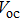† Corresponding author. E-mail:
Colloidal quantum dot (CQD) solar cells have attracted great interest due to their low cost and superior photo-electric properties. Remarkable improvements in cell performances of both quantum dot sensitized solar cells (QDSCs) and PbX (X = S, Se) based CQD solar cells have been achieved in recent years, and the power conversion efficiencies (PCEs) exceeding 12% were reported so far. In this review, we will focus on the recent progress in CQD solar cells. We firstly summarize the advance of CQD sensitizer materials and the strategies for enhancing carrier collection efficiency in QDSCs, including developing multi-component alloyed CQDs and core-shell structured CQDs, as well as various methods to suppress interfacial carrier recombination. Then, we discuss the device architecture development of PbX CQD based solar cells and surface/interface passivation methods to increase light absorption and carrier extraction efficiencies. Finally, a short summary, challenge, and perspective are given.
Solar energy as renewable energy has drawn increasing attention, developing high efficiency and low-cost photovoltaic techniques has been regarded as a direct and effective way to solve energy-shortage and environmental pollution problems. Although traditional silicon-based solar cells and Cu(In,Ga)Se2, CdTe thin-film solar cells possess high efficiency and mature manufacturing technologies,[1–3] it is still tough to further reduce the cost for wide application. Therefore, efforts toward seeking new materials and developing new technologies for low-cost solar cells are ongoing. Currently, some new thin film solar cells are developing rapidly, such as perovskite solar cells (PSCs),[4,5] dye-sensitized solar cells (DSCs),[6,7] organic photovoltaics (OPVs),[8] quantum dot solar cells (QSCs),[9–11] inorganic–organic hybrid solar cells,[12,13] etc., which are found to be promising for next generation solar cells.
Colloidal quantum dots (CQDs) as potential absorbing materials have been explored for more than 20 years duo to their specific optoelectronic and optical properties. They feature size- and composition-dependent absorption onset, high extinction coefficient, and multiple exciton generation (MEG) effect, especially the MEG effect would lead to incident-photon-to-current efficiencies of over 100%.[14–17] Besides, the CQDs allow energy level matching between desired donor and acceptor materials, which is crucial to efficient photovoltaic devices. Therefore, CQD-based solar cells offer the possibility of boosting the PCE beyond the traditional Schockley-Queisser limit of 33%.[18,19] Various device structures have been developed for CQDs based solar cells, including Schottky solar cells, depleted heterojunction solar cells, extremely thin absorber (ETA) solar cells, quantum dot sensitized solar cells (QDSCs), and inorganic–organic heterojunction solar cells. A remarkable progress has been achieved just in the last couple of years by developing more efficient CQDs synthesis techniques, optimizing device architectures, and improving charge transportation. Amongst, QDSCs and PbX (X = S, Se) CQD-based thin film solar cells exhibit better cell performance, and a PCE of 12.34% for QDSCs and a certified PCE of 13.4% for PbS CQD based solar cells have been achieved.[20,21]
Several review articles have been published on quantum dot solar cells. Emin et al. summarized the working principle and the architecture of various CQDs-based solar cells.[22] Yamaguchi et al. focused on various dynamic processes including photo-generation, spatial separation, transfer, and recombination for CQD-sensitized solar cells and CQD heterojunction solar cells.[23] Zhong et al. discussed the QD deposition methods and charge recombination control in QDSCs.[24,25] Sargent et al. summarized the architecture and charge-extraction strategies of CQD solar cells. In this review, advances in QDSCs and PbX based CQD solar cells in the last couple of years will be focused on, including CQD materials and approaches of suppression charge recombination for QDSCs, the progress in device architecture and surface passivation methods of PbX CQD based solar cells.[26,27] Finally, a brief summary, challenge, and prospect will be given.
