† Corresponding author. E-mail:
‡ Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 51332005, 51372066, 51172060, 51202055, and 21103056).
SBA-15 (mesoporous SiO2) is used to stabilize and transfer F− in the NH4BF4 CVD reaction for the first time, and a large-scale crystalline h-BN phase can be prepared. We successfully fabricate hollow h-BN capsules with collapsed surfaces in our designed NH4BF4 CVD system. Optimum temperature conditions are obtained, and a detailed formation mechanism is further proposed. The successful SBA-15-assisted NH4BF4 CVD route is of importance and enriches the engineering technology in the h-BN single-source CVD reaction.
Chemical vapor deposition (CVD) technology is of great importance to fabricate hexagonal boron nitride (h-BN) micro-/nano-materials.[1–5] The traditional plasma-enhanced and thermal CVD technologies are still widely applied to h-BN growth,[6,7] and the most commonly used precursors include multi-source precursors with ammonium (NH3), such as NH3–boron halide (BX3, X = F, Cl, or I), NH3–boron (BxHy), NH3–organoborane (RzBxHy, R = C, F, Cl, or I), NH3–organoborate [B(OR)3, R = CnH2n + 1], etc;[2,8–10] and single-source ones, i.e., some organic or inorganic molecules which contain boron and nitrogen components, for instance, borazine (B3N3H6), chloroborazine (B3N3H6 − xClx), ammonia borane (BNH6), and so on.[5,11–13] Generally speaking, almost all of the multi-source CVD routes possess considerable complexity, and the final h-BN properties depend strongly on the vapor phase composition, the controlled target temperature, and the selective substrate in the reaction system.[5,9–13] In contrast, the single-source precursors can not only make the synthetic route much easier, but also improve the h-BN product.[5,11–13] Actually, both the frequently used single- and multi-source precursor materials are disadvantageous to the controllable CVD routes and the final h-BN micro-/nano-materials because of their deucedly-instable, highly-flammable/explosive, extremely-poisonous, and carbon-contaminated properties.[5,11–13] Furthermore, the above-mentioned single-source materials and their synthetics have been quite costly to date. Therefore, a stable, hazard-free, and carbon-free single-source precursor is highly desirable for the h-BN CVD system, and designing its feasible CVD route is also of great significance for the BN growth technology.
As a commonly-used chemical, ammonium tetrafluoroborate (NH4BF4), which is a molecule containing boron and nitrogen, is highly-stable in the ambient atmosphere and very cheap on the market. As reported by Yang et al., a reaction was carried out in an autoclave in the temperature range of 400–600 °C via NH4BF4 pyrolysis in metallic Na flux,[14] suggesting that NH4BF4 would be able to be used as a single-source precursor in the h-BN CVD system due to its nature of high-temperature sublimation. However, an explosion occurred in the autoclave, easily at such a high temperature in experiments, and it could not be controlled precisely either. MgCl2-assisted NH4BF4 CVD was subsequently carried out to successfully obtain a large-scale h-BN phase,[15] indicating the feasibility of using NH4BF4 in the single-source CVD reaction. As elaborated in that investigation, the key to h-BN formation in an NH4BF4 single-source CVD system is to stabilize the by-product HF generated from the NH4BF4 decomposition. Due to the chemical-cycled equilibrium, cycle reaction of NH4BF4 occurs in the high-temperature phase (NH4BF4 → HF + NH3BF3; 4NH3BF3 → BN + 3NH4BF4; 4HF + BN → NH4BF4), revealing that h-BN formation only happens by effectively depressing the HF content in the reaction zone. The HF can be efficiently limited by forming a stable MgF2 phase (via a reaction of HF with MgCl2 vapor), resulting in the chemical equilibrium towards h-BN direction and forming h-BN in large-scale. Unfortunately, other chemicals that could play a similar role as MgCl2 have not yet been found or reported for the NH4BF4 CVD system.
In this study, mesoporous SiO2 (SBA-15) is utilized to effectively stabilize and transfer the HF in the single-source NH4BF4 CVD reaction system. As a result, a hollow-capsular h-BN structure is successfully obtained. The formation mechanism is proposed for the first time.
To obtain SBA-15, the mixture of tetraethoxysilane (TEOS) and P123 (EO20PO70EO20, Sigma-Aldrich) was stirred at room temperature in a dilute hydrochloric acid solution (pH < 1) for 20 h. The solid product was collected, washed, and air-dried at room temperature. Then the obtained product was calcined at 500 °C for 6 h.[16]
The designed CVD reaction based on NH4BF4 in the presence of SBA-15 was carried out in a conventional furnace equipped with a horizontal alumina-ceramic tube (Fig.
X-ray powder diffraction (XRD) was recorded by a Japan SmartLab Rigaku XRD diffractometer with Cu K radiation (0.154181 nm). The scanning electron microscopy (SEM) and transmission electron microscopy (TEM) images were taken with Japan Hitachi S-4800 field-emission SEM (FESEM) (3 kV, resolution ∼ 10 nm) and USA TECNAI20 TEM instruments (200 kV, resolution ∼ 0.2 nm), respectively. The Fourier transform infrared spectra (FTIR) were measured by using a Germany Bruker OPUS 80V FTIR spectrometer. The Brunauer–Emmett–Teller (BET) parameters and nitrogen–adsorption-desorption isotherms were obtained from a USA Quanta chrome-Instruments 1900 automated-gas-sorption analyzer (77 K).
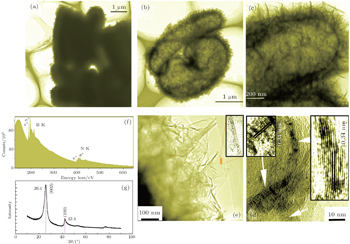
After the NH4BF4 CVD process (at 1050 °C), the as-collected residues exhibit the collapsed surfaces that differed from the smooth ones of raw SBA-15, as shown in Figs.
Further TEM measurements indicate that the washed samples completely convert to hollow capsular microstructures from the pristine solid micro-capsules (unwashed) (Figs.
In order to obtain the optimum temperature, we did a lot of work. From the XRD patterns of different-temperature samples, we know that the optimum temperature should be fixed at 1050 °C in our experiments, in light of a completely-washed-removal SiO2 treatment for the relatively pure h-BN (Fig.
In Fig.
Figure
The formation mechanism is proposed and demonstrated in Fig.

