† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 51472016 and 51272015).
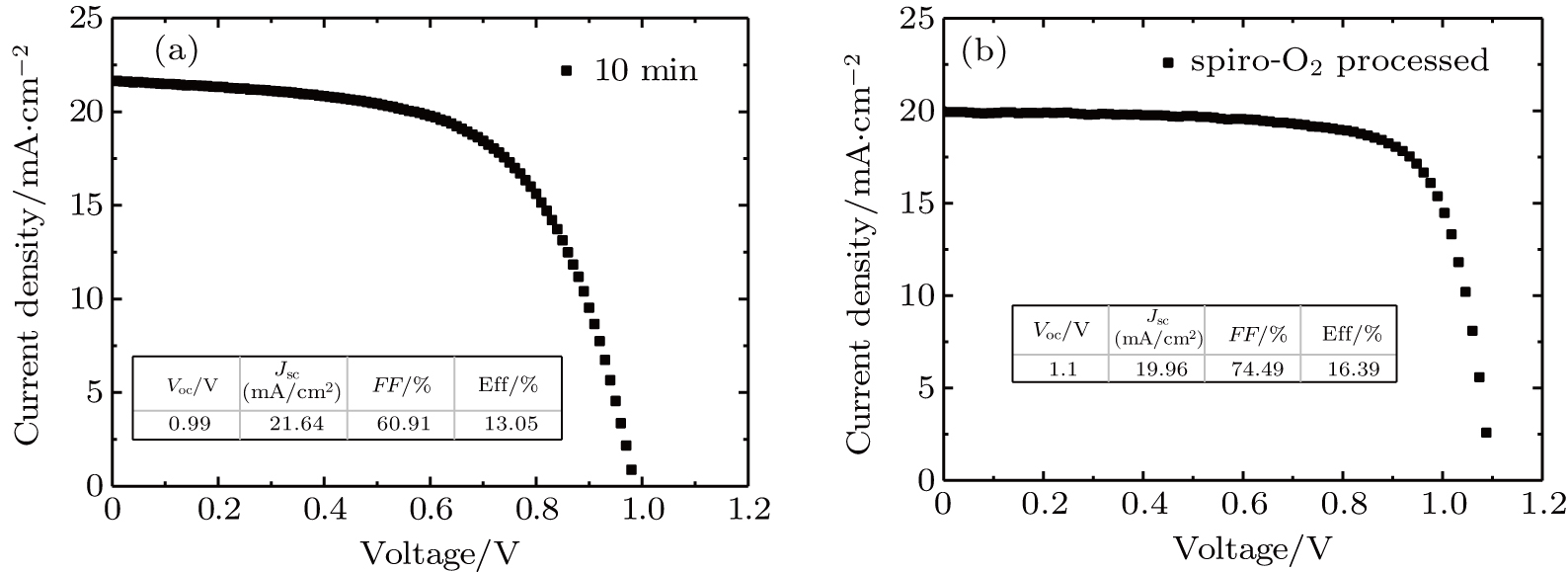
We demonstrate a simple and fast post-deposition treatment with high process compatibility on the hole transport material (HTM) Spiro-MeOTAD in vapor-assisted solution processed methylammonium lead triiodide (CH3NH3PbI3)-based solar cells. The prepared Co-doped p-type Spiro-MeOTAD films are treated by O3 at room temperature for 5 min, 10 min, and 20 min, respectively, prior to the deposition of the metal electrodes. Compared with the traditional oxidation of Spiro-MeOTAD films overnight in dry air, our fast O3 treatment of HTM at room temperature only needs just 10 min, and a relative 40.3% increment in the power conversion efficiency is observed with respect to the result of without-treated perovskite solar cells. This improvement of efficiency is mainly attributed to the obvious increase of the fill factor and short-circuit current density, despite a slight decrease in the open-circuit voltage. Ultraviolet photoelectron spectroscopy (UPS) and Hall effect measurement method are employed in our study to determine the changes of properties after O3 treatment in HTM. It is found that after the HTM is exposed to O3, its p-type doping level is enhanced. The enhancement of conductivity and Hall mobility of the film, resulting from the improvement in p-doping level of HTM, leads to better performances of perovskite solar cells. Best power conversion efficiencies (PCEs) of 13.05% and 16.39% are achieved with most properly optimized HTM via CH3NH3I vapor-assisted method and traditional single-step method respectively.