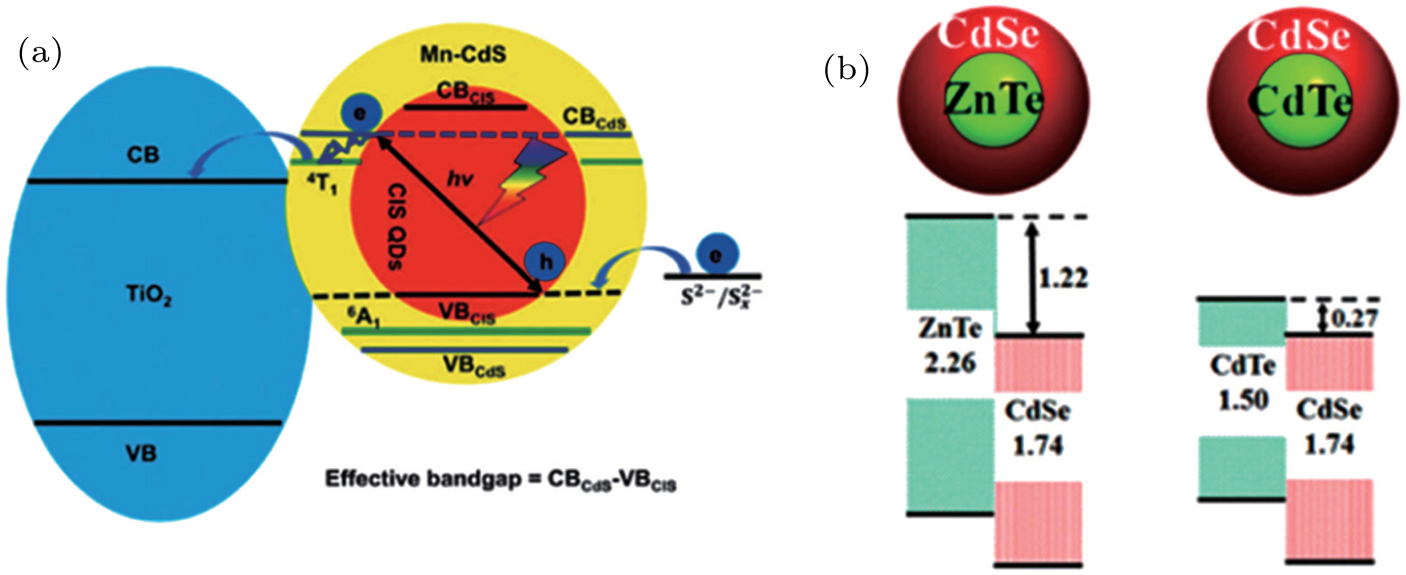
Figure 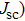

 | Fig. 1. (color online) (a) Architecture and working mechanism of QDSC (FTO: fluorinated tin oxide; VB: valence band; CB: conduction band; Ox: oxidized species; Red: reduced species.[28] (b) Main charge transfer processes occurred at TiO2/QD/electrolyte interfaces including six major recombination paths: (i) recombination between photo-generated electrons in TiO2 and sub-bands (or traps ) in QDs; (ii) recombination between photo-generated electrons in TiO2 and holes in QDs; (iii) recombination between photo-generated electrons in QDs and oxidized species in the electrolyte; (iv) irradiative recombination between electrons and holes in QDs; (v) photo-generated electrons captured by traps; (vi) recombination between photo-generated electrons and oxidized species in the electrolyte. |
There are two ways to sensitize TiO2 photoanode by in-situ and ex-situ methods. In-situ deposition of QDs (CdS, CdSe, PbS, PbSe, etc.) on mesoporous TiO2 film surface mainly can be realized by chemical bath deposition (CBD) or successive ionic layer adsorption and reaction (SILAR) method. This deposition method will lead to high QDs loading, but it is difficult to precisely control the QDs size distribution, therefore, the surface trap state density of the QDs is generally high, which hinders the further development of QDSCs.[29–31] In the ex-situ method, the CQDs are synthesized first, then adsorbed on the TiO2 photoanode in the aid of surface linker. For this method, the QDs are size-controlled and high-quality with low surface trap states, but the low QDs surface coverage and low electron collection efficiency caused by organic long chain ligands on the CQD surface will lead to unsatisfactory performance, thus the 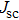
Ideal QD sensitizers for QDSCs need to have narrow band gaps, high conduction band edge to ensure enough light absorption, and smooth electron injection from QDs to TiO2. In QDSCs, CdS, CdSe, and CdS/CdSe co-sensitizers are the most studied QDs via in-situ method,[32,33] but their relatively narrow light absorption range limits the 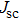



PbS or PbSe CQDs have a high absorption coefficient of 1×105–5×105 cm−1 and a wide range of tunable band gaps owning to their large Bohr exciton radius, which is in favor of high 

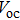
Developing new CQDs materials with wider photo-response range is one of the most effective ways to further improve QDSCs performance. Ternary or multi-component alloyed CQDs are good choice, which are strongly dependent on the synthetic techniques, such as CuInS2, CdSeTe, CuInSe2, CuInSe1−xSx, Zn–Cu–In–Se, CuInTe2−xSex, etc,[45,48–52] as shown in Fig.
 | Fig. 2. (color online) Highly efficient QDSCs based on alloyed CQDs. (a) Schematic energy level diagram and corresponding device performances of CdSe, CdS, and CdSeTe CQDs.[44] (b) Schematic of CuInTe2−xSex CQD and EQE curve of the corresponding CuInTe2−xSex based QDSCs.[45] (c) J–V curves of CuInS2-Zn and CuInS2-based champion cells.[46] (d) The champion device based on Zn–Cu–In–Se alloyed CQDs with a certified PCE of 11.61%.[47] |
Besides, type-II core-shell CQDs can also extend the spectral response range due to narrower effective band gap; meanwhile, the type-II level structure is beneficial to spatial separation of electrons and holes, thus prolonging excited-state lifetimes. This is crucial to multi-excition generation and efficient QDSCs.[61] Type-II core-shell CQDs can be obtained in either aqueous or organic systems. Much recent research efforts have been devoted to preparation of various type-II core-shell CQD structures, like CdSe/ZnSe, CdTe/CdSe,[62] ZnTe/CdSe, ZnSe/CdS, CuInS2/CdS,[63] etc. As shown in Fig. 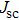
Recently, QDSCs have experienced rapid development, and the cell efficiency has been significantly improved from 5% in 2012 to current 12%. In fact, the preparation of high quality CQDs and development of a capping ligand-induced self-assembly approach have made a tremendous contribution, which gradually show great advantages over the in-situ deposition method (SILAR and CBD). Sharp excitonic absorption peak and relatively high photoluminescence (PL) intensity are usually observed in high-quality CQDs, indicating its low non-radiative recombination rate, high crystallinity, and low trap state density. However, such a distinct feature cannot be achieved by SILAR or CBD deposition methods. Moreover, for highly efficient QDSCs, effective suppression toward charge recombination and selection of superb counter electrode materials are also important.
