† Corresponding author. E-mail:
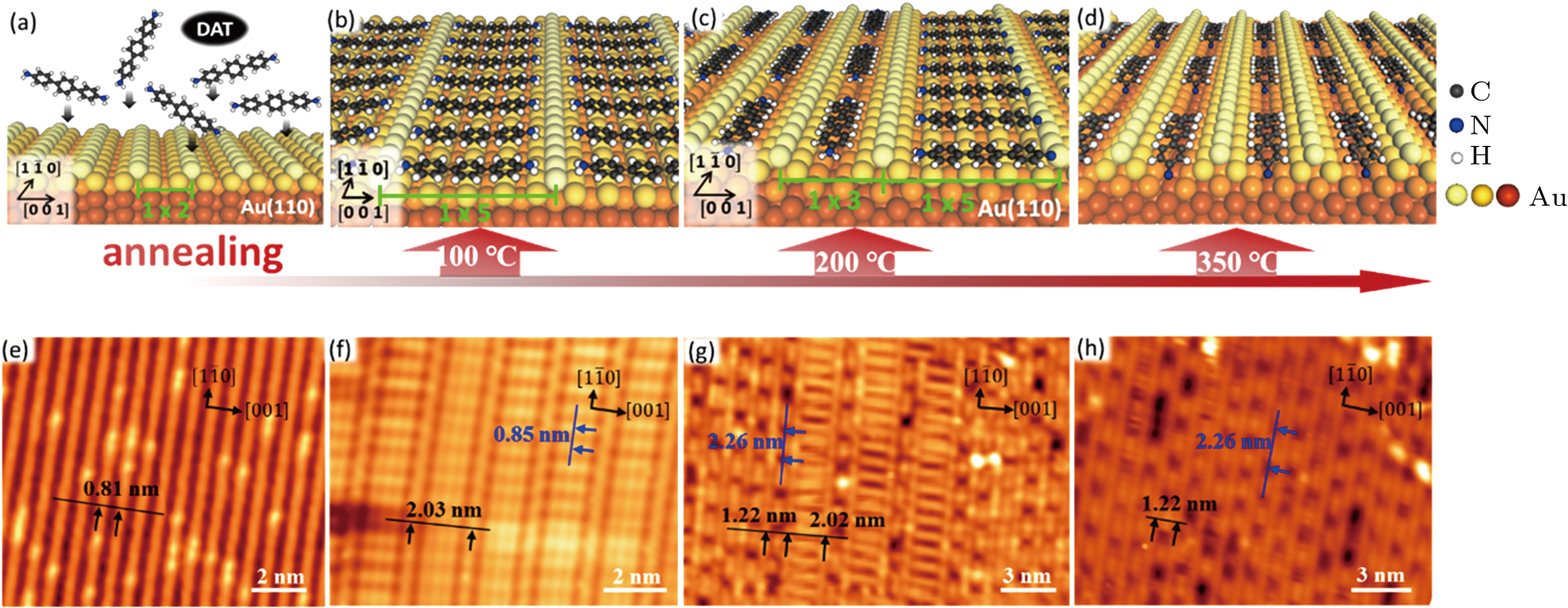
Here we report the thermo-controllable self-assembled structures of single-layer 4, 4″-diamino-p-terphenyl (DAT) molecules on Au (110), which are investigated by scanning tunneling microscopy (STM) combined with density functional theory (DFT) based calculations. With the deposition of monolayer DAT molecules on Au (110) and subsequent annealing at 100 °C, all DAT molecules adsorb on a (1 × 5) reconstructed surface with a ladder-like structure. After annealing the sample at about 200 °C, STM images show three distinct domains, including DAT molecules on a (1 × 3) reconstructed surface, dehydrogenated molecules with two hydrogen atoms detached from one amino group (−2H-DAT) on a (1 × 5) reconstructed surface and dehydrogenated molecules with four hydrogen atoms detached from two amino groups (–4H-DAT) on a (1 × 3) reconstructed surface through N–Au bonds. Furthermore, after annealing the sample to 350 °C, STM image shows only one self-assembled structure with −4H-DAT molecules on a (1 × 3) reconstructed surface. Relative STM simulations of different self-assembled structures show excellent agreements with the experimental STM images at different annealing temperatures. Further DFT calculations on the dehydrogenation process of DAT molecule prove that the dehydrogenation barrier on a (1 × 5) reconstructed surface is lower than that on (1 × 3) one, which demonstrate the experimental results that the formation temperature of a (1 × 3) reconstructed surface is higher than that of a (1 × 5) one.