Organometal halide perovskite solar cells are dramatically emerging as one of the most competitive solar technologies and revolutionizing the field of photovoltaics.[1–4] With desirable physical properties including broadly tunable bandgaps,[5] high absorption coefficients,[6] high carrier mobilities,[7] extremely long carrier diffusion lengths,[8,9] and good crystallinity,[10,11] organometal halide perovskite materials have been demonstrated to be excellent photovoltaic materials. Since the first version of the perovskite solar cell based on the sensitizer concept in 2009,[12] the power conversion efficiencies (PCEs) of perovskite-based devices have been enhanced from 3.8%[12] to 22% or more.[13] With the continuous development of the perovskite solar cells, this photovoltaic technology will achieve economic feasibility in the near future.
The common device structure of perovskite solar cells consists of a transparent conductive substrate, a compact TiO2 hole-blocking layer, an organolead halide perovskite light-absorption layer combined with a porous scaffold, a hole transporting layer, and a metal counter electrode. Studies have mainly focused on hole-blocking layer optimization,[14] scaffold layers design,[10,15] high quality perovskite film formation methodologies,[16,17] alternative device structure,[18,19] and inherent mechanisms.[9,20,21] Hole transport material (HTM), as one of the key components in perovskite solar cells, separates photo-excited electron–hole pairs and transports the holes to the external circuit.[22] Therefore, the performance of perovskite solar cells is highly dependent on the properties of HTM. The 
In the present work, we investigate the CH3NH3PbI3-based device with a structure of glass/fluorine-doped tin oxide (FTO)/TiO2 compact layer/mesoporous TiO2layer/ CH3NH3PbI3/Co-doped p-type Spiro-MeOTAD/silver (Ag). The perovskite films are prepared via vapor-assisted solution processed deposition method.[26] The HTM films are treated in dry O3 at room temperature for 3 different lengths of time: 5 min, 10 min, and 20 min, before evaporating Ag metal contact. The control samples are completely fabricated without O3 treatment. It is observed that the performances of CH3NH3PbI3-based devices are strongly affected by the O3 treatment of the films, which leads to a substantial increase in the PCE of the device with the significant increase of the fill factor and current density. The processing conditions and corresponding results will be discussed in detail.
