, which had about 21% improvement on pure CuGaTe2.[107] However, the introduction of nanophase Cu2Se into CuGaTe2 reduced the value of S due to the increase of carrier concentration.[32] Benefiting from the greatly reduced value of κL, the final zT approached to 1.2 at 834 K for the CuGaTe2/3vol% Cu2Se sample. By introducing a proper amount of grapheme into CuInTe2, Chen et al. also observed that the κL successfully decreased and also the value of zT was improved.[108]
Recently, high entropy alloy (HEA), which consists of multiple principal elements in an equimolar manner or in a close to equimolar ratio manner, has been proposed as a novel type of alloy with intriguing properties in structure and function.[109,110] Due to the chemical complexity (e.g., large atomic size differences) and high configuration entropy, HEA may significantly introduce extra phonon scattering and also offer a new way to adjust electrons, and resultantly affect the electrical and thermal properties. The HEA was introduced into the context of TE by Shafeie et al. as a potential candidate for future TE material.[111] In this scenario, Liu et al. devised a strategy of using entropy as the global gene-like performance indicator, which shows how multicomponent thermoelectric materials with high entropy could be designed (Fig. 
 | Fig. 8. (color online) (a) Schematic diagrams of the lattice framework in multicomponent materials compared with an ordinary binary compound. (b) Schematic diagram of the entropy engineering with multicomponent TE materials. (c) Maximum confgurational entropy (in units of kB per formula unit) as a function of material solubility parameter δ for given multicomponent TE materials, where n is the number of components. (d) Plots of maximum zT versus confgurational entropy in Cu2(S/Se/Te)-, (Cu/Ag)(In/Ga)Te2-, and Cu8Ge(Se/Te)6-based multicomponent TE materials.[26] Reproduced from Ref. [26] with permission from WILEY-VCH. |
Nano-oxide additives are commonly used to reduce κL by introducing additional phonon scattering into TE materials, and the oxides are usually stable in the host material because of their intrinsically strong bonding.[113–119] In 2016, Luo et al. first discovered the in-situ displacement reaction between the ZnO additive and the CuInTe2 matrix at 714 K by differential scanning calorimeter (DSC) and transmission electron microscopy (TEM) as displayed in Figs. 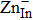

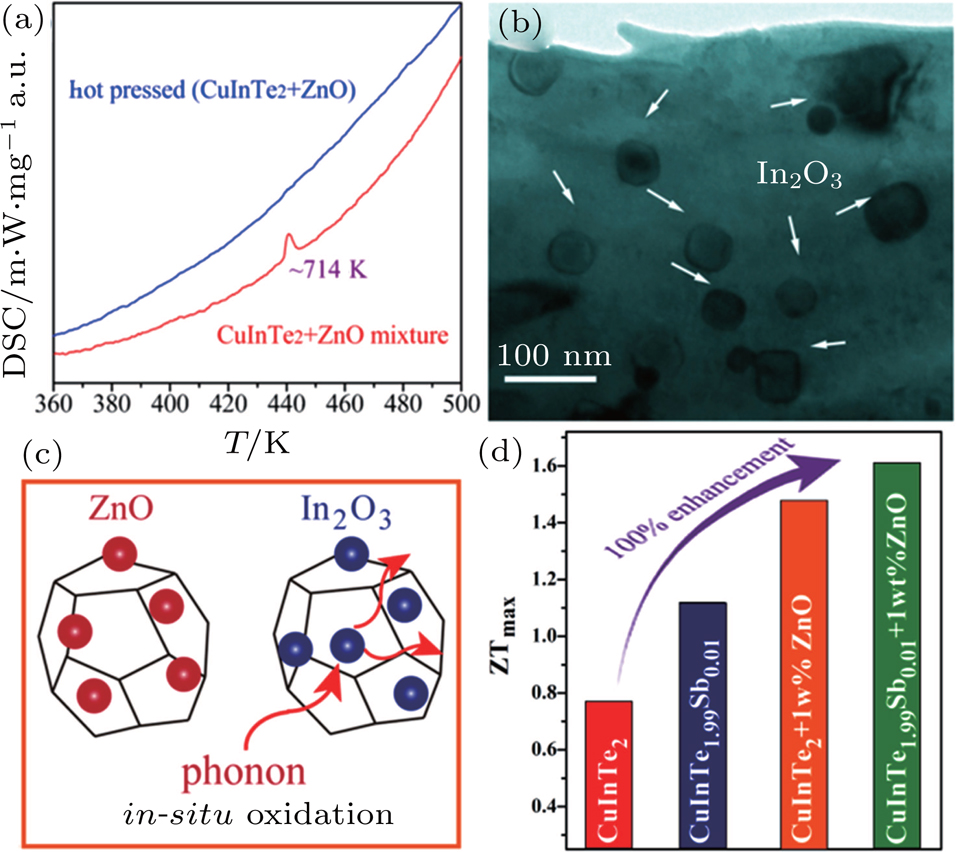 | Fig. 9. (color online) (a) DSC curves of CuInTe2+2.0-wt% ZnO additives before and after hot press process. (b) Highly magnified TEM images of CuInTe2 matrix with dispersed In2O3 nanoparticles. Reproduced from Ref. [25] with permission from Elsevier. (c) Scheme of in-situ displacement reaction. (d) Stepping increase of zT at 823 K in the CuInTe2 samples by Sb substitution, ZnO adding, and combined Sb substitution, and ZnO adding.[25,26] Reproduced from Ref. [26] with permission from WILEY-VCH. |
 | Fig. 10. (color online) (a) Schematic diagram of the structure of the fabricated diamond-like module, and b) plots of output voltage (black) and power (blue) versus current for TE module based on diamond-like materials. Temperature differences between the cold side and the hot side of the module are 420, 470, and 520 K, respectively.[35] |
Since research on the TE properties of MDLCs is still in the early stage, most of the attention is paid to enhancing the zT s and the feasibility to fabricate an efficient TE device based on MDLCs is still an open question. In 2017, researchers started to address this inevitable question and finally opened the door for practical applications of MDLCs TE materials. Due to the obtained high TE performance in (Sn, Bi)-codoped nanocrystalline Cu3SbSe4 sample, Liu et al. used them to fabricate an innovative ring-shaped TE generator with a single p-leg, which could be used to recover the waste heat from exhaust gas pipes of vehicles.[59] The results showed that an open circuit voltage near 20 mV was obtained for a single TE element when the temperature gradient was maintained at 160 K. Then, the first TE module including both n and p-type MDLCs was fabricated by Qiu et al. as shown in Fig.