Currently, the PCEs of QDSCs are still unsatisfied in comparison to those of PSCs and the theoretical efficiency of 44%, which are often attributed to QD surface states or back electron transfer at the solid–liquid interface. The surface traps sometimes called trap states may interfere with electrons injected from QDs to TiO2. Passivation of surface states is often adopted by molecular modification or deposition of another semiconductor material, which is usually realized by a solution process. ZnS as the passivation layer by SILAR method has already been a routine procedure to prepare QDSCs. It is demonstrated that ZnS passivation can enhance the charge injection efficiency largely by reducing carrier trapping and recombination in QDs, and play an important role in the stability and the cell efficiency: (i) to prevent the QDs photocorrosion in the electrolyte and improve the device stability; (ii) to prevent reverse electron transfer from TiO2 to electrolyte and increase charge efficiency; (iii) to reduce surface trap states of QDs and thus increasing electron injection efficiency. Furthermore, ZnSe is found to be another efficient passivation material to replace ZnS. Wang et al. suggested that ZnSe may be a more efficient passivation layer than ZnS, which was attributed to a type II energy band alignment between CdS/CdSe QDs (core) and ZnSe layer (shell), leading to more efficient electron–hole separation and slower electron recombination.[65] Besides, wide bandgap metal oxide materials, such as MgO, ZrO2, SiO2, and Al2O3 deposited on TiO2/QDs surface can also be used as energy barriers to prevent the recombination between photogenerated electrons in TiO2 and holes in the electrolyte. Atomic layer deposition (ALD) technology is one of the most reliable ways to prepare passivation layer due to its conformal coating, high purity, and molecular-level control in thickness and composition.[66–68]
Furthermore, the post-treatment method is adopted to passivate defects and suppress the recombination. Zhong et al. employed a ZnS/SiO2 double layer onto TiO2/QDs surface through SILAR (ZnS) and hydrolysis (SiO2) processes in sequence, interfacial recombination was remarkably inhibited with higher 


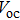
 | Fig. 4. (color online) (a) A modified passivation method toward the photoanode by using an amorphous TiO2 (am-TiO2)/ZnS/SiO2 barrier layer, and (b) CdSeTe QDSCs based on this passivation method exhibit a PCE of 9.28%.[69] |
 | Fig. 5. (color online) CdSeTe QDSCs based on fumed silica (SiO2) modified polysulfide electrolyte achieved a certified PCE of 11.3% and better stability. PEG: polyethylene glycol.[58] |
As we know, the 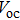

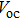



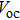
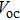
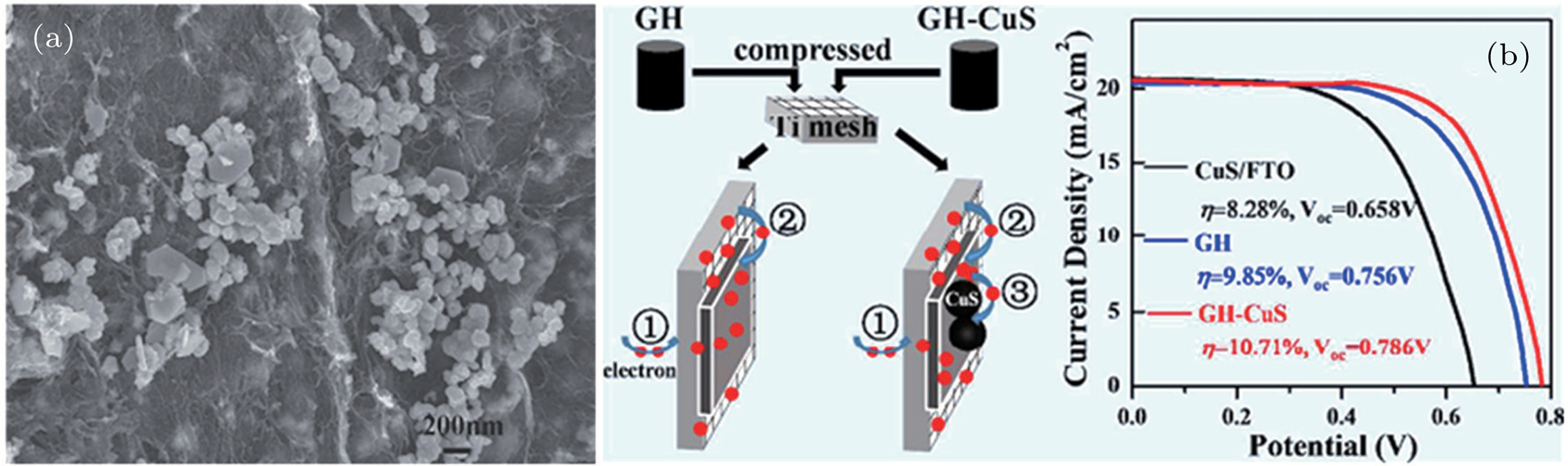 | Fig. 6. (color online) (a) FESEM image of graphene hydrogels (GHs)-CuS hybrid. (b) Fabrication of GH-CuS CE and the cell performance of CdSeTe QDSCs with various CEs.[80] |
| Table 1.
