† Corresponding author. E-mail:
Project supported by the Joint Fund of the National Natural Science Foundation of China and Chinese Academy of Sciences (Grant No. U1332104).
The structural stability of Zn2GeO4 was investigated by in-situ synchrotron radiation angle dispersive x-ray diffraction. The pressure-induced amorphization is observed up to 10 GPa at room temperature. The high-pressure and high-temperature sintering experiments and the Raman spectrum measurement firstly were performed to suggest that the amorphization is caused by insufficient thermal energy and tilting Zn–O–Ge and Ge–O–Ge bond angles with increasing pressure, respectively. The calculated bulk modulus of Zn2GeO4 is 117.8 GPa from the pressure-volume data. In general, insights into the mechanical behavior and structure evolution of Zn2GeO4 will shed light on the micro-mechanism of the materials variation under high pressure and high temperature.
Zn2GeO4 (ZGO) is an important ternary oxide. Recently, ZGO nanomaterial as a kind of nano-luminescence material has become the focus of research, since it has many miraculous new optical properties. The ZGO material possesses fluorescence and wide bandgap properties which are applied to current thin-film photoluminescence devices and field-emission displays, and it is about 40% brighter than the traditional ZnO phosphor.[1–3] In addition, the ZGO can be used as a promising high-capacity anode for lithium batteries due to the excellent rate capability,[4] and as a stable photocatalyst to decompose water and organic pollutant.[5,6] On the other hand, the ZGO was studied as the analog of Mg2SiO4 which is a dominant mineral in the upper mantle initially.[7] Because the Zn2+ with the unfulfilled 3d orbitals displays similarities to Mg2+ and the pressure of Mg2SiO4 phase transition is relatively high, it can be reduced by Ge4+ instead of Si4+, Zn2+ instead of Mg2+. Studying the influence of pressure and temperature on the structural stability and possible phase transition will provide us an insight into the potential applications to extreme conditions.
In this article, we investigate the hexagonal phenacite structure (willemite) of ZGO up to 24 GPa by means of the in-situ x-ray diffraction, high-pressure Raman spectroscopy, and high-pressure and high-temperature (HP-HT) sintering at a cubic press. The pressure-induced amorphization (PIA) is observed at ambient temperature, while the phase transformations from hexagonal phenacite to tetragonal spinel at 1200–1400 °C, 5 GPa, to decomposition above 1500 °C are found. The results are different from some early reports. The ambient-pressure phase of ZGO transformed to cubic spinel and tetragonal spinel at pressures of 3–7.2 GPa,[8] decomposed different minerals at 25–27 GPa, 1000–1400 °C.[12] The composition, pressure, and temperature dependence of the structure transformation are important tools for studying the properties of materials. In general, pressure and temperature can result in disorder in lattice, so we infer that the kinetic hindrance may be one of the mechanisms of PIA. Furthermore, the high-pressure Raman spectrum measurement is firstly performed to illustrate the route of PIA, and the instability of ZGO mainly comes from tilting Zn–O–Ge and Ge–O–Ge bond angles with increasing pressure.
The study aims to better understand the mechanism of PIA, synthesize, and determine the crystal structures of ZGO at HP-HT. There are a few works on the pressure and temperature effect on the properties of materials by HP-HT sintering at a cubic press. The approach particularly suits to obtain and observe the stability of cubic and tetragonal spinel,[8] or investigate these structures of decomposition products from hexagonal phenacite ZGO.[12] Besides, amorphous ZGO possesses better characteristics as anode materials for Li-ion batteries[9] and we can obtain different structures and amorphous bulk solids of ZGO by changing the pressure and temperature at the cubic press.
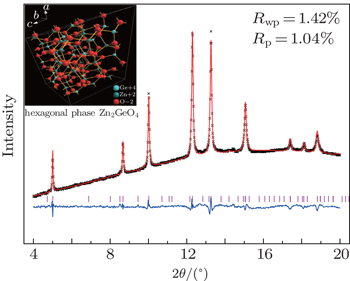
ZGO was synthesized by solid-state reactions using the starting materials GeO2 (99.9999%) and ZnO (99.9%) in a stoichiometric molar ratio: ZnO:GeO2 = 2:1. The mixed powder was heated to 1200 °C for 12 h in a muffle furnace with a heating rate of 10 °C/min. The annealed sample was confirmed to be the pure hexagonal structure by a powder x-ray diffraction with Cu-Kα radiation at ambient condition, which agrees with the refined result of 1.3 GPa diffraction data shown in Fig.
