† Corresponding author. E-mail:
Project supported by the National Key Research and Development Program of China (Grant No. 2017YFA0303600) and the National Natural Science Foundation of China (Grant Nos. 11474207 and 11374217).
According to first principle simulations, we theoretically predict a type of stable single-layer graphene oxide (C2O). Using density functional theory (DFT), C2O is found to be a direct gap semiconductor. In addition, we obtain the absorption spectra of the periodic structure of C2O, which show optical anisotropy. To study the optical properties of C2O nanostructures, time-dependent density functional theory (TDDFT) is used. The C2O nanostructure has a strong absorption near 7 eV when the incident light polarizes along the armchair-edge. Besides, we find that the optical properties can be controlled by the edge configuration and the size of the C2O nanostructure. With the elongation strain increasing, the range of light absorption becomes wider and there is a red shift of absorption spectrum.
During the past few decades, surface plasmons (SPs) in nanostructures have attracted a great deal of attention in nanoscience field, because they have remarkable effects on molecular spectroscopy,[1,2] optical waveguides,[3] optoelectronic devices,[4] bio-medical treatments,[5] etc. The SPs are of the harmonic oscillations of free electrons in metal nanostructures. The resonance frequency of SPs is affected by the factors of size,[6,7] shape,[8] and surrounding environment.[9] Usually, to excite plasmon, we mainly use traditional metal nanomaterials such as nanoparticles of gold, silver, copper, etc. However, there is a huge problem that the surface plasmons of the optoelectronic device of precious metal decay quickly in the propagation process. Moreover, the plasmonic device of precious metal has a relatively single performance of resonance mode, which cannot meet the requirements for the diversification of modern electronic devices. To overcome the difficulty, scientists have made great efforts for a long time and found that the metal-free materials can offset the insufficiency, and graphene is the most representative material.
Graphene is one of the most promising two-dimensional (2D) materials, which is structured by a planar honeycomb monolayer of carbon atoms.[10] It permits electrons to flow freely across its surface with particularly low scattering due to its electronic structure-Dirac cone.[11] In the applications of plasmon excitation, graphene has three advantages over precious metals: firstly, π plasmon has a long-range charge transfer excitation; secondly, electrons do not easily decay on the surface of graphene; thirdly, graphene has several vibration modes of energy. Due to the excellent properties of graphene, scientists have also done a lot of research on graphene derivatives, such as the substitutional doping of carbon atoms in graphene with transition metals (TMs) can tune the electronic and magnetic properties of graphene.[12] Moreover, van der Waals heterostructure of graphene and g-GaN can tune the Schottky barrier.[13] In addition, graphene oxide (GO) is also a prominent material of graphene derivatives. A lot of experiments have shown that GO has some superior properties compared with graphene, such as its excellent surface activity,[14] which can be eliminated by small molecules or polymers.[15] Besides, GO has an important status in 2D material for improving the thermal, electrical and mechanical properties.[16–18] Current research on GO is not systematic nor comprehensive, therefore, it is necessary to do more research on GO.