Some advances in highly efficient QDSCs in the past few years. . |
| Table 2.
Some advances of PbX CQDs based solar cells in the past five years. . |
PbX (X = S, Se) CQDs possess tunable near-infrared absorption, multiple exciton generation effect, and solution-processed ability, which have emerged as attractive candidates for thin film photovoltaics.[88] PbS and PbSe CQDs are the most common types of CQD materials used in quantum dot thin film solar cells, which possess narrow bulk bandgaps of 0.41 eV and 0.27 eV, respectively, making them ideal candidates for tuning the absorption spectra in the near-IR range of the sun’s spectrum.[89] Hot-injection method has been employed to prepare PbS or PbSe CQDs, that is, S or Se precursor solution is injected into Pb precursor solution, and precursor decomposition and rapid nucleation processes are typically involved. The most common precursors for PbS and PbSe syntheses are lead oxide (PbO), oleic acid, and bis(trimethylsilyl)sulfide ((TMS)2S) or bis(trimethylsilyl)selenide ((TMS)2Se).[90,91] CQD sizes can be controlled by varying the injection temperature, precursor concentration, conversion rate, and saturation degree, the excitonic peaks are in the range of 880–1600 nm, corresponding to CQD diameters of 3–6.5 nm.[92]
However, the benefits of the tunability and processability of CQDs are counter-balanced by the challenge of achieving superior device performance. In the meantime, high light absorption and charge collection efficiencies are two major requirements to increase the solar cell efficiency. Progress in device architecture as well as advances in managing CQDs surface chemistry have recently led to significant improvement on PbX CQDs based solar cells and the best certified PCE of 13.4% has been achieved.[20] Here, recent progress of PbX CQDs solar cell performance mainly on device architecture engineering, surface chemistry programming, and structured electrodes to improve carrier collection, especially the significant advances in CQD surface passivation, will be reviewed.
As shown in Fig.
