† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 61205193), the Project of Science and Technology of Jilin Province, China (Grant No. 20140520107JH), the Technology Foundation of Jilin Provincial Department of Human Resources and Social Security, China (Grant No. RL201306), and the Science Foundation for Young Scientists of Changchun University of Science and Technology, China (Grant No. XQNJJ-2015-03).
Biofunctional europium (III)-doped ZnS (ZnS:Eu) nanocrystals are prepared by a sol–gel method. The characteristic luminescence of ZnS:Eu is used as a probe signal to realize sensitive immunoassay. The luminescence intensity of the Eu3+ in the ZnS matrix shows strong concentration dependence, and the optimal doping concentration is 4%. However, the emission wavelengths of the ZnS:Eu nanocrystals are not dependent on doping concentration nor the temperature (from 100 K to 300 K). Our results show that these features allow for reliable immunoassay. Human immunoglobulin, used as a target analyte, is captured by antibody modified ZnS:Eu probe and is finally enriched on gold substrate for detection. High specificity of the assay is demonstrated by control experiments. The linear detection range is 10 nM–800 nM, and the detection limit is about 9.6 nM.
The rapid development of nanotechnology and biomedicine has greatly aroused interest in the research of semiconductor nanomaterials for biomedical applications.[1] CdSe quantum dots and their core shell structures are the most active luminescent materials currently used in the study of biological markers.[2] However, the toxicity of heavy metal ions has received much attention of researchers and restricts its applications in the fields of biology, medicine, pharmacy, etc.[3,4] Therefore, the development of a new non-toxic matrix material as a fluorescent marker is necessary. The ZnS nanomaterials are non-toxic and exhibit good biological compatibility and chemical stability in the physiological environment, even in a high salt alkali environment.[5] The ZnS nanomaterials can be activated by appropriate element doping, which can greatly improve their luminous performance.[6]
Rare-earth ions are an attractive candidate for doping due to the unique optical properties originating from their special features of electronic configurations.[7] Eu3+ ion, as a typical member of rare-earth ions, is one of the most important activators in red luminescent materials.[8] The Eu3+-activated luminescent materials are widely used in fluorescent lamps, cathode ray tubes, and plasma display panels.[9] However, immunoassay based on the fluorescence spectrum of Eu3+ is nearly ignored. Recently, ultrasensitive immunoassay based on CdS:Eu nanoparticles was reported by Liu et al.[10] The strategy is based on the quenching of the electrochemiluminescence from CdS:Eu. In fact, the fluorescence spectrum of Eu3+ is comprised of several narrow and intense emission lines, which can be used as a characteristic signal to carry immune detection.[11] Since the nontoxicity and easy doping of ZnS, Eu3+-doped nanomaterial with using ZnS as the matrix will be conducive to the obtaining of an environmental friendly fluorescent immune detection.[12]
Herein, ZnS:Eu nanomaterials are prepared by a sol–gel method and biofunctionalized by introducing carboxyl groups. The optical property of the ZnS:Eu nanomaterials is optimized by doping concentration. The wavelength positions of the emission lines are anti-interference by temperature, which provides an accurate and reliable detection. The detection results show that rare-earth-doped ZnS nanomaterials can be used in immunoassays.
The Zn(AC)2⋅2H2O and Na2S⋅9H2O were obtained from Sigma–Aldrich. Thioctic acid (TA), 3-Mercaptopropionic acid (MPA), N-Hydroxysulfosuccinimide sodium salt (NHSS), and Eu(NO)3⋅6H2O were purchased from Aladdin Reagent Company Limited (Co., LTD). 1-ethyl-3-[3-dimethylaminopropyl] carbodiimide (EDC), Bovine serum albumin (BSA), goat anti-human immunoglobulin (IgG) and human IgG were purchased from Beijing Dingguo biotechnology Co., LTD. All chemicals were of analytical grade in purity and directly used without being purified further.
Aqueous synthesis not only solves the problem of the water solubility of the nanomaterials, but also is simpler, less toxic and less expensive than the metal–organic chemical method. Thus, ZnS:Eu nanomaterials were prepared by a modified aqueous method.[13] The improvement was that the carboxyl groups were introduced by MPA to gain a biofunctional surface. The detailed process is as follows: 300-μl MPA was added into 50-ml 75-mM Zn(AC)2⋅2H2O aqueous solution; 50-ml 0.75-mM Eu(NO)3⋅6H2O was added in the mixing process for the 1% doped ZnS:Eu sample. After mixing for 20 min, 50-ml 75-mM Na2S⋅9H2O solution was slowly added to the mixed solution to provide element sulphur. Stirring continued for 3 h to obtain ZnS:Eu sample. The concentration of the Eu3+ was amplified to prepare 2% (3%, 4%, and 5%) doped samples.