The in-situ high-pressure angle-dispersive x-ray diffraction (ADXRD) measurement was performed at the 4W2 beamline of the Beijing Synchrotron Radiation Facility (BSRF) with a wavelength of 0.6199 Å. The high pressure was generated in a Mao-Bell type diamond anvil cell (DAC) with a pair of 300 μm culet sized diamond anvils. Before the experiment, the ZGO powders and a few ruby grains with a diameter of about 2 μm were loaded into an approximately 100 μm diameter hole drilled in a stainless-steel (T301) gasket and a 4:1 methanol–ethanol was used as a pressure-transmitting medium to decrease the uniaxial stress effect. Then, the gasket was placed on the diamond anvil and compressed to the target pressure at room temperature. Pressure was determined from the Ruby fluorescence shifts and the error is not over 1 GPa.[10] The incident x-ray beam was produced from a Si (111) monochromator and was focused down to 21 μm (vertical) × 8 μm (horizontal) full width at half maximum (FWHM) by a pair of Kirkpatrick–Baez mirrors. The diffraction patterns at various pressures were collected with a Mar345 image plate. The geometrical parameters of the detector were calibrated with a high purity CeO2 powder. An FIT2D software was used to integrate and correct the collected 2D images into one-dimensional profiles. Finally, the diffraction patterns were indexed and refined with the GSAS program packages.
The Raman spectrum measurement was also performed at different pressures using a solid-state laser with a wavelength of 532 nm (RGB laser system, Germany). Prior to the experiment, the Raman system was calibrated using the peak at 520 cm−1 of monocrystalline silicon and the uncertainty is 0.5 cm−1. The high-pressure device was the same as ADXRD’s. The Raman laser beam was focused within ∼5 μm on the surface of the sample, and its power was 50 mW and the exposure time was 10 s.
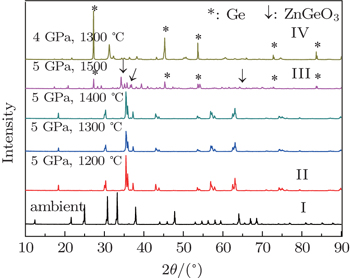
The HP-HT sintering experiments were performed on a 6 × 14 MN cubic press. During the experiment, the samples were firstly compressed to a target pressure (4–5 GPa) at room temperature, then heated to the temperature (900–1500 °C) for 15 min. Finally, these sintered samples ground to powder were identified and characterized by powder x-ray diffraction at room temperature.
The in-situ x-ray diffraction data of ZGO is shown in Fig.
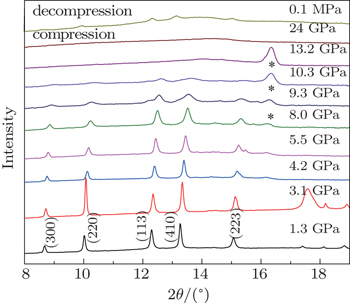 | Fig. 2. Representative ADXD patterns of ZGO under various pressures. The peaks labeled with an asterisks are from (T301) gasket. |
The datum of HP-HT sintering experiments are shown in Fig.
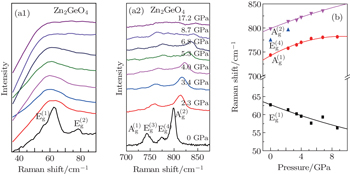
The PIA has been extensively studied in many materials.[15,17,19,21] Concurrently, the mechanism of PIA has been debated through the ages. A few factors such as framework structures, insufficient thermal energy, non-hydrostatic condition, increase in the coordination number and crystalline anisotropy have been considered to underlie PIA.[13,14,16,22] It is generally believed that the PIA easily arises from insufficient thermal energy in such a poly-tetrahedral packing structure.[16] PIA in the material of ZGO also contributed to the GeO4 and ZnO4 tetrahedron framework structure.