The self-assembly of two-dimensional (2D) organic molecules on various substrates has been investigated extensively in the past two decades,[1–11] and has been demonstrated to be an effective way to form well-ordered and interesting structures, such as Kagome lattice,[1,2,9] Sierpinski triangle,[4,11] and so on. In contrast to nanostructures constructed through the intermolecular van der Waals force,[4,5] nanostructures formed through the coordination[1,12–14] with specific functional properties, such as tunable size,[15] and topologies,[15] have better potential application in several aspects, such as catalysis,[16] gas storage,[16] and magnetism.[17] 4, 4″-diamino-p-terphenyl (DAT) is a linear molecule with three non-planar phenyl rings and an amino group attached to each end. Because of its fascinating physical properties, DAT has been used to construct polymer light-emitting diodes by vapor deposition polymerization.[18] Notably, DAT molecule has been reported to chemically bound to the substrate through the thermo-controlled dehydrogenation of the amino group.[19,20] Furthermore, the interplay between adsorbates and Au (110) easily induces the formation of distinct surface reconstructions,[21–23] making Au (110) an ideal template to construct novel nanostructures for organic molecules,[24] especially linear molecules,[25,26] due to two growth modes, namely the end-to-end mode and the side-by-side mode.[25,27]
In this article, we construct different nanostructures of single-layer DAT molecules on Au (110) by modulating the annealing temperatures. The configurations and the formation mechanism of different structures were systematically explored by STM experiments and DFT calculations. After annealing at 100 °C, DAT molecules were found to form one ladder-like structure in a (1 × 5) surface reconstruction with their long molecular axes parallel to the [001] direction of the Au (110) substrate. While the monolayer sample was annealed at 200 °C, both the (1 × 5) and (1 × 3) surface reconstructions were formed on Au (110) with –2H-DAT molecule in (1 × 5) reconstruction and both intact DAT and –4H-DAT molecules in (1 × 3) reconstruction. Notably, after annealing at 350 °C, –4H-DAT molecules in (1 × 3) surface reconstruction covered the whole Au (110) surface. Further calculations proved that in comparison with –2H-DAT molecules in (1 × 5) surface reconstruction, –4H-DAT molecules in (1 × 3) surface reconstruction were more difficult to form.
Our experiments were accomplished in a homemade UHV-VT-STM with the base pressure better than 

DFT calculations were performed by Vienna ab-initio simulation package (VASP) code.[28] The projector augmented wave (PAW)[29] method was used to describe the interaction between the ions and electrons, and the Perdew–Burke–Ernzerhof (PBE)[30] generalized gradient approximation (GGA)[31] exchange–correlation functional was employed. The plane-wave energy cutoff is 400 eV. The molecule–substrate system was modeled within the supercell approach and contained five layers of Au atoms with adsorbed molecules on one side. The vacuum layer is about 18 Å. Two supercells with 3 × 5 and 8 × 3 of Au (110) were constructed. In structural relaxations, two bottom Au layers were fixed, while the other Au atoms and the molecule were fully relaxed until the force for each atom was less than 0.02 eV/Å. Van der Waals (vdW) interactions were included using the dispersion corrected DFT-D2 method of Grimme.[32] The nudged elastic band (NEB).[33] calculation was performed to investigate the dehydrogenation pathway for the molecules on surface.
Figure 
As shown in the high-resolution STM image (Fig.
While the monolayer sample was annealed at 200 °C, we observed the co-existence of (1 × 5) and (1 × 3) surface reconstructions with side-by-side and end-to-end molecular adsorption configurations, respectively (Fig.
