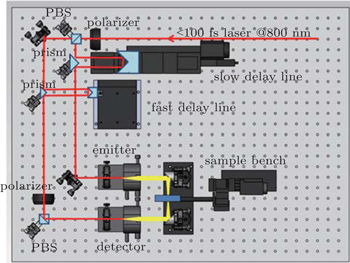
† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant No. 61205101), the Shenzhen Municipal Research Foundation, China (Grant Nos. GJHZ201404171134305 and JCYJ20140417113130693), and the Marie Curie Actions-International Research Staff Exchange Scheme (IRSES) (Grant No. FP7 PIRSES-2013-612267).
Spectroscopic measurements and terahertz imaging of the cornea are carried out by using a rapid scanning terahertz time domain spectroscopy (THz-TDS) system. A voice coil motor stage based optical delay line (VCM-ODL) is developed to provide a rather simple and robust structure with both the high scanning speed and the large delay length. The developed system is used for THz spectroscopic measurements and imaging of the corneal tissue with different amounts of water content, and the measurement results show the consistence with the reported results, in which the measurement time using VCM-ODL is a factor of 360 shorter than the traditional motorized optical delay line (MDL). With reducing the water content a monotonic decrease of the complex permittivity of the cornea is observed. The two-term Debye relaxation model is employed to explain our experimental results, revealing that the fast relaxation time of a dehydrated cornea is much larger than that of a hydrated cornea and its dielectric behavior can be affected by the presence of the biological macromolecules. These results demonstrate that our THz spectrometer may be a promising candidate for tissue hydration sensing and practical application of THz technology.
Bound between microwaves and the infrared, the terahertz (THz) waves bridge the gap between electronics and optics and hold a wealth of intriguing and highly complex THz-matter interactions in physical, chemical and biological systems.[1] Recent research in THz medical imaging suggested that THz systems may be ideal for mapping the distribution and movement of water in physiologic tissues, especially in corneal tissues.[2,3] Over the last 3 decades, terahertz time-domain spectroscopy (THz-TDS) has emerged as a promising technique for numerous prospective studies, particularly for biomedical studies[4–10] as it gives direct access to the complex dielectric response of the bio-samples. Recent reports of the unique THz maps for the in vivo sensing of the corneal tissue[2,3] warrant further investigation into the THz properties of the cornea. To date, no experimental work on the dielectric properties of the cornea has been reported in the THz region, to our knowledge.[11,12]
It is important to know the THz dielectric properties of corneal tissue for understanding the interaction between THz radiation and cornea, from both fundamental and practical point of view. The drawback of conventional THz-TDS systems is the use of the motorized optical delay line (MDL) to realize the time delay between the THz pulse and the optical detection pulse. In particular, the traditional mechanical stage for time-delay scanning remains the bottleneck due to its reciprocating motion. It is difficult to introduce such a THz-TDS system into a high-speed performance. Hence, different high-speed systems have been proposed. For example, high-speed THz-TDS system based on asynchronous optical sampling (AOS) method was proposed as a promising method without any mechanical delay.[13–15] However, it is limited by the high cost of using two femtosecond (fs) lasers, the complex of phase lock system, and the dependence of temporal scan window on laser repetition rate. Various types of fast THz pulse measurements have been proposed, such as single-shot THz measurement, oscillating optical delay line, rotary optical delay line, rotation helicoid mirrors, circular involute stages, fiber stretcher, etc.[16–21] Hence, the simplest way to reduce time-delay scanning is to use a rapid scanning mechanical delay stage. The conventional MDL has a rather simple and robust structure, but in practice, it is not easy to provide a large delay range while the scanning speed is keeping sufficiently high. There is an inherent trade-off between the range and the speed. Voice coil motors (VCMs) are direct drive actuators in which a permanent magnet field and a coil winding are used to generate a force. The characteristics of the VCM include simple structure, high thrust density, rapid response and easy control,[22] hence the VCM may be the ideal actuator for low-cost, high-speed and long-range linear optical delay line. Since the first VCM based optical delay line (VCM-ODL) was presented for the measurement of picosecond (ps) laser pulses,[23] the first THz-TDS system with high dynamic range and fast measurement speed has been developed and commercialized in recent years.[24] However, the acquisition time of one pulse trace varies with the chosen delay range, since the swing speed of the voice coil is maintained to be constant. Consequently, the effective scanning speed of this system depends on the scan range of delay line and the dynamic range depends on the number of averaged time traces.