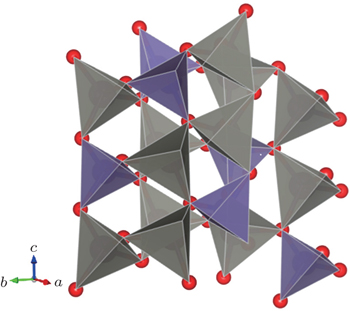 | Fig. 4. The crystal structure of hexagonal ZGO. The dark gray tetrahedrons represent four coordinate Zn atoms while the blue tetrahedrons represent Ge atoms. |
Figure
The speculation is also confirmed by analyzing the P–V/V0 (Fig.
The pressure effect on the framework structure of ZGO brings about some disordered clusters due to Zn–O–Ge linkage bonds rupture, such as tetrahedron, hexahedron or octahedron unit cell. However, there is insufficient thermal energy to form a bond between two polyhedrons. In other words, the procedure of clusters nucleation and growth is hindered so that high-pressure equilibrium phase cannot be formed, maintaining the amorphous state. Here, the phase transformation can be performed at HP-HT. This is because these clusters are arranged in order by enough thermal energy under high pressure, so that the grown clusters probably long range orderly transform to cubic spinel and tetragonal spinel structure. In other words, high temperature can supplement the dynamic to rearrange these polyhedrons to form the phases. At low temperature and high pressures, the transformation to an amorphous phase is observed, which is a general phenomenon of the open-packed structure.[11,16]
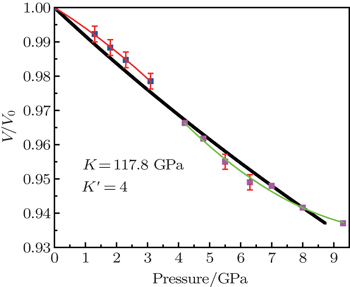
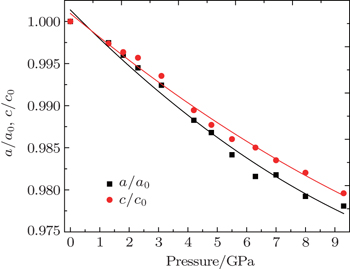
The compression behavior of ZGO under high pressure is reflected in the pressure–volume data plotted in Fig.
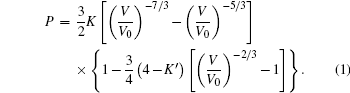
Here, V/V0 is the ratio of unit-cell volume compressed to 10 GPa comparing to ambient conditions. K is the bulk modulus, and K′ is the pressure derivative of K. The bulk modulus of material can be calculated by fixing the zero-pressure volume (V0) to its measured value. The EOS of the hexagonal ZGO is determined: V0 = 1671.4 Å, K = 117.8 GPa, K′ = 4, standard error of the bulk modulus is 11.7 GPa compared to standard EOS. The bulk modulus is an important parameter, which can present the material incompressibility. The pressure evolution of lattice parameters is shown in Fig.
The phase transition and compressibility of ZGO were investigated at pressure up to 24 GPa with room-temperature ADXRD and Raman. The HP-HT experiments were performed at cubic press to 5 GPa, 1500 °C. Pressure-induced irreversible amorphization of ZGO arises up to 10 GPa at room temperature, the hexagonal structure transforms to phase II–IV with increasing temperature, indicating that the PIA is connected with kinetic hindrance. Furthermore, the high-pressure Raman spectrum measurement illustrates that the instability of the framework structure mainly comes from tilting Zn–O–Ge and Ge–O–Ge bond angles. The compression curve of ZGO yields an averaged bulk modulus of 117.8±11.7 GPa. The material with these low frequency modes and highly flexible framework structure easily exhibits the PIA property. The study provides an effective method to make an amorphous substance using high pressure. Moreover, we can obtain different structure bulk solids of ZGO by changing the pressure and temperature at the cubic press.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 |