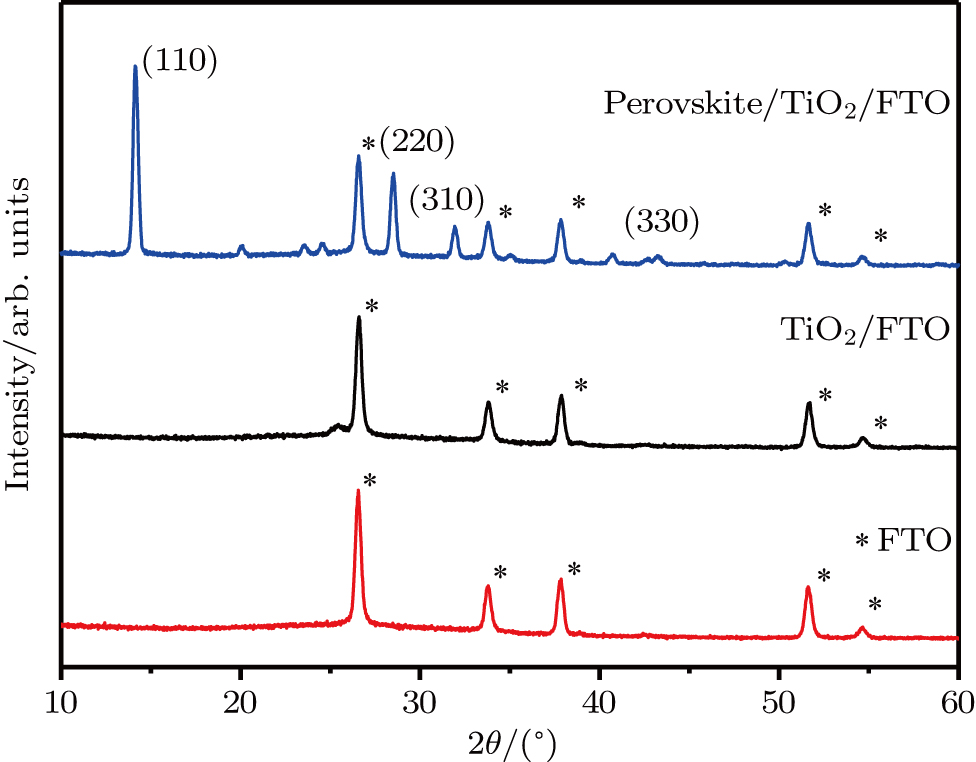
CH3NH3PbI3-based perovskite solar cells were fabricated under ambient condition. The x-ray diffraction (XRD), scanning electron microscope (SEM), UV-visible spectroscopy, ultraviolet photoelectron spectroscopy (UPS) and Hall effect measurement are used to characterize the material; I-V measurement is used to determine the photovoltaic performance of device.
The 35 mm × 35 mm square substrates, fluorine-doped tin oxide coated glasses (10 Ω/sq), were cleaned by ultrasonication in a detergent diluted washing solution, rinsed with deionized water and ethanol, dried in nitrogen, and then treated under O3/ultraviolet for 30 min.
The clean FTO glasses were coated with 0.15-M and 0.3-M titanium diisopropoxide bis (acetylacetonate) at 3000 rpm for 30 s subsequently. After being heated on a hotplate at 125 °C for 15 min, the coated substrates were annealed in an oven at 550 °C for 30 min in air. The substrates were treated in 0.05-M TiCl4 aqueous solution at 70 °C for 30 min and rinsed with deionized water, followed by being annealed at 550 °C for 20 min in air to form a compact n-type layer of TiO2 (c-TiO2).
Mesoporous TiO2 films were deposited on the substrates by spin coating TiO2 paste at 3000 rpm for 30 s, followed by being heated on a hotplate at 125 °C for 15 min and annealed at 550 °C for 30 min in air subsequently to form a layer of mesoporous TiO2 (mp-TiO2). The FTO/c-TiO2/mp-TiO2 substrates were preserved in nitrogen at 80 °C for 15 h.
The 460-mg/ml precursor of PbI2 solution was prepared by dissolving PbI2 into DMF, then heated to form clean yellow and clear solution. The PbI2 solution in DMF was spin-coated on the FTO/c-TiO2/ mp-TiO2 substrates at 5000 rpm. for 10 s, and dried at 100 °C for 15 min. CH3NH3I powder was spread out around the PbI2 coated substrates with a petri dish covering on the top, and heated at 120 °C for the desired time to form perovskite films. After cooling down, the as-prepared perovskite films were washed with isopropanol, dried and annealed at 80 °C for 30 min.
The perovskite films were coated with the HTM (Co-doped p-type Spiro-MeOTAD) by using the spin-coating method at 2000 rpm for 20 s. The spin-coated Spiro-MeOTAD films were treated with O3, respectively, for 5 min, 10 min, and 20 min as shown in Fig.