In this paper, we propose a rapid scanning terahertz time domain spectroscopy system based on a fast-scan optical delay line. This delay line powered by a VCM stage with high-speed and high-precision can provide a delay range over 130 ps, reaching the scanning rate of up to 50 Hz, which is independent of its scan range. Thanks to the VCM stage, the system drastically reduces the acquisition time and achieves a high dynamic range in conjunction with fast measurement speed. The developed THz-TDS is used to measure the liquid water and ex-vivo rabbit cornea in a range of 0.1 THz–0.5 THz. In addition, we also investigate the possible origins of the spectrum differences among the samples by fitting the parameters of the double Debye model to our experimental data. Moreover, a proof-of-principle study on THz imaging of the water loss in the cornea is also performed.
Figure
VCM-ODL combines with a digital, high-precision position sensor with a resolution of 0.1 μm. Compared with the system (time resolution of 1.3 fs) described in Ref. [24], our system determines the delay position with an uncertainty below 200 nm, and thus achieves a higher time resolution of 0.67 fs. Meanwhile, such a delay line can provide a maximum continuous moving speed of 2 m/s with a stroke of 20 mm, corresponding to the highest scan rate of 100 Hz with the maximum delay range over 130 ps. Consequently, our system could reach the highest rate of 13000 ps/s, 10 times higher than that of 1200 ps/s (reported in Ref. [24]). Moreover, the scan rate of this delay line can vary with the chosen speed of the VCM stage, which does not depend on the delay range.
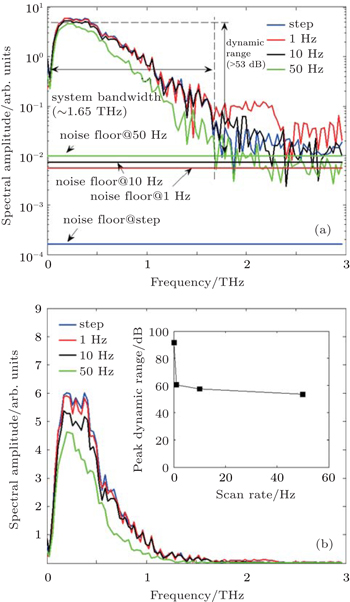
In order to examine the performance of our system, we carry out several independent tests in terms of THz spectral amplitude at different scan rates under the same measurement environment. Figure
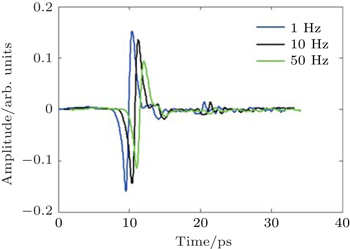 | Fig. 2. Typical THz signal measured with our system with using the fast delay line at 1 Hz (blue curve), 10 Hz (black curve shifted +1 ps), and 50 Hz (green curve shifted +2 ps), respectively. |
Figure
Because THz radiation is strongly absorbed in water, it is ideal for determining the water content in corneal tissue. In this study, we measure THz spectrum at 1 Hz mode as the overlapping spectra with step-scan mode below 1.65 THz indicate the promising reliability in 1 Hz mode for rapid scanning THz-TDS. We perform the THz spectroscopic measurements of the cornea with different amounts of water content caused by different dehydration times.[26] The transmission spectra in a range of 0.1 THz–0.5 THz have been monitored to determine the refractive indexes and absorption coefficients of the water and the cornea. Figure
 | Fig. 4. Optical properties, refractive indexes, and absorption coefficients for distilled water (black solid line) and hydrated cornea (blue dashed line) versus THz frequency. |
For describing the picosecond relaxation process in a material, e.g., in the water and corneal tissue, it is convenient to express the THz response in relative complex permittivity 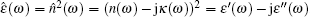
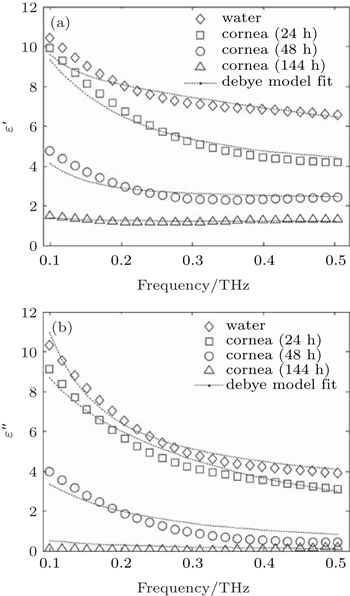
Figure
The Debye theory describes the reorientation of molecules which could involve translational and rotational diffusion, hydrogen bond arrangement and structural rearrangement. The dielectric properties in the low THz frequency range of water have been characterized and well described by the double Debye model with a slow and a fast relaxation process.[30] Moreover, the Debye theory has been also used to understand the interaction of THz radiation with biological tissues.[28,31] To better understand the change in dielectric properties of the cornea with the dehydration, we employ the double Debye dielectric relaxation model given by

Further, a genetic algorithm (GA) is chosen to fit the data to the Debye model by Eq. (
| Table 1. Double Debye coefficients for water and cornea. . |
In Table
Considering the data in Table