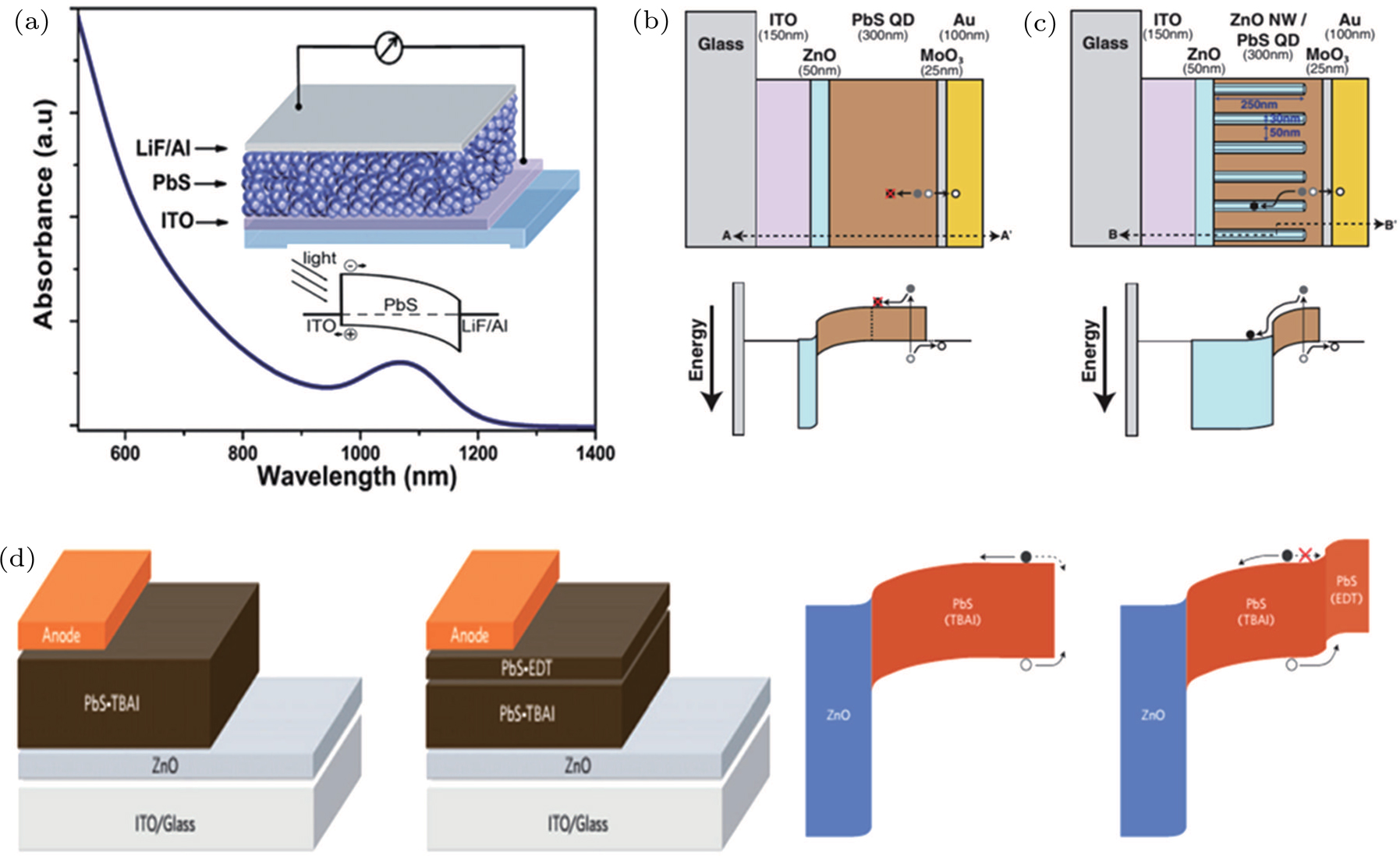 | Fig. 7. (color online) The evolution progress of CQD solar cell architectures. (a) The schematic of Schottky CQD solar cell structure and diagram of energy levels.[93] The schematics and energy band diagrams of (b) planar depleted heterojunction solar cell and (c) bulk heterojunction solar cell.[94] (d) Device architectures and energy bending in ZnO/PbS-TBAI and ZnO/PbS-TBAI/PbS-EDT devices at short-circuit conditions.[95] |
In early research stage of CQD based solar cells, a Schottky structure was employed, as shown in Fig. 


In 2010, planar depleted heterojunction CQD solar cells were developed to overcome the limitation of Schottky solar cells by permitting somewhat thicker active layers and moving the depletion layer to the front contact to suppress recombination losses.[105] Figure 

Bulk depleted heterojunction architectures suggest a path forward for further improving the cell performance. As shown in Fig. 

 | Fig. 8. (color online) The introduction of n-PCBM buffer layer leads to enhanced device performance. The black lines represent the control device, and the red lines represent the n-PCBM modified device.[111] |
From Schottky to heterojunction solar cells, the device performance has experienced a rapid improvement, and the remarkable improvement of PCE has been achieved in less than a decade. Notably, prior solar cells employed p-type CQDs absorber and n-type electrode (such as TiO2) to form a rectifying junction and efficient photovoltaic devices.[110] Current depleted-heterojunction solar cells rely on careful tailoring toward the band offset between p-type CQDs and TiO2 electrode. In contrast, quantum homo-junction can overcome this drawback wherein p-type CQDs and n-type CQDs are employed, as shown in Fig.
 | Fig. 9. (color online) (a) The structure and energy band alignment diagrams of quantum junction and depleted heterojunction. (b) The simulated device performance (
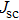
|
 | Fig. 10. (color online) Comparison of iodide-, chloride-, and bromide-treated CQD solids and corresponding devices. (a) Three devices performance tested in air; EQE of iodide (b) and bromide (c) solid-state ligand-exchanged devices observed before and after air exposure; (d) Fermi level (


|
It is demonstrated that increasing doping concentration in PbS-EDT layer can shift the distribution of the depletion region towards the PbS-TBAI layer, allowing for higher carrier mobility and more efficient charge extraction. For example, NaHS was used to treat PbS-EDT layer, leading to threefold increase and thus enhanced 
