† Corresponding author. E-mail:
Project supported by the Scientific Research Starting Fund for Doctor of Mudanjiang Normal University, China (Grant No. MNUB201508), the Youth Science Foundation of Heilongjiang Province, China (Grant No. QC2016007), and the National Natural Science Foundation of China (Grant No. 61604065).
Carbon nanodots (CDs) with visible absorption band and TiO2 are integrated to enhance the photosensitivity of TiO2. The CD/TiO2 nanocomposites show obvious CD-coverage-dependent photocatalytic performance. The CD/TiO2 nanocomposites with moderate CD coverge exhibit the highest photocatalytic activity after being irradiated with visible light, which is more excellent than that of TiO2. Too little CD coverage could result in poor visible light absorption, which limits the photocatalytic performance of CD/TiO2 nanocomposites. While, too much CD coverage weakens the photocatalytic activity of CD/TiO2 nanocomposites by restraining the extraction of conduction band electrons within TiO2 to generate active oxygen radicals and the electron transfer (ET) process from CDs to TiO2. These results indicate that rational regulation of CD coverage and the realization of efficient ET process are important means to optimize the photocatalytic performance of CD/TiO2 nanocomposites.
Recently, great attention has been paid to solar energy conversion, due to the problems of environment and energy.[1–3] Semiconductor TiO2 has been widely used in the field of photocatalysis, which is attributed to its unique features such as low cost, good stability, and environmental friendliness.[4–6] While, the large bandgap of TiO2 (∼ 3.2 eV) severely restricts its practical application progress of photocatalysis,[4–7] considering the fact that TiO2 responds only to the ultraviolet region (lower than 5% of the full solar spectrum).[8] Therefore, it is important to expand the photosensitive spectrum range of TiO2 to visible region. In recent years, great efforts have been made to sensitize the narrow bandgap quantum dots (QDs) and organic dyes on TiO2 for improving the performance of TiO2 photocatalysts.[9–12] However, most of QDs with superior performances contain heavy metal elements and the photochemical stability of organic dyes is generally unsatisfactory.[13,14]
Carbon nanomaterials, which include carbon nanotubes, fullerene, graphene, carbon nanodots (CDs), and so on, are expected to replace the use of QDs and organic dyes in bioimaging, optoelectronics, photovoltaics and photocatalysis.[14–19] Especially, CDs have received significant attention due to their low toxicity, biocompatibility, good stability and water solubility.[20–24] Moreover, CDs could be easily coupled to TiO2 by many methods.[25–28] Recently, a lot of efforts have been made to employ the CD/TiO2 nanocomposites in photocatalysis. The photocatalytic performances of CD/TiO2 nanocomposites were pretty excellent under visible light in comparison with TiO2.[25–33] For example, Yu et al. prepared CD/TiO2 nanocomposites with a simple hydrothermal process and reported that the photocatalytic H2 evolution rate of the CD/TiO2 nanocomposites was obviously improved compared with that of TiO2 after being irradiated with visible light, due to the sensitization effect of CDs.[25] Ming et al. designed a new CD/TiO2 hybrid photocatalyst by integrating CDs with TiO2 through an easy hydrothermal method, which exhibited an excellent photocatalytic performance under visible light.[32]
It is known that the exposure of active sites on TiO2 surface is essential for excellent photocatalytic activity.[31] In addition, the sensitization of CDs on the surface of TiO2 could expand the absorption band of TiO2 photocatalysts to visible region and enhance the interfacial charge separation process, which can improve the photocatalytic performance of CD/TiO2 nanocomposites.[28,29,31,34] While, the sensitized CDs could also occupy some active sites on TiO2 surface, which play an important role in photocatalysis.[31] Therefore, rational regulation of CD coverage has a great influence on the photocatalytic activity of CD/TiO2 nanocomposites, which is proposed as an urgent consideration for the design of CD/TiO2 nanostructure photocatalysts. However, the influencing mechanisms of CD coverage are less explored and understood in CD/TiO2 nanocomposites.
In this work, we prepare CDs with visible absorption band locating in the range from 400 to 600 nm by the microwave synthesis method and design the CD/TiO2 nanocomposites with different CD coverages. The CD/TiO2 nanocomposites exhibit expanded photosensitive spectrum range compared with TiO2 and the photocatalytic performance of CD/TiO2 nanocomposites is measured by photodegradation experiment of Rhodamine B (RhB) dye molecules after being irradiated with visible light. The experimental results are well reproducible. CD-coverage-dependent photocatalytic performance of the CD/TiO2 nanocomposites is studied and possible mechanisms are analyzed through time-resolved photoluminescence (PL) spectrum.