In this paper, we theoretically predict a stable single-layer structure of graphene oxide (C2O). There are two oxygen atoms and four carbon atoms in the primitive cell. We use the first principles theory to study the stability and the electron structure of C2O. Through the calculations of the cohesive energy, formation energy and phonon spectrum of C2O, we obtain that the structure of C2O is stable. In addition, this type of C2O is a direct gap semiconductor from analyzing the electronic band structures and density of states (DOS). To study the optical properties of C2O nanostructures, TDDFT is used. By analyzing the structure of this type of C2O, we find that the structure of C2O is anisotropic when the light polarizes along different directions, which means that the C2O is a promising candidate of photoelectric switch. We focus on studying the optical properties of C2O nanostructures when the light polarizes along the armchair-edge direction. We consider the influence on the optical response by changing the edge configuration and size of the C2O nanostructure. What is more, the influence of changing the elongation strain has also been considered.[19] These studies will provide some theoretical support for the practical application of the optoelectronic devices which are made from C2O nanostructure. The rest of the paper is organized as follows. In Section
All of our calculations were based on first-principle simulations. To study the stability, electronic band structure, density of states, and optical properties of single layer C2O, DFT was used in the generalized gradient approximation (GGA), which is expressed by the Perdew–Burke–Ernzerhof (PBE) functional.[20] The CASTEP[21] package was used with norm-conserving pseudopotentials and a plane-wave cutoff energy of 830 eV. Our structure model was fully relaxed, until the forces were less than 0.01 eV/Å and the energy tolerances were less than 5 × 10−6 eV per atom. A vacuum spacing between the 2D single-layer structures was 20 Å. We adopted the Monkhorst–Pack scheme for k-point sampling of the Brillouin zone with 21 × 21 × 1 for C2O. The phonon dispersion of C2O was calculated by the linear response method.[22] The calculations of the optical properties of C2O nanostructures were performed by the real-space TDDFT code OCTOPUS.[23] We described the oxygen atoms, carbon atoms and hydrogen atoms by the Troullier–Martins pseudopotential.[24] The GGA and PBE functional for the exchange–correlation potential were used in both the ground-state and the excited-state calculations, respectively. The excitation spectra induced by an impulse excitation were extracted by the Fourier transform of dipole strength. We defined the simulation zone by assigning a 6-Å-radius sphere around each atom and a uniform mesh grid of 0.2 Å. In the real-time propagation, Kohn–Sham wave function is evolved for 6000 steps in time steps of Δt = 0.003ℏ eV−1.
The crystal structure of C2O is given in Fig.
| Table 1.
Results for optimized geometries of C2O, obtained by using DFT with the PBE exchange–correlation functional. . |
In addition, we perform the phonon-dispersion calculation of C2O. As shown in Fig.
Furthermore, we calculate the electronic band structure and density of states (DOS) of C2O, through using both PBE and the screened hybrid functional of Heyd–Scuseria–Ernzerhof (HSE06). As shown in Fig.
We investigate the periodic structure of C2O. As shown in Fig.
Many experiments show that materials will have many unique properties, such as surface effect, quantum effect, and volume effect, when the macroscopic material is processed into the nanometer size. These small-scale nanostructures can be used in the fields of biological plasmon imaging, medical treatment, and quantum plasmon transfer. Moreover, in the practical application, the optoelectronic devices need higher and higher integration level and the sizes of primary elements need to be processed to be smaller and smaller until they can be processed into nanoscale. However, as is well known, because of the quantum confinement effect,[27] the optical properties of nanostructure material would have a considerable difference compared with those of macroscopic material. Therefore, in the rest of the paper, we focus on studying the optical properties of C2O nanostructures.
We use Cartesian coordinates and fix atoms in the XY plane, to deal with the rectangular C2O nanostructures that the armchair-edge is parallel to the X-axis direction, and the zigzag-edge is perpendicular to the X-axis direction. In addition, at the edge of rectangular C2O nanostructures, the dangling σ bonds at the edges are passivated by hydrogen atoms. Here, we regard the carbon ring (including oxygen atoms) as a basic unit, then the rectangular C2O nanostructures with different sizes are named X × Y, where X represents the number of basic units in the X-axis direction, Y refers to the number of basic units in the Y-axis direction.