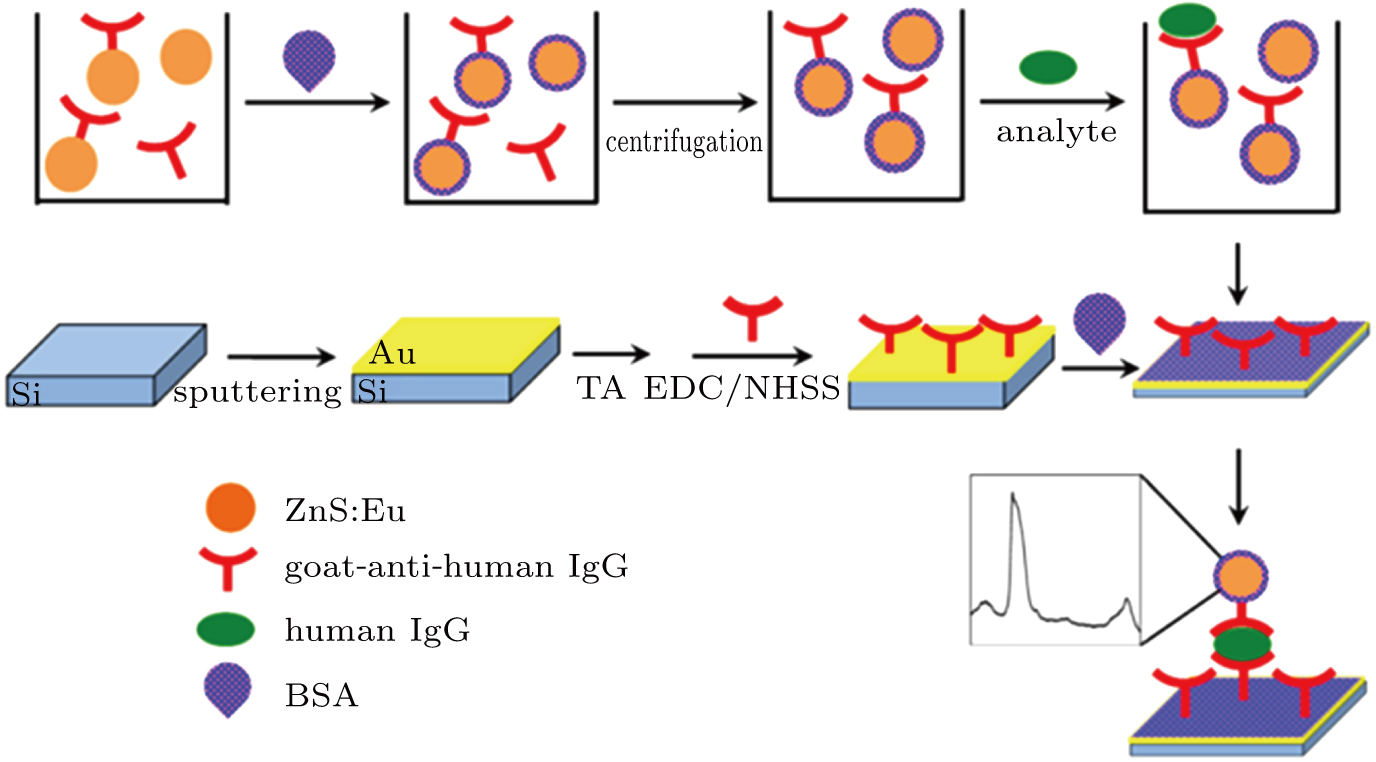
The principle for preparing the fluorescent probe and the sandwich immunoassay is depicted in Fig.