TiO2 nanoparticles (P25, average size 25 nm, 20% rutile and 80% anatase phase) were purchased from Degussa. Urea (99.5%), citric acid (99.8%), and RhB (99%) were all purchased from Aladdin.
The CDs were synthesized with the microwave synthesis method.[23,35] The urea of 4 g and citric acid of 2 g were loaded into a beaker. Some deionized water (∼ 15 mL) was added and then a clear solution was formed. For the formation of CDs, the obtained clear solution was then heated for about 10 min in a 750 W microwave oven. In the process of heating, the reaction mixture gradually evolves from clear solution to dark-brown solid. After that, the dark-brown solid CDs were dispersed in deionized water and purified five times with a centrifugal speed of 10000 rpm to remove clustered CDs.
The CD/TiO2 nanocomposites with different CD coverages were prepared by changing the mixing mass ratio of CDs and P25 TiO2 from 1:10, 1:50, 1:200, 1:500 to 1:1000. Specifically, they were that 2, 0.4, 0.1, 0.04, and 0.02 mL of CD solutions in deionized water (5 mg/mL) were simply dispersed in 20 mL of TiO2 solution in deionized water (5 mg/mL). The mixed solutions were magnetically stirred for about 24 h to complete the adsorption process of CDs on TiO2 surface. After that, the reaction products were dispersed in some deionized water and then purifyed to eliminate unbonded CDs several times until no fluorescence was detected in the supernatant with the centrifugal speed of 4000 rpm. The obtained CD/TiO2 nanocomposites were dried at a temperature of 80 °C in a vacuum oven.
The photodegradation rates of RhB dye molecules under visible light were measured to evaluate the photocatalytic performances of CD/TiO2 nanocomposites with different CD coverages. The CDs, P25 TiO2 and CD/TiO2 nanocomposite solutions (5 mg/mL) were prepared in deionized water. Each (100 μL) of these solutions, 100 μL of RhB solution in deionized water (100 mg/L) and some deionized water (2.8 mL) were mixed in a 1 cm×1 cm quartz cuvette. After that, the obtained mixed solutions were kept in darkroom with constant stirring to achieve the balance between adsorption and desorption of RhB dye molecules. After being kept in darkroom for 2 h, the mixed solutions were irradiated under visible light (λ > 420 nm) from a solar simulator (Zolix SS150) equipped with a 420 nm cut-off filter, during which magnetic stirring was constantly kept. The absorption spectra of RhB dye molecules were in situ measured once every 10 min with the USB4000 spectrometer. The reference light for measuring the absorption spectra of RhB dye molecules was from a HL-2000 light source (Ocean Optics) and imported by a QP82-SMA-BX fiber (Ocean Optics). The transmission light was then exported by a QP1000-2-SR fiber (Ocean Optics) and collected by USB4000 spectrometer. 510 nm cut-off filter was used to avoid exciting the CDs by reference light.
The absorption spectrum of CD aqueous solution was measured with Shimadzu UV-3101PC UV-Vis-NIR spectrophotometer. The PL spectra of CDs and CD/TiO2 nanocomposites in deionized water were recorded in a 10 mm×1 mm quartz cuvette by Hitachi F-7000 spectrophotometer. The diffuse reflectance spectra of CD/TiO2 nanocomposites with different CD coverages were also measured by Hitachi F-7000 spectrophotometer with an integrating sphere scanning from 200 to 700 nm with BaSO4 used as the reference. The morphology of CD/TiO2 nanocomposites was recorded on TECNAI G2 transmission electron microscope (TEM). The time-resolved PL spectra were recorded on LifeSpec-II PL lifetime spectrometer (Edinburgh Instruments) with a diode laser (with a picosecond pulse, laser wavelength of 405 nm, pulse width of 74.5 ps) used as an excitation source. The detector was Hamamatsu H5773-04 photomuitplier (response time: 250 ps) and the instrument response function was about 440 ps.
The CDs are prepared with microwave synthesis method.[23,35] As seen from Fig.