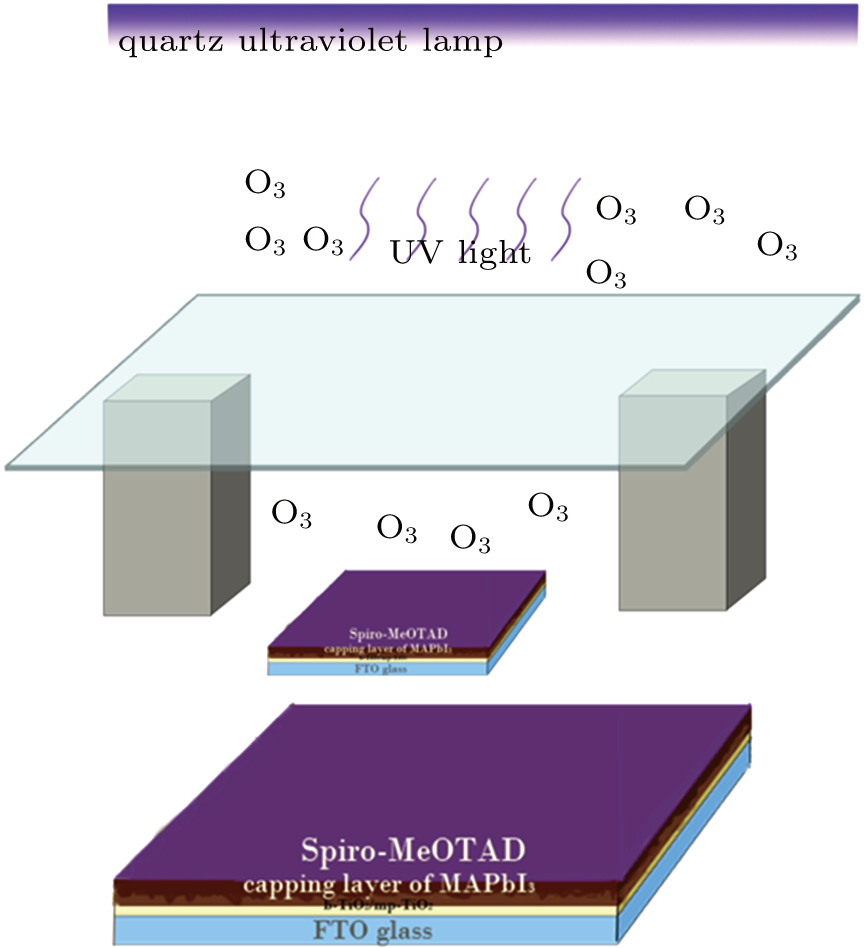 | Fig. 1. (color online) Schematic procedure for the fast O3 treatment to Spiro-MeOTAD films. Soda-lime glass excludes the direct UV rays. |
Finally, the counter electrode was deposited by thermal evaporation of silver under a vacuum of 5 × 10−5 Torr (1 Torr = 1.33322 × 102 Pa). The active area was a bit larger than 0.07 cm2 and was finally tested with a mask whose area was just 0.07 cm2.
The I–V characteristics of the devices were measured by using the Keithley 2400 source meter under the calibrated solar simulator equipped with an AM 1.5 filter at 100 mW/cm2. The I–V curves were obtained from the reverse scan at a scan rate of 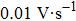
Ultraviolet photoelectron spectroscopy (UPS) analysis was conducted by using Beijing synchrotron radiation light source. The charging effects were taken into account by referring the measured spectra to the binding energy peak of Au 4f at 84 eV. The conductivity and Hall mobility were recorded on the HL5500PC Hall Effect measurement system for Co-doped p-type Spiro-MeOTAD film deposited on a 0.5 cm × 0.5 cm2 square glass substrate.
CH3NH3PbI3-based solar cells are fabricated via vapor-assisted solution process.
Figure
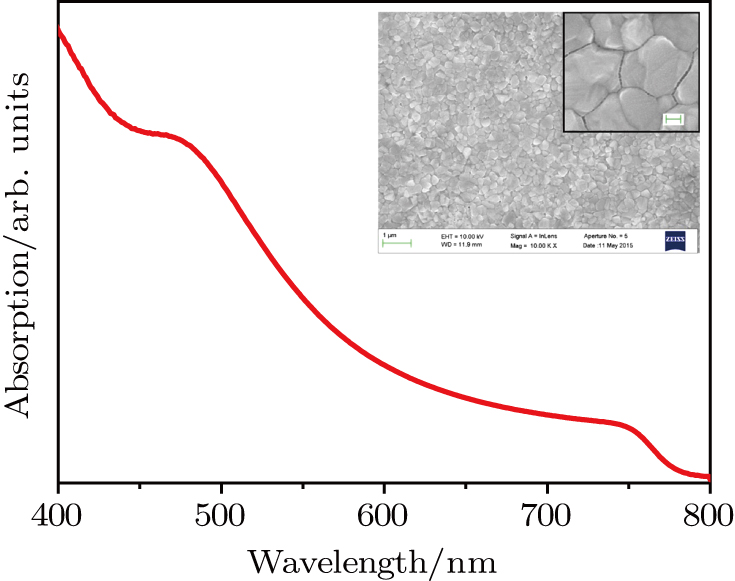 | Fig. 3. (color online) Absorption spectrum of CH3NH3PbI3 film and top-view SEM images for the sample of FTO/TiO2/CH3NH3PbI3. (Insert image with higher resolution, scale bar: 100 nm). |
The changes of the electronic properties of the as prepared Spiro-MeOTAD film and film after O3 exposure are probed by UPS with a synchrotron radiation light source. As can be seen in Fig.