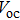
The halide treatment has promoted the CQD photovoltaics development in the past few years. Ning et al. successfully obtained air-stable n-type PbS CQDs based on iodide ligands, whose electronic properties can be controlled by the halide doping density.[124] A graded-doping structure was further constructed, which presented a higher 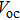

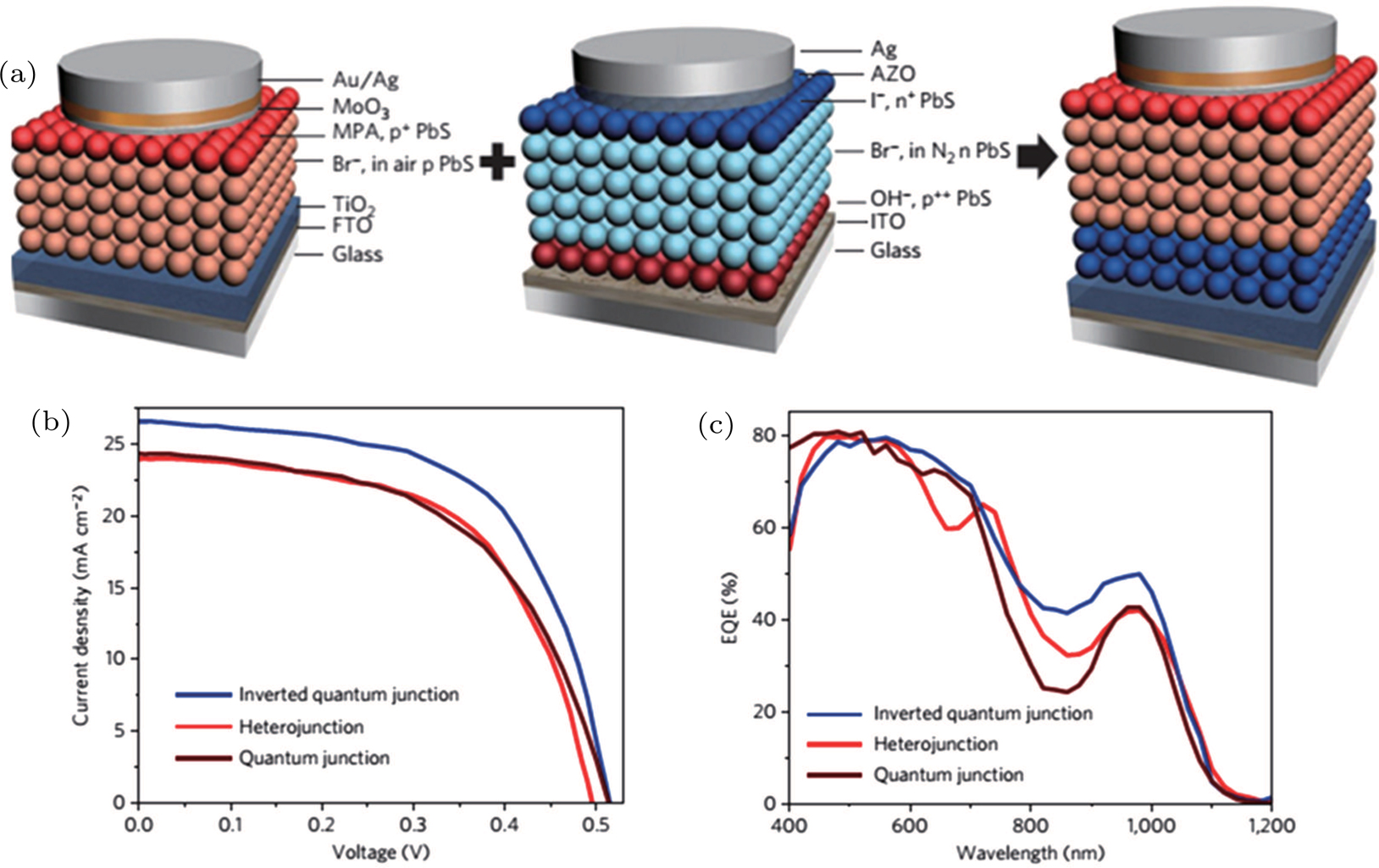 | Fig. 11. (color online) (a) The inverted quantum junction device is fabricated by combing the best p-type film in the heterojunction device and the best n-type film in the quantum junction device. (b) Current–voltage character and (c) EQE of heterojunction, quantum junction, and inverted quantum junction devices.[123] |
While notable advances in performance have been accomplished through the aforementioned efforts, further progress relies on improving the quality of the light-absorbing film itself. The inherent large surface-to-volume ratio of CQD results in high electronic trap states due to unsaturated dangling bonds on the CQDs surface, which increase the carrier recombination and thus reduce the charge extraction efficiency.[126] For this reason, a series of strategies have been developed to passivate the trap states at each step in processing, and ligand strategies are proven to be efficient paths to decrease recombination losses, such as organic–inorganic hybrid passivation, atomic passivation, and perovskite-matrix passivation. Generally, the as-prepared CQDs are passivated by long chain ligands (such as oleic acid (OA)) to control the CQD size and prevent their aggregation, but meanwhile these ligands produce insulating barriers between CQDs that militate against efficient carrier transport. Hence, much attention has been paid to the development of new ligand strategies that minimize the interparticle spacing to promote carrier transport and lower the defect density to reduce recombination loss.[17,95,123,127–129] The ligand exchange that replaces long chain ligands with shorter chain ones is an essential process to enhance charge transport via solution ligand exchange or solid-state ligand.