The Au substrate assembly we used in the present investigation (for details, see the research work of Meyerhoff et al.)[15] was obtained by depositing a layer of Au on a single-crystal silicon in vacuum. Because Au and other sulfur containing groups are prone to coordination processes,[16] carboxyl groups were introduced by immersing the Au substrate in a 2% ethanol solution of TA for 24 hours. In each of the following steps, the substrates were carefully rinsed and then dried by using a nitrogen flow. The substrate was then immersed in an EDC/NHSS aqueous solution for 45 min to activate carboxylate groups, followed by a goat anti-human IgG (660 nM) solution at 37 °C with continual rocking for 2 hours. The BSA aqueous solution (1 wt%) was also introduced to avoid nonspecific absorption by incubation for 2 hours at 37 °C. Finally, the Au substrate with immobilized antibody was prepared.
Control experiments were performed to verify the immune function of the nanomaterials. First, the gold substrate was directly immersed into the prepared ZnS:Eu probe solution for 2 h as the blank test. Second, a specified concentration of human IgG molecules (1 μM) was added to the probe solution and rocked for 2 h for analyte capturing. After being centrifuged and redispersed, the solution containing the probes/analyte complexes was prepared. The Au substrate with immobilized antibody was immersed in the solution for 2 h at 37 °C with gentle shaking. Then, the substrate was carefully rinsed and then dried using a nitrogen flow. To verify the sensitivity, five control experiments were performed. The 800 nM (or 600 nM, 400 nM, 200 nM, 100 nM, 10 nM) human IgG was added to the probe solution with all other experimental conditions being the same as those in the second experiment. An unspecific control experiment was also carried out to evaluate the selectivity, where the analyte (human IgG) was replaced with 1-μM rabbit IgG.
Transmission electron microscopy (TEM) images were obtained on a JEOL JEM-2200FS microscope operated at 200 kV. Selected area electronic diffraction (SAED) attached to the TEM was used to analyze the crystal structure. The structure was further determined by x-ray diffraction (XRD, Rigaku D/max 2500). The existence of Eu element and the bonding state were confirmed by x-ray photoelectron spectroscopy (XPS, VG ESCALAB LKII) with a binding energy reference of 284.6 eV for a C 1s line. Fluorescence investigations were performed on the samples by using a Shimadzu RF-5301PC fluorescence (FL) spectrometer with an excitation wavelength of 396 nm. Photoluminescence (PL) investigations of the sample were carried out with a Horiba LabRam HR evolution spectrophotometer excited with a 532-nm laser. For temperature-dependent photoluminescence, the temperature variation range is from 300 K to 100 K.
The morphology and the structure of the synthesized sample are investigated by TEM and SAED measurements. Figure
The interplanar distances calculated by using the three diffraction rings are 3.10 Å, 1.90 Å, and 1.57 Å, which coincide with those of the (111), (220), and (311) planes of cubic zinc-blende ZnS, respectively. The results of XRD measurement further confirms the crystal structure (Fig.
 | Fig. 3. (color online) XRD pattern of the ZnS:Eu nanomaterials with different doping concentrations. |
The influence of Eu3+ doping concentration on the luminescence intensity is discussed in this paper. Figure
Except for the doping concentration, the influence of temperature on the fluorescence is also investigated. Considering the fact that biomolecular may lose activities under high temperature, the temperature range in PL measurement is from 300 K to 100 K. A 532-nm laser is chosen as the excited light to obtain more detailed information about the emissions from Eu3+ ion.[23] With respect to the fluorescence excited by a xenon lamp (as shown in Fig.
The principle used for this assay is similar to a sandwiched enzyme-linked immunosorbent assay (ELISA). Compared with the ELISA, the step of adding an enzymatic substrate is omitted, and the enzyme is replaced by ZnS:Eu nanocrystal, which can provide simpler and more robust assays. The 4% doped ZnS:Eu nanomaterial presents the optimal fluorescence and is chosen as the biolabel to perform immunoassays. A 532-nm laser is used as an excitation source. Figure
An immunoassay is performed by using biofunctionalized ZnS:Eu semiconductor nanocrystals as fluorescent probes. The optimal doping concentration to obtain ZnS:Eu nanomaterials with high fluorescent emission is 4%. The further increasing of the doping concentration will lead to fluorescence quenching. The photoluminescence peak position of the emission originating from 5D0 → 7F2 transition is always around 617 nm when the temperature varies from 100 K to 300 K, which allows the accurate determination of human IgG. The fluorescent probes show high specificity for the target analyte, and the calculated detection limit is about 9.6 nM. The strategy of using Eu3+ doped zinc-based semiconductor can be extended to other materials, such as ZnO, and may play an important role in future biomedicine.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] |