† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 11574276, 51302249, and 51503185) and the Doctoral Fund of the Ministry of Education of China (Grant No. 20114101110003).
Solid solutions of In2(1−x)(HfMg)xMo3O12 are synthesized by solid state reaction with the aim to reduce the phase transition temperature of In2Mo3O12 and improve its thermal expansion property. The effects of (HfMg)6+ incorporation on the phase transition and thermal expansion are investigated. It is shown that the monoclinic-to-orthorhombic phase transition temperature obviously decreases and the coefficient of thermal expansion (CTE) of the orthorhombic becomes less negative and approaches to zero with increasing the content of (HfMg)6+. A near zero thermal expansion covering the case at room temperature (RT) is achieved for the solid solutions with x ≥ 0.85, implying potential applications of this material in many fields.
The negative thermal expansion (NET) phenomena in some materials have received considerable attention in the past few years.[1–7] NTE materials have the promising potential applications in tailoring the coefficient of thermal expansion (CTE) of materials to reduce thermal stress.[3,8–10] However, a series of NTE materials such as BiNiO3, LaCu3Fe4O12, and Mn3NX needs extreme synthesis conditions, like high pressure/temperature or exclusion of oxygen,[5,11–13] which restricts their applications in industry. Recently, some ceramics consisting of an open-framework with low thermal expansion or NTE[14–21] and low synthesis requirements, such as A2O(A = Ag/Cu),[18] ABO3 (A = Bi/Pb, B = Ni/Ti),[5] AMO5 (A = Ta/Nb, M = V/P),[15,16] AM2O7 (A = Zr/Hf, M = V/P),[17] AM2O8 (A = Zr/Hf, M = Mo/W)[1] and A2M3O12 (A is transition metal or rare earth, M is Mo or W) have been extensively investigated. The A2M3O12 family is extremely attractive due to its chemical flexibility within the orthorhombic Pbcn (No. 60) space group, and it possesses a variation of linear CTE from low positive to large negative values.[22–32] This orthorhombic feature of A2M3O12 makes it possible to be synthesized in solid solution state with controllable CTEs. Nevertheless, its heavy hygroscopicity[25–28] and abrupt change in thermal expansion property caused by phase transition from monoclinic to orthorhombic with increasing temperature[29–32] are still serious obstacles for practical applications of the A2M3O12 family.
In2Mo3O12 is a typical material of the A2M3O12 family with the monoclinic-to-orthorhombic phase transition temperature of about 608 K to 610 K.[31] Miller et al.[30] reported an In(HfMg)0.5Mo3O12 material and its monoclinic-to-orthorhombic structure transition occurring around 430 K. It means that the thermal expansion properties of In2Mo3O12 and In(HfMg)0.5Mo3O12 will be abruptly changed from giant positive to negative or near zero around their corresponding phase transition temperatures. The abrupt changes in thermal expansion deteriorate their mechanical properties and induce cracking, thus seriously hindering the applications of these materials. In order to reduce the phase transition temperature of In2Mo3O12, Cr3+ was reported to be introduced into In2Mo3O12,[32] but the phase transition temperature is still in a range of 523 K–673 K, which is much higher than room temperature (RT). It is because the phase transition (monoclinic-to-orthorhombic) temperature region of Cr2Mo3O12 is as high as 623 K–673 K.[29] Reducing the phase transition temperature of In2Mo3O12 to below room temperature (RT) and improving its thermal expansion property are still very attractive challenges.
In order to reduce the phase transition temperature of In2Mo3O12, the most effective method is to partially replace In3+ with a larger trivalent ion such as Y3+ or Yb3+. However, this may introduce crystal water at the same time such as in the cases of Yb2−xAlxMo3O12[33] and Fe2−xYxMo3O12.[27] Crystal water deteriorates the thermal expansion properties of materials because the releasing and admission of crystal water lead to serious expanding and contracting of the lattice during temperature circling. Besides, HfMgMo3O12 was shown to adopt an orthorhombic structure down to very low temperature.[34] It is expected that simultaneous doping with Hf4+ and Mg2+ into In2Mo3O12 can not only reduce the phase transition temperature but also avoid introducing crystal water. At the same time, charge balance in the formula is maintained. In this paper, solid solutions of In2(1−x)(HfMg)xMo3O12 are synthesized by solid state reaction in order to reduce the phase transition temperature of In2Mo3O12 and improve its thermal expansion property. It is found that the phase transition temperature decreases as the amount of (HfMg)6+ is increased and it is lower than RT when x ≥ 0.85. With further increasing the amount of (HfMg)6+, the near zero thermal expansion covering the case at RT is achieved so that the application scope of NTE materials can be further expanded.