In earlier research, CQD solar cells mainly relied on organic ligands, such as 1,2-ethanedithiol (EDT) and mercaptopropionic acids (MPA), to passivate CQD surfaces and the corresponding device efficiencies have already reached over 5% by 2011. However, in consideration of the limited absorber thickness of MPA or EDT passivated CQD solar cells as well as their drawbacks of oxidation and thermal degradation, inorganic ligand-passivated CQD solar cells have been developed. Atomic halide ligands were one of ideal choices among various passivation schemes due to their strong passivation and good air-stability of the resulting CQD solids as well as a lower density of trapped carriers than that of CQD with organic ligands.
Tang et al. reported an atomic-ligand strategy which utilized monovalent halide anions including cetyltrimethylammonium bromide (CTAB), hexadecyltrimethylammonium chloride (HTAC), and tetrabutylammonium (TBAI) to enhance electronic transport and successfully passivated surface defects in PbS CQDs films, resulting in the device with FTO/n-TiO2/p-PbS/Au architecture with 6% PCE.[129] Recently, TBAI has already been used as an air stable exchange ligand to produce high efficiency photovoltaic devices. Chuang et al. demonstrated high-performance CQD solar cells through engineering the band alignment, in which TBAI and 1, 2-ethanedithiol (EDT) were used as the inorganic and organic ligands for solid-state ligand exchange.[95] In Fig.
 | Fig. 12. (color online) (a) Surface-bound molecules shift quantum dot energy levels (leading to p- or n-type behavior) by inducing variable surface dipoles. (b) At the interface between quantum dot layers with different surface binding groups, another dipolar shift modifies the energy-level alignment crucial to device performance.[130] |
Lan et al. demonstrated that molecular iodine can improve trap passivation in PbS CQDs and decrease the trap density, thus leading to an increased diffusion length and allowing the thickness of the CQDs active layer increased without compromising charge extraction, finally presenting a certified PCE of 9.9%.[132] Unfortunately, the highly reactive nature of molecular iodine can lead to uncontrolled fusion of PbS CQDs. In order to solve this problem, they further employed a co-solvent system to enable methylammonium iodide (MAI) to access the PbS CQDs to realize improved passivation, and the resulting device achieved a recorded PCE of 10.6%, the highest certified PCE published in the literature so far.[133]
CQD films are generally fabricated by lay-by-layer spin coating, spray coating, inkjet printing, and dip coating, all of which involve repeated quantum dot deposition, solid-state ligand exchanges, and rinse steps, resulting in low quantum dot utilization and high solvent consumption. In contrast, fabricating CQD films based on pre-exchanged CQD inks, namely, the solution-based ligand-exchange method, the CQD film can be coated in a single deposition step, which was developed by Fischer.