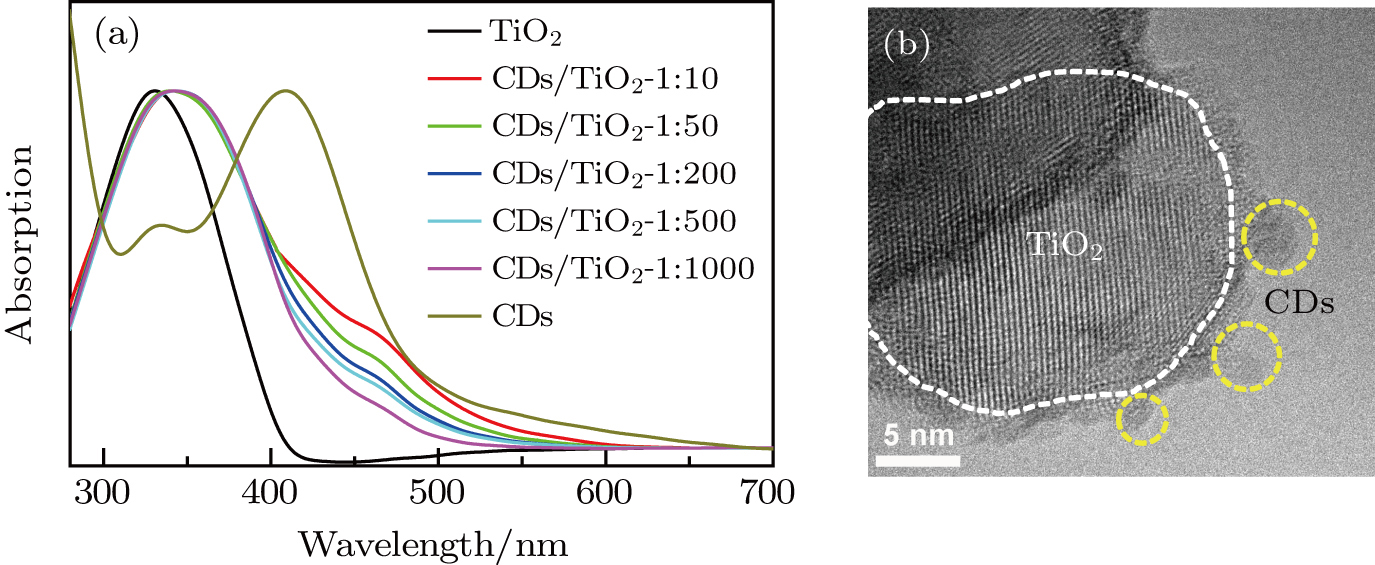 | Fig. 1. (color online) (a) Absorption spectra (normalized) of the CDs, TiO2, and CD/TiO2 nanocomposites with different CD coverages, (b) TEM image of the CD/TiO2 nanocomposites. |
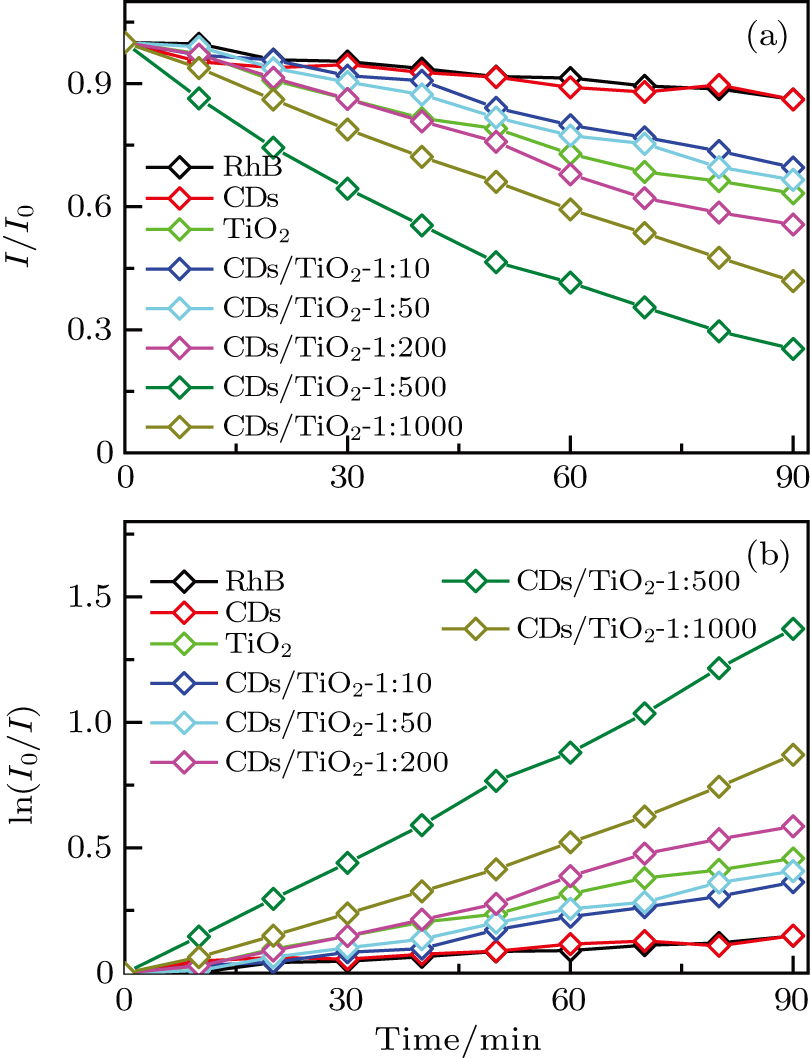
The photodegradation rates of RhB dye molecules after being irradiated with visible light (λ > 420 nm) are measured to evaluate the photocatalytic performance of CD/TiO2 nanocomposites. In the photodegradation process, it is generally considered that photogenerated electrons and holes within CD/TiO2 nanocomposites under visible light are extracted by O2 and OH− in water to generate active oxygen (O2·) and hydroxyl (·OH) radicals, respectively, thereby degrading the RhB dye molecules.[28,29,32–34] Figure
Figure
Charge separation in CD/TiO2 nanocomposites under visible light is considered to be the crucial process to improve photocatalytic performance of TiO2, because it is a key step for the generation of O2· and ·OH to photodegrade organic dye molecules.[25,28–31,34] The dynamic behaviors of electron transfer (ET) from CDs to TiO2 give rise to primary photophysical processes to achieve charge separation between them after being irradiated with visible light.[28,38] Thus, there is a strong need to study the ET dynamic processes between CDs and TiO2 for better understanding the effect of CD coverage on the photocatalytic activity of CD/TiO2 nanocomposites. Figure
 |
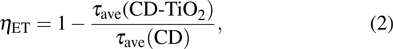 |
where τave(CD-TiO2) and τave(CD) are the average PL lifetimes of CD/TiO2 nanocomposites and CDs in deionized water, respectively. Two- or three-order exponential decay kinetics is used to fit PL decay spectra of CDs and CD/TiO2 nanocomposites and then the kET and ηET are calculated as shown in Table
| Table 1. Fitting results for the PL dynamic behaviors of CDs and CD/TiO2 nanocomposites in deionized water and calculation values of kET and ηET. |
Three feasible consumption ways for conduction band electrons of TiO2 exist in the CD/TiO2 nanocomposites, which are electron extraction by O2 in water, electron recombination back to the CDs, and electron capture into TiO2 defect states[28,29] as seen from Fig.