The formed NH3·BF3 vapor not only has a high thermal stability, but also works well as a CVD precursor for h-BN formation[15]
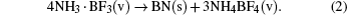
However, the decomposed HF by-product can strongly corrode the pre-formed BN, especially at a relatively high temperature (the as-formed BN can be completely consumed by HF), again generating NH4BF4

Therefore, the chemical vapor balance by the cycle reactions (Eqs. (

In order to verify the SiF4 generation in the SBA-15-assisted NH4BF4 CVD reaction system, we added a water-cooling-trap system (with a plastic bottle) in the exhaust gas before the NaOH-solution-cooling-trap system. When the reaction system cooled to room temperature naturally, the water solution of the plastic bottle was dried to obtain a small quantity of white powder. We tested the EDS of this powder, and found that the Si element emerged (Fig.
The as-introduced SBA-15 can disequilibrate the NH4BF4-cycle reactions by effectively stabilizing and transferring the formed HF in the cycle process. It is predicted that when the temperature is fixed at 1050 °C, the decomposed HF seriously erodes the formed h-BN surface layers, resulting in more collapsed breakages on the surface while h-BN growth occurs along the fracture edges by a CVD process of NH3·BF3 [proposed as (4NH3· BF3(v) + 3SiO2(s) → BN(s) + 3SiF4(v) + 6H2O(v)], leading to a disordered surface with a dense few-layered h-BN structure. Actually, SBA-15 has acted as an in-situ template for the h-BN deposition. Although SiF4 could react with H2O to generate SiO2 again [SiF4(v) + 2H2O(v) → 4HF(v) + SiO2(s)], the constant Ar flow could effectively remove SiF4 and H2O from the depositing reaction region, which does not influence the h-BN formation on the SBA-15 surfaces. Here, the F− can be transferred by forming an intermediate SiF4 in the CVD system, benefiting for the h-BN formation.
We successfully synthesized a hollow h-BN capsule structure by using the novel SBA-15-assisted NH4BF4 CVD system. The hollow capsule shells were composed of relatively well-aligned h-BN few-layers, and the collapsed surfaces were made up of disordered ultrafine h-BN nanosheets (one-layered and few-layered). The effective F− stabilization and transference of the HF are vital for h-BN deposition and growth in the NH4BF4 CVD reaction.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 |