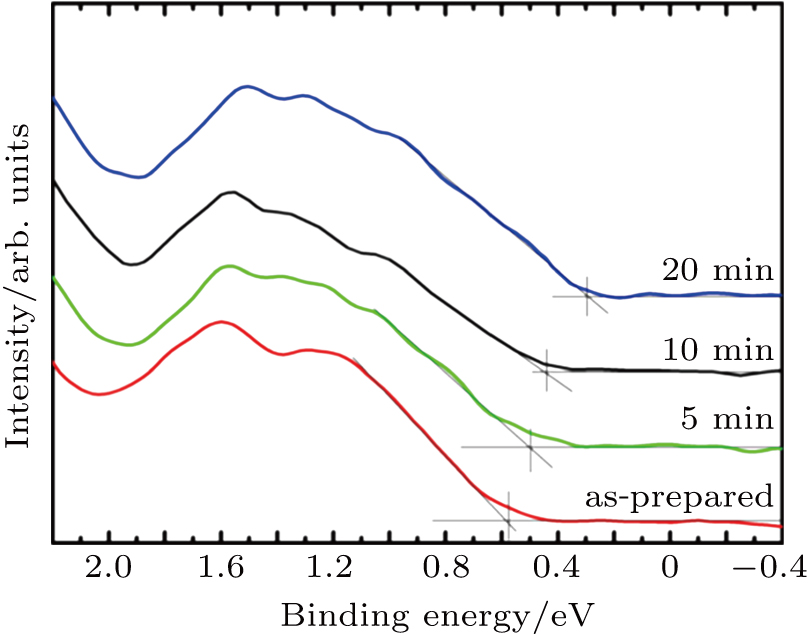 | Fig. 4. (color online) UPS corresponding to the HOMO region of the as-prepared sample and samples with different O3 treatment times. |
To record the carrier density, Hall mobility and conductivity of the Co-doped p-type Spiro-MeOTAD film, the Hall effect measurement is adopted. The sample is obtained by spinning on 0.5 cm × 0.5 cm square glass at 2000 rpm for 20 s. The thickness of the film is 500 nm. Hall mobility (μ), sheet carrier density (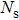
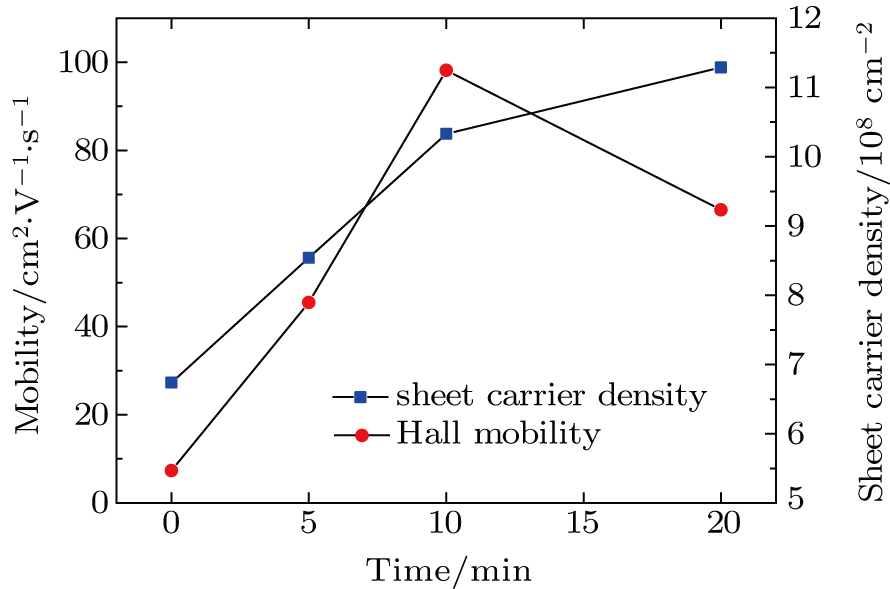 | Fig. 5. (color online) Time-dependent carrier mobility and sheet carrier density of the spin-coated Spiro-MeOTAD film on glass substrate. |
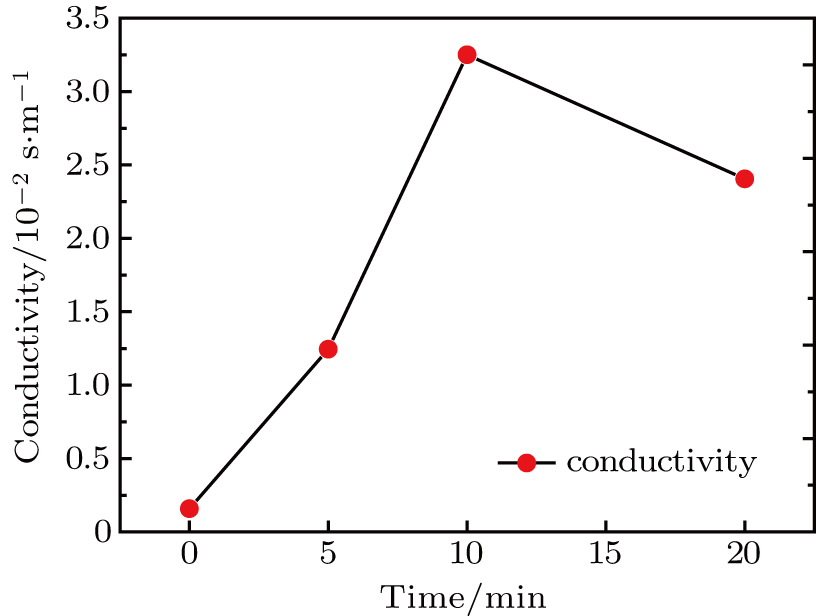 | Fig. 6. (color online) Conductivity of the spin-coated Spiro-MeOTAD film on glass substrate versus O3 treatment time. |
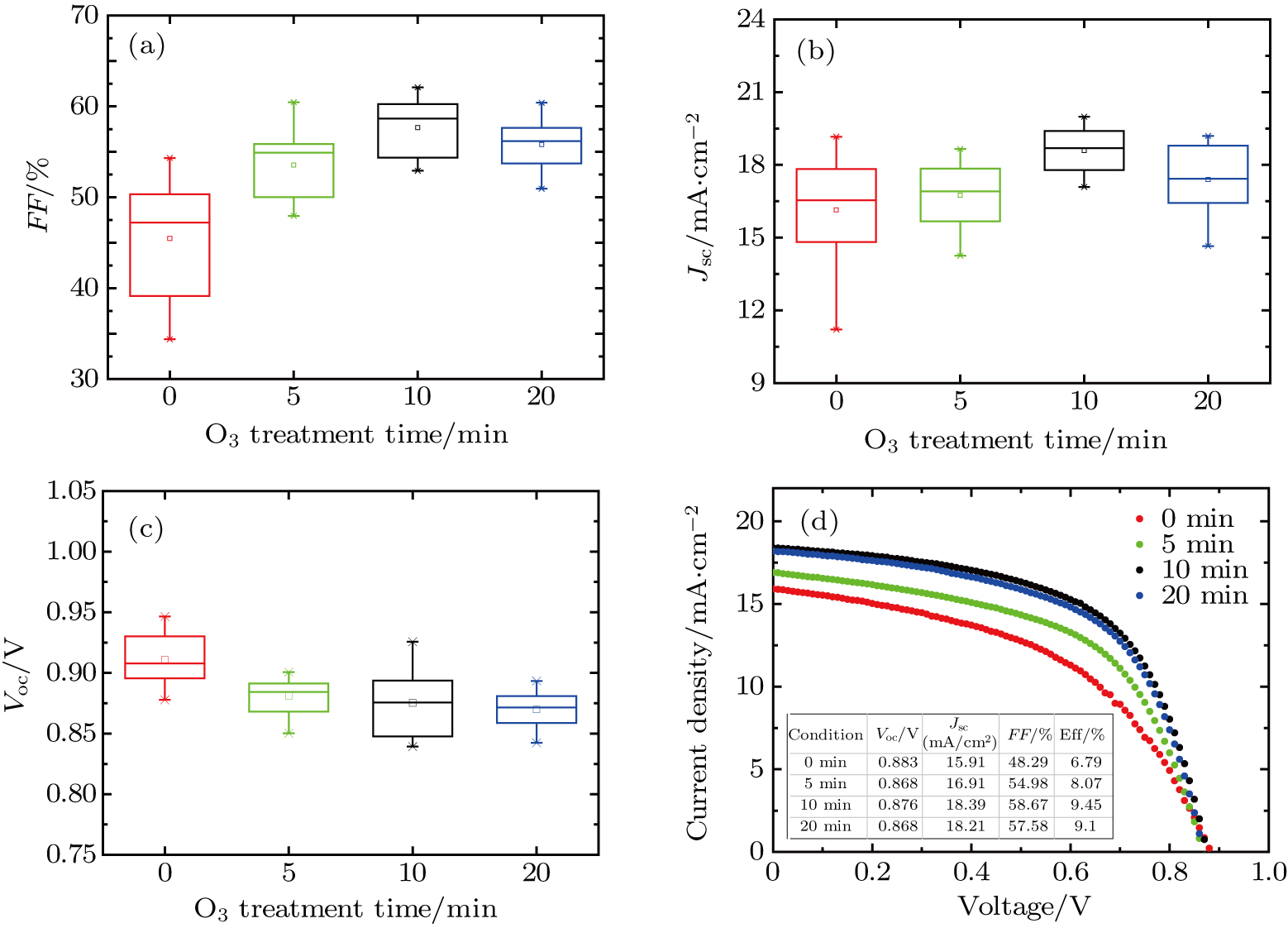
To further confirm the effect of O3 treatment, we examine the exposure-time-dependent photovoltaic performance of CH3NH3PbI3-based solar cells employing Spiro-MeOTAD as HTM with different exposure times. J–V curves of average performance devices as representative devices are shown in Fig. 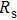
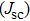
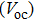
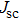
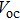
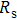
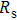
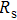
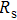
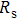
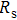
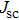
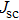
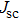
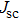
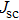
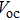
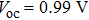
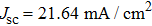
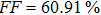
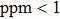
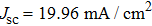
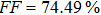
In this work, O3 is adopted to treat co-doped p-type Spiro-MeOTAD film at room temperature before evaporating Ag metal contact when fabricating CH3NH3PbI3-based perovskite solar cells under ambient condition. The O3 treatment procedure enhances p-doping level in this hole transport material which is evidenced by the HOMO level approaching Fermi level after O3 treatment. This leads to the increase in the carrier density, improvement in the carrier mobility and optimization in conductivity of Spiro-MeOTAD, which is confirmed by Hall effect measurement. Results suggest that the enhancements in the conductivity and carrier mobility lead to better device performance and 10-min treatment turned out to be favorable for the optimization, thus achieving a 40.3% increment in the PCEs: an averaged enhancement is 9.4%. The values of η reach, respectively, 13.05% in ambient air via CH3NH3I vapor-assisted method and 16.39% in glove box via traditional single-step method, indicating that the device achieves the best performance. This presents a short-time and effective process to improve the efficiency of organometal halide perovskite solar cells
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] |