Figure
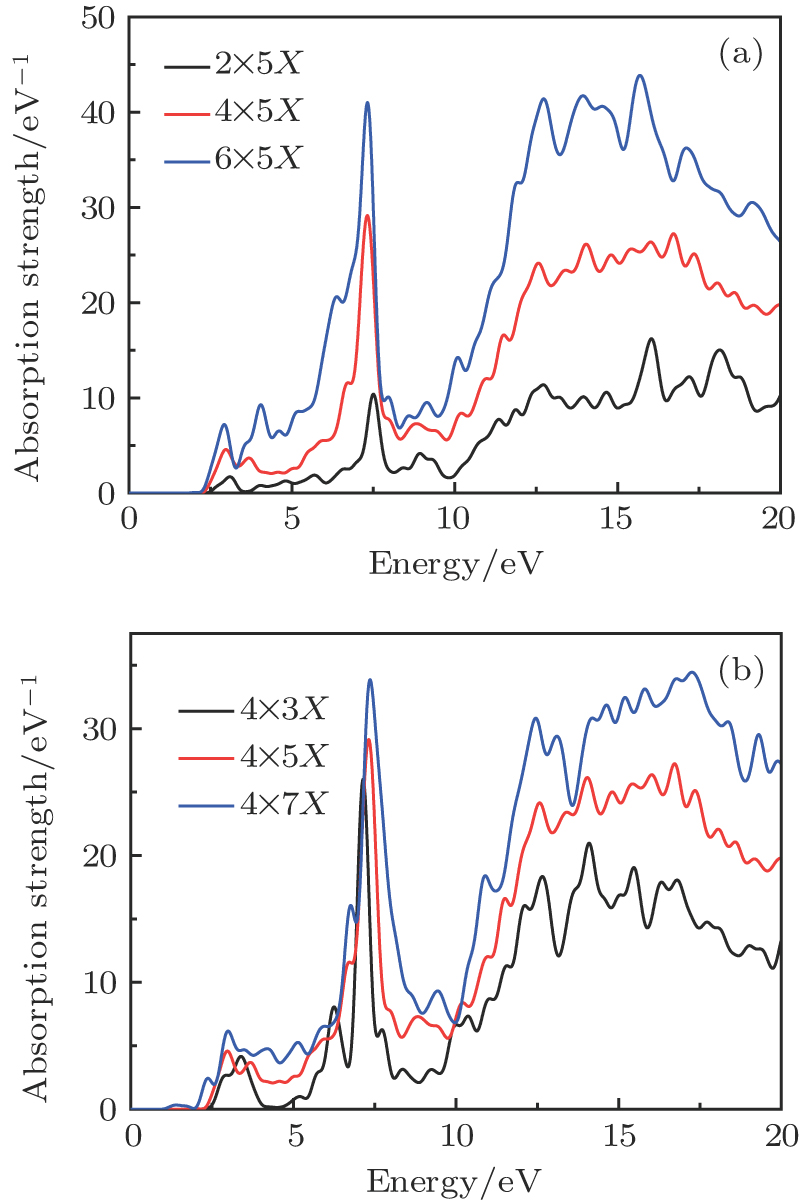
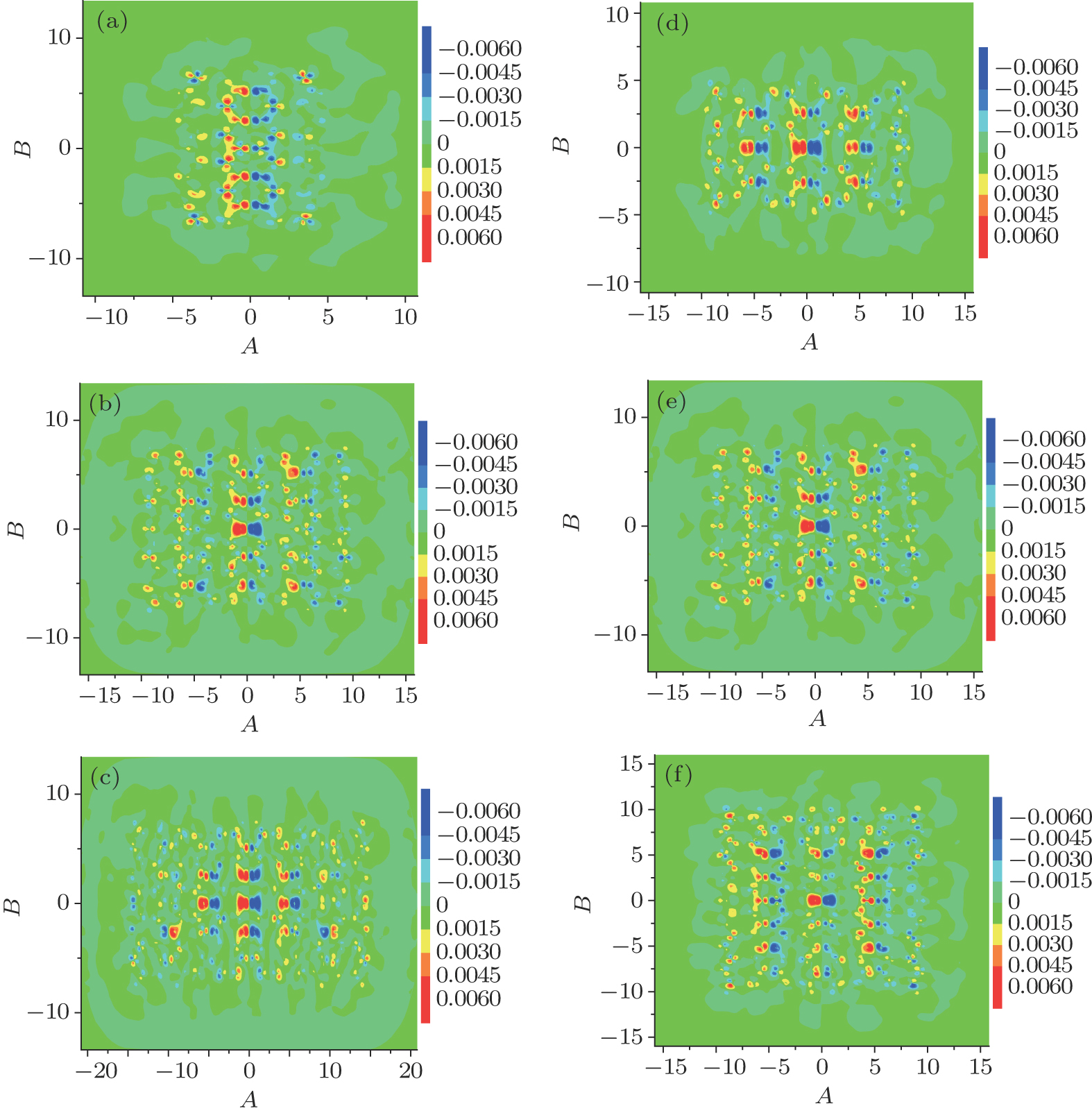
To study the effects of the size and the edge configuration of rectangular C2O nanostructures on their optical properties. We study the 2 × 5, 4 × 5, 6 × 5, 4 × 3, 4 × 7 rectangular C2O nanostructures, which are shown in Fig.
 | Fig. 4. (color online) 2 × 5, 4 × 5, 6 × 5, 4 × 3, 4 × 7 rectangular C2O nanostructures, respectively, in 2D Cartesian coordinate system. |
As shown in Fig.
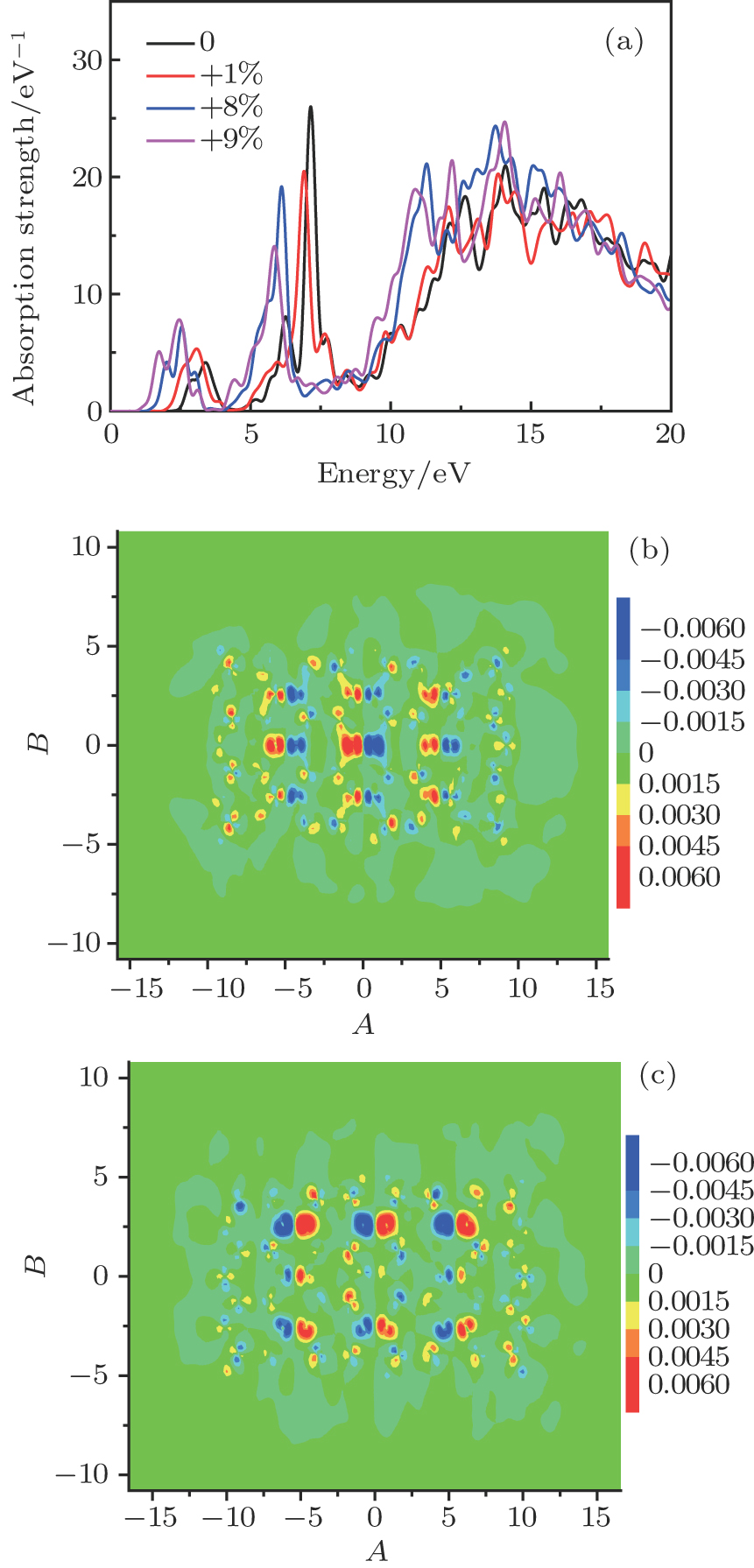
To be applied to a real system, C2O should be grown on a flexible substrate, which may cause a lattice constant mismatch to inevitably occur.[32,33] From many experimental and theoretical studies, we find that the strain has an effect on the optical properties. We can freeze the lattice constants by using the mechanical biaxial strain, which is different from the optimized value. We use η = (a − a0)/a0 to represent a biaxial strain, from which a0 represents an optimized lattice constant, and a refers to the lattice length which is along the strain direction.