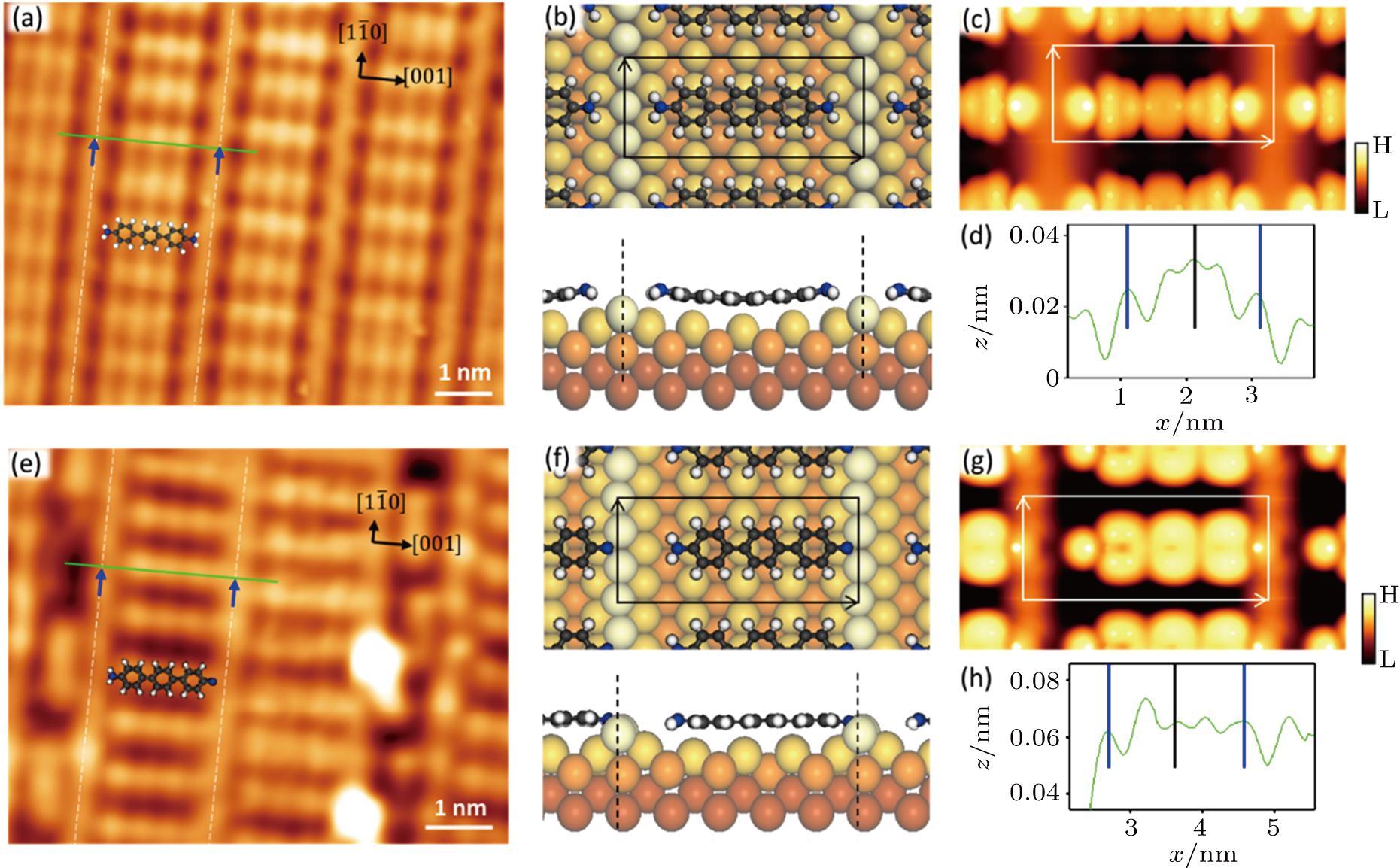
Another structure of molecules on a (1 × 3) reconstructed surface (see Fig.
In order to reveal the configuration of the molecules and how many hydrogen atoms are detached from the original ones, we carried out DFT calculations and did STM simulations. The results are shown in Figs.
In order to know the structure of the sample at a higher temperature, we annealed the sample at 350 °C, and the surface completely changed, as shown in Fig.
The calculated dehydrogenation processes of the amino groups at both ends of DAT molecules in reconstructed channels are shown in Fig.
By combining the STM experiments with DFT calculations, we systematically explored the thermo-controlled configuration transformations and formation mechanism of the self-assembled single-layer DAT molecules on Au (110) surface. At a low annealing temperature of 100 °C, DAT molecules only form one ladder-like structure in the (1 × 5) surface reconstructed channels. At a high annealing temperature of 200 °C, the amino groups of the molecules are partially dehydrogenated. The –2H-DAT and –4H-DAT molecules can be found in both the (1 × 5) and (1 × 3) surface reconstructions, respectively. At a higher annealing temperature of 350 °C, there are only –4H-DAT molecules in the (1 × 3) surface reconstructions. The investigation on these novel self-assembled structures, which is combined with surface reconstruction and dehydrogenation process, enriches our knowledge of the self-assembly behavior of organic molecules on metal surfaces.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] |