By the way, the electron recombination back to the CDs and electron capture into TiO2 defect states are inferred to be less effective than electron extraction by O2 in water, which is important for efficient photocatalytic activity. Because the effective electron recombination back to the CDs and electron capture into TiO2 defect states could also consume conduction band electrons of TiO2 injected from photoexcited CDs and then promote the ET from CDs to TiO2, which cannot cause the PL decay of the CD/TiO2 nanocomposites with low CD coverage (mass ratio: 1:1000) to be much shorter than that of CD/TiO2 nanocomposites with high CD coverage (mass ratio: 1:10). This accords well with the fact that the PL quenching (Fig.
In this work, we expand the photosensitive spectrum range of TiO2 to visible region by the sensitization of CDs. The CD/TiO2 nanocomposites show obvious CD-coverage-dependent photocatalytic performance under visible light. The CD/TiO2 nanocomposites with moderate CD coverage exhibit the best photocatalytic performance under visible light, which is more excellent than that of pure TiO2. The PL dynamic study demonstrates that too little CD coverage results in poor visible light absorption, which limits the photocatalytic activity of the CD/TiO2 nanocomposites, while too much CD coverage could restrain the extraction of conduction band electrons of TiO2 injected from photoexcited CDs to generate O2· and ET process from CDs to TiO2 by reducing the surface active sites of TiO2, thus weakening the photocatalytic activity of CD/TiO2 nanocomposites. These results indicate that rational regulation of CD coverage and the realization of efficient ET process are important means to optimize the photocatalytic performance of CD/TiO2 nanocomposites. We think that a clear understanding of the CD-coverage-dependent photocatalytic activity is significant for further designing the excellent CD/TiO2 nanostructure photocatalysts.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] | |
| [39] | |
| [40] | |
| [41] | |
| [42] |