[34] Positive values of η represents the elongation. The band gaps of strained C2O as a function η are presented in Fig.
In this work, we theoretically predict a highly stable single-layer C2O. We perform first principles simulations to investigate the stability and the electronic structure of C2O. By computing the phonon spectrum, cohesive energy and formation energy, we draw a conclusion that the C2O is kinetically stable. Through analyzing the electronic band structures of C2O and the density of states of C2O, we find this structure is a direct-gap semiconductor. According to TDDFT, we carry out a systematic study of the optical properties of monolayer C2O nanostructure with rectangular geometry. Because of the existence of anisotropy in C2O structure, its absorption spectra show optical anisotropy, which means it would be used as a photoelectric switch. We focus on the optical absorptions of the single-layer C2O nanostructures when the incident light polarizes along the armchair-edge direction. With the increasing of width along the armchair-edge, the absorption strength becomes stronger and the resonance point is red-shifted. In low-energy resonance area, this phenomenon will improve the utilization region of solar energy. With increasing width along the zigzag-edge direction, the absorption strength becomes stronger. The optical properties can be controlled by the edge configuration and size of the C2O nanostructure. In addition, elongation strain also has an effect on the optical properties of C2O nanostructure. With the elongation strain increasing, there is a red shift and the range of absorption spectra become wider. Ours research results reveal that C2O is a promising candidate as a photoelectric material.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] |