In this review, we mainly discuss the features in crystal structure and electronic structure, and summarize the effective strategies to enhance zT s for MDLCs. Due to the unique diamond-structure-accompanied properties: flat valence band and large local distortion, MDLCs usually possess both relatively high Seebeck coefficients and low thermal conductivities. A zT value as high as 1.6 is achieved in Pb-free MDLC by entropy engineering or by in-situ displacement reaction, thus highlighting the promising prospect of MDLCs for TE applications. However, more efforts are still needed to promote the practical TE applications of MDLCs. On the one hand, the zT value especially the average one cannot be so good as that of highly efficient PbTe-based TE material, so it is of great importance to further increase the zT s by new strategies.[9,120–122] Recently, it was demonstrated that dislocation arrays, which effectively scatter phonons and maintain high carrier mobility could concurrently optimize the electrical and thermal transport properties, leading to a great improvement of both power factor and zT in CoSb3- or (Bi,Sb)2Te3-based TE materials.[123,124] Pei et al. also noted that the dense in-grain dislocations indeed took the major responsibility for the decrease of κL through scattering the remaining mid-frequency phonons thereby resulting in the extremely low κL approaching to the amorphous limit.[125] Hence, dislocation engineering is a good direction for further obtaining advanced MDLC TE materials. Besides, the resonant levels play an important role in enhancing both the average power factor and zT value in SnTe, which is mainly due to the increased S.[126,127] Nanoheterostructure-based nanocomposites (e.g., PbTe–Ag2Te, PbTe–BiSbTe) present both the improved power factor and the depressed κL because of minority carrier blocking.[128,129] Can the resonant levels also increase S or does the nanoheterostructure block the minority carrier in MDLC?
The poor TE properties in n-type MDLCs also hamper the TE applications of this promising typed material, because a TE device requires both a high-performance n-type leg and a high-performance p-type leg. In the literature, the highest zT in reported n-type MDLCs is still inferior to that of the p-type one, such as 0.126 at 450 K for CuFeInTe3,[130] 0.264 at 500 K for CuFeS2, and 1.1 at 900 K for AgInSe2.[35,36] Meanwhile, it seems difficult to change the intrinsic p-type conduction into an n-type one for most of MDLCs with high TE performance. For instance, zinc-doping heavily at the copper site could not make the n-type Cu3SbSe4 available.[131] The origin of p-type electrical transports in most of MDLCs might be linked to the presence of intrinsic Cu vacancies.[132] Thus, discovering and realizing n-type MDLCs with high TE performance is urgently needed in the future research. In addition, the stability investigation of MDLCs under thermal cycling or large current, which is of great importance in practical applications, should also be focused on.[133]
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [52] | |
| [53] | |
| [54] | |
| [55] | |
| [56] | |
| [57] | |
| [58] | |
| [59] | |
| [60] | |
| [61] | |
| [62] | |
| [63] | |
| [64] | |
| [65] | |
| [66] | |
| [67] | |
| [68] | |
| [69] | |
| [70] | |
| [71] | |
| [72] | |
| [73] | |
| [74] | |
| [75] | |
| [76] | |
| [77] | |
| [78] | |
| [79] | |
| [80] | |
| [81] | |
| [82] | |
| [83] | |
| [84] | |
| [85] | |
| [86] | |
| [87] | |
| [88] | |
| [89] | |
| [90] | |
| [91] | |
| [92] | |
| [93] | |
| [94] | |
| [95] | |
| [96] | |
| [97] | |
| [98] | |
| [99] | |
| [100] | |
| [101] | |
| [102] | |
| [103] | |
| [104] | |
| [105] | |
| [106] | |
| [107] | |
| [108] | |
| [109] | |
| [110] | |
| [111] | |
| [112] | |
| [113] | |
| [114] | |
| [115] | |
| [116] | |
| [117] | |
| [118] | |
| [119] | |
| [120] | |
| [121] | |
| [122] | |
| [123] | |
| [124] | |
| [125] | |
| [126] | |
| [127] | |
| [128] | |
| [129] | |
| [130] | |
| [131] | |
| [132] | |
| [133] |