The starting materials of In2O3, MgO, HfO2, and MoO3 were mixed according to the mole ratios of the destination of materials of In2(1−x)(HfMg)xMo3O12 with x = 0.0, 0.1, 0.3, 0.5, 0.7, 0.75, 0.8, and 1.0. The mixtures were ground for 2 h in an agate mortar followed by sintering at 1073 K for 8 h in a furnace.
XRD measurements were carried out with a Bruker D8 Advance x-ray diffractometer. Raman spectra were recorded with a Renishaw inVia Raman spectrometer with 532-nm laser wavelength excitation. The differential scanning calorimeter (DSC) curves were obtained by the simultaneous thermal analyzer of Netzsch STA 449F3 in a temperature range of 298 K–673 K, under the 50 mL/min nitrogen (N2) purge, with a heating rate of 10 K/min. The linear CTE was measured with a dilatometer of Linseis DIL L76 in an atmosphere environment and with a heating rate of 5 K/min.
Figure
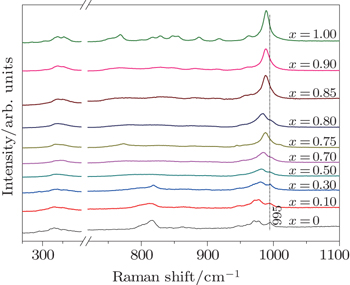
According to previous reports, the Raman spectra are sensitive to the monoclinic-to-orthorhombic phase transition.[27,35,36] In order to confirm the phase transition after the introduction of (HfMg)6+, Raman spectra of the solid solutions of In2(1−x)(HfMg)xMo3O12 are measured and shown in Fig.
Figure
 | Fig. 3. (a) The DSC of the solid solutions of In2(1−x)(HfMg)xMo3O12 and (b) linear fit and interpolation of the phase transition temperature vis (HfMg)6+ content. |
The progressively weakening and shifting to lower temperature of the endothermic peak indicate that the monoclinic-to-orthorhombic phase transition requires less energy with increasing the content of (HfMg)6+, the physics behind which could be understood by the difference in electronegativity between (HfMg)6+ and In3+. The electronegativity values of Hf3+ (1.30) and Mg2+ (1.31) are smaller than that of In3+ (1.78). With increasing the content of (HfMg)6+, the bridging oxygen connected with Hf4+ or Mg2+ gains more negative partial charge, which enhances the oxygen-oxygen repulsion force and makes the monoclinic-to-orthorhombic structure transition easier. When the sample with a higher content of (HfMg)6+ is heated, its phase transition requires a lower temperature and less heat, resulting in the weakening and shifting of the endothermic peak.
Figure
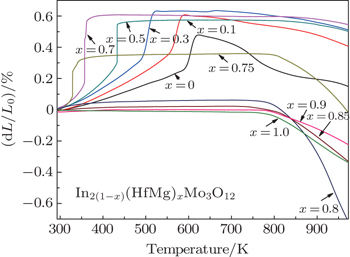 | Fig. 4. Variations of relative length of the solid solutions of In2(1−x)(HfMg)xMo3O12 with temperature. |
The mechanisms for the near-zero CTE can be understood by the cation size effect of the six-coordinated In3+, Hf4+, and Mg2+ in the center of octahedron. As is well known, the NTE increases with a larger A3+ cation size for the A2M3O12 family in the orthorhombic structure. The cation radius of In3+ is 0.080 nm while the average size of (HfMg)6+ is 0.0715 nm. The In3+ cation size plays a decisive role for the NTE in In2Mo3O12 above 623 K. However, with increasing the substitution amount of (HfMg)6+ for In3+, the NTE is reduced and even down to near-zero CTE due to the smaller average cation size. On the other hand, the thermal expansion of the In(HfMg)0.5M3O12 is anisotropic,[30] the a- and c-axis contract while the b-axis expands on heating. The two factors result in the average thermal expansion of the cell volume approaching to zero.
Solid solutions of In2(1−x)(HfMg)xMo3O12 are synthesized and the effects of (HfMg)6+ incorporation on the phase transition and thermal expansion are investigated. It is shown that the monoclinic-to-orthorhombic phase transition temperature obviously decreases with increasing the content of (HfMg)6+. The CTE of the orthorhombic structure becomes less negative and approaches to zero with increasing the content of (HfMg)6+. A near zero thermal expansion over the range from RT to 773 K is achieved for the solid solutions with x ≥ 0.8. The In2(1−x)(HfMg)xMo3O12 solid solutions with near-zero thermal expansion property covering the case at RT hold great potential applications in many fields.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 | |
| 34 | |
| 35 | |
| 36 |