[135,136] Ning et al. reported the first photovoltaic device based on solution-phase inorganic-ligand-exchanged n-type CQD ink, wherein methylammonium iodide (MAI) was used as ligand.[124] Recently, organohalide perovskites (CH3NH3PbX3, X = I, Br, Cl) have been used to passivate CQDs by both solid-state ligand exchange and solution ligand exchange in view of their long hole and electron diffusion lengths, broad visible absorption range, and high charge mobility, more importantly, CH3NH3PbX3 shows perfect lattice matching with PbS CQDs. Most recently, methylammonium lead triiodide perovskite (MAPbI3)-ligand passivated PbS CQD films have been prepared, followed by annealing to form perovskite thin shells on the PbS CQD surfaces, then combined with PbS-EDT CQDs to fabricate hybrid quantum junction solar cells, presenting a high PCE of 8.95%.[137] A device with the structure of ITO/ZnO/[PbX3]−/[PbX]+-PbS/EDT-PbS/Au (X = I, Cl, Br) was also fabricated, as shown in Fig. 

 | Fig. 13. (color online) (a) Suggested formation dynamics of [PbX3]−/[PbX]+ capped CQD solids. (b) HDR EQE measurement. (c) 
|
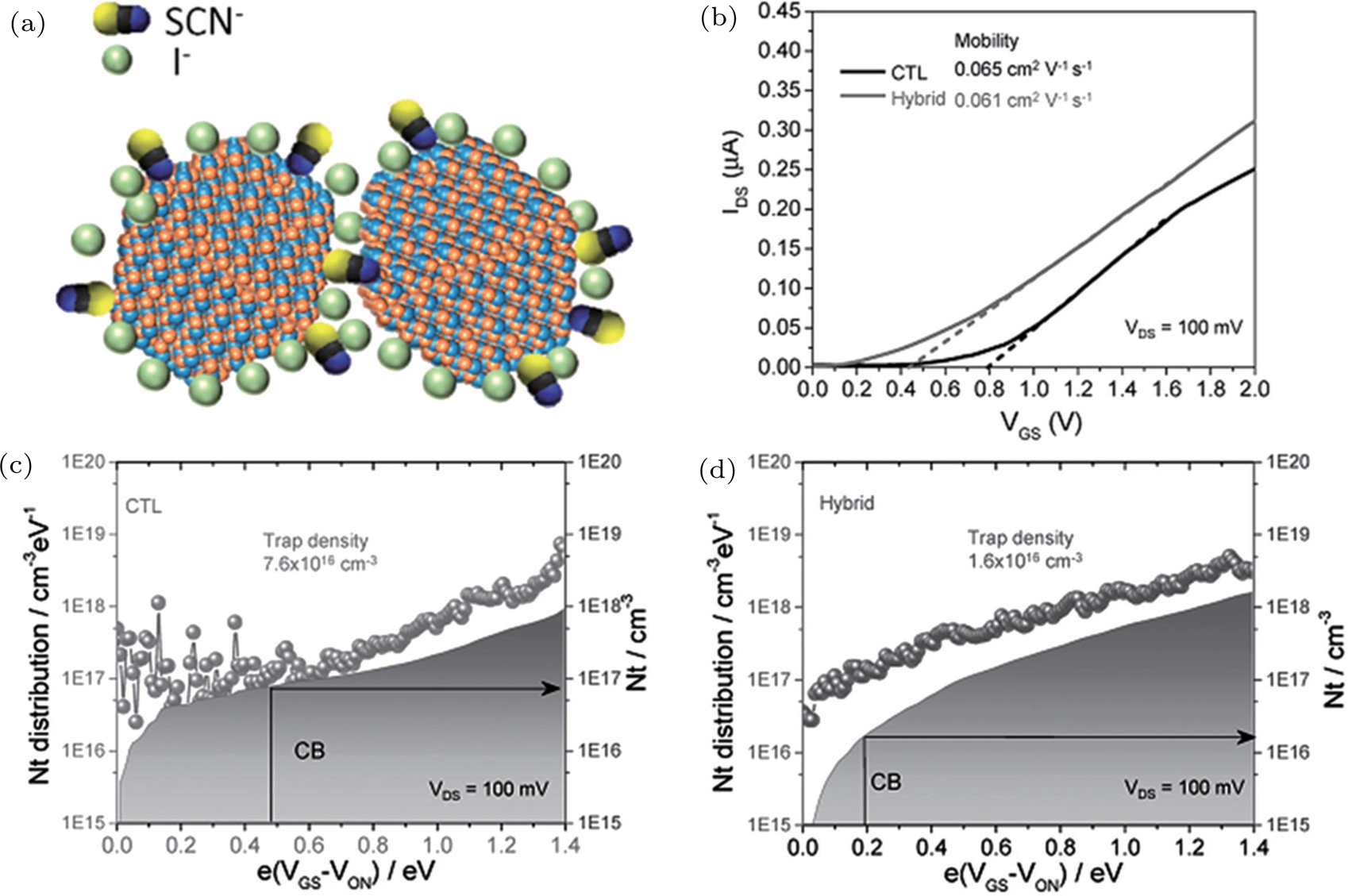 | Fig. 14. (color online) (a) Schematic diagram of PbS CQD capped by I− and SCN− and separated by SCN−. (b) Transfer characteristics of CTL (I− capped CQDs) and hybrid CQD FETs. (c) and (d) Trap state densities (Nt) of CTL and hybrid CQD FETs.[134] |
We review the recent advance in QDSCs and PbX CQD based solar cells, including high-quality CQD materials with good photoelectric properties and efficient strategies in suppressing carrier recombination, which made main contribution to the impressive enhancement of the device performance. For QDSCs, developing new QD sensitizer materials with relatively high conduction band and narrower bandgap is still crucial for enhancing 

| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] | |
| [43] | |
| [44] | |
| [45] | |
| [46] | |
| [47] | |
| [48] | |
| [49] | |
| [50] | |
| [51] | |
| [52] | |
| [53] | |
| [54] | |
| [55] | |
| [56] | |
| [57] | |
| [58] | |
| [59] | |
| [60] | |
| [61] | |
| [62] | |
| [63] | |
| [64] | |
| [65] | |
| [66] | |
| [67] | |
| [68] | |
| [69] | |
| [70] | |
| [71] | |
| [72] | |
| [73] | |
| [74] | |
| [75] | |
| [76] | |
| [77] | |
| [78] | |
| [79] | |
| [80] | |
| [81] | |
| [82] | |
| [83] | |
| [84] | |
| [85] | |
| [86] | |
| [87] | |
| [88] | |
| [89] | |
| [90] | |
| [91] | |
| [92] | |
| [93] | |
| [94] | |
| [95] | |
| [96] | |
| [97] | |
| [98] | |
| [99] | |
| [100] | |
| [101] | |
| [102] | |
| [103] | |
| [104] | |
| [105] | |
| [106] | |
| [107] | |
| [108] | |
| [109] | |
| [110] | |
| [111] | |
| [112] | |
| [113] | |
| [114] | |
| [115] | |
| [116] | |
| [117] | |
| [118] | |
| [119] | |
| [120] | |
| [121] | |
| [122] | |
| [123] | |
| [124] | |
| [125] | |
| [126] | |
| [127] | |
| [128] | |
| [129] | |
| [130] | |
| [131] | |
| [132] | |
| [133] | |
| [134] | |
| [135] | |
| [136] | |
| [137] | |
| [138] | |
| [139] | |
| [140] | |
| [141] | |
| [142] | |
| [143] |