Besides THz spectroscopy, we propose a THz image extension for our THz-TDS system. It moves the sample in the THz focal plane where a THz temporal waveform is recorded for each step of the XY-scanning stage. THz waves interacting with the cornea respond to the characteristics by changing their temporal behavior. Thus, this image of a cornea consists of M × N pixels, and is at one time instance along a discretized time domain with P points. By stacking these M × N arrays over time, a three-dimensional (3D) time domain array (M × N × P) is generated. With the fast Fourier transform (FFT) of the time domain data, THz spectral image of the cornea can be reconstructed. For proof-of-principle experiments, we monitor the loss of water from the cornea by using the THz spectral amplitude imaging of the ex-vivo rabbit cornea sample. In order to keep the sample flat and to avoid the geometry variations during the experiment, the cornea was placed in the sample cell during dehydration.[26] The cornea placed in the sample cell is dehydrated in drying oven for over 144 h to remove its water and the mass of the corneal tissue decreases as water loses. During the dehydration, we carry out the THz imaging measurement (Fig.
The size of the scanned area is 21 mm × 21 mm and the step size is 0.5 mm. The THz spectrum for each pixel could be obtained by the rapid scanning THz-TDS technique at 1 Hz mode. The data acquisition time for one pixel is about 1.7 s for neighboring pixels in the same line, resulting in a total measurement time about 53 min. Figure
According to the performance of our system, the choice of THz imaging frequency is motivated by the trade-off among the image contrast, the spatial resolution and signal quality, which is observed to be well balanced at 0.3 THz. Figure
It is apparent that THz radiation is sensitive to variations of hydration bonding and water content, hence THz spectroscopy and imaging can be an excellent non-contact probe of water content in corneal tissue. Future research will attempt: i) to further quantify the relationship between THz properties (such as dielectric functions) and corneal hydration, in particular, corneal disease-related hydration; ii) to extend the frequency to higher region in order to ensure a precise determination of the fast relaxation time and better understanding of the mechanisms behind the relaxation processes; and iii) to develop a compact mobile and more cost-effective THz system in reflection geometry. It is expected that THz technique will become the clinically-viable technology to non-invasively assess the hydration changes of ocular diseases in vivo.
In this work, a rapid scanning terahertz time domain spectroscopy system is successfully developed by using a fast-scan optical delay line based on the VCM stage. Measurements at the scanning rate in value up to 50 Hz demonstrate the capability of the VCM as the actuator for a fast delay line. Compared with by using MDL, the measurement time is shortened by a factor of 360 by using the ODL-VCM, which holds great potential as a cost-effective linear optical delay line in a rapid scanning THz-TDS system. THz spectroscopy is applied to liquid water with the great agreement with the reported results. Further, the real and imaginary parts of the dielectric constants of the corneas with different amounts of water content in the lower THz region (0.1 THz to 0.5 THz) are measured, revealing that dehydrating the sample can reduce the dielectric coefficients of the samples. The two-term Debye relaxation model is used to describe the experimental results of the dielectric functions satisfactorily. Both two relaxation times for the corneal tissue are much longer than those of water sample, indicating that the biomolecules may have effects on the structure and properties of the cornea sample. Our results also demonstrate that the developed THz-TDS system can be an excellent tool for investigating the hydration in biological sample. Moreover, THz spectral imaging technique is successfully used to monitor the water loss of an ex-vivo cornea. The proposed and developed THz-TDS system demonstrate the advantages as a biomedical tool for non-contact sensing of tissue hydration, especially for cornea diagnostic in the field of ophthalmology.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 | |
| 34 | |
| 35 | |
| 36 | |
| 37 |