† Corresponding author. E-mail:
Project supported by the National Natural Science Foundation of China (Grant Nos. 51461033, 51571126, 51541105, and 11547032), the Natural Science Foundation of Inner Mongolia, China (Grant No. 2013MS0110), and the Inner Mongolia University of Science and Technology Innovation Fund, China.
Resource-saving (PrNdCe)2Fe14B sintered magnets with nominal composition (PrNd)15−xCexFe77B8 (x = 0−10) were prepared using a dual alloy method by mixing (PrNd)5Ce10Fe77B8 with (PrNd)15Fe77B8 powders. For Ce atomic percent of 1% and 2%, coercivity decreases dramatically. With further increase of Ce atomic percent, the coercivity increases, peaks at 6.38 kOe in (PrNd)11Ce4Fe77B8, and then declines gradually. The abnormal dependence of coercivity is likely related to the inhomogeneity of rare earth chemical composition in the intergranular phase, where PrNd concentration is strongly dependent on the additive amount of (PrNd)5Ce10Fe77B8 powders. In addition, for Ce atomic percent of 8%, 7%, and 6% the coercivity is higher than that of magnets prepared by the conventional method, which shows the advantage of the dual alloy method in preparing high abundant rare earth magnets.
(PrNd)2Fe14B permanent magnets are a high energy product and are widely used in numerous devices, promoting the technology development in the areas of computers, motors, miniature speakers, and wind turbines, etc.[1–3] However, their mass production and wide application give rise to the unbalanced use of rare earth and the backlog of Ce.[4–11] Ce ion is in a strongly mixed-valent α-like state rather than the trivalent γ state, so it is nonmagnetic and the magnetocrystalline anisotropy is low in Ce2Fe14B.[12,13] Furthermore, the phase diagram and phase constitution of Ce–Fe–B are different from those of Nd–Fe–B,[14,15] and the minor phases possibly lead to a large variation of magnetic properties. It is a big challenge to obtain relatively high magnetic properties in the resource-saving and low cost Ce2Fe14B magnets using the conventional sintered method.[16–19] The rare earth substitution could optimize the phase constitution and thus enhance the coercivity in (PrNdCe)2Fe14B magnets,[20–22] and the dual alloy method is a feasible way to improve the magnetic properties by mixing (NdCe)2Fe14B with Nd2Fe14B powders.[23] For reducing the cost of this type of magnet, the amount of Ce should be further increased in the resource-saving permanent magnets, therefore, it is necessary to probe how the Ce element affects the magnetic properties in (PrNdCe)2Fe14B magnets. In this paper, we investigate the magnetic properties of (PrNdCe)2Fe14B sintered magnets prepared by mixing Ce-rich powders of (PrNd)5Ce10Fe77B8 (the atomic ratio of Pr/Nd is 0.8) with (PrNd)15Fe77B8 powders. It is found that the coercivity is abnormally dependent on the Ce content, and that in high Ce content magnets prepared by the dual alloy method the coercivity is higher than that of conventional sintered magnets. It is expected that these investigations could serve as a reference for optimizing the composition designing in preparing high abundant rare earth magnets.
Two kinds of ingots with nominal compositions (PrNd)5Ce10Fe77B8 and (PrNd)15Fe77B8 were prepared, respectively, by induction melting of PrNd, Ce, Fe and FeB under the protection of high-purity argon. The 1 wt% excessive of rare earth elements was used to compensate the weight loss in the induction melting process. The ingots were broken to small pieces and then crushed to powders in a grinder. (PrNd)5Ce10Fe77B8 and (PrNd)15Fe77B8 powders were mixed in molar ratios of 0, 1/10, 1/5, 3/10, 2/5, 1/2, 3/5, 7/10, 4/5, 9/10, and 1, respectively, corresponding to the nominal compositions of (PrNd)15Fe77B8, (PrNd)14Ce1Fe77B8, (PrNd)13Ce2Fe77B8, (PrNd)12Ce3Fe77B8, (PrNd)11Ce4Fe77B8, (PrNd)10Ce5Fe77B8, (PrNd)9Ce6Fe77B8, (PrNd)8Ce7Fe77B8, (PrNd)7Ce8Fe77B8, (PrNd)6Ce9Fe77B8, and (PrNd)5Ce10Fe77B8. Ball milling was performed in petroleum ether for 45 min and the particle size were decreased to 3–5 μm. The fine mixed powders were compacted to green pellets after alignment under a 2 T pulsed magnetic field and then cold isostatic pressed at 300 MPa. The vacuum sintering was carried out at 1040–1080 °C for 2 h, and then the as-sintered magnets were annealed at 900 °C and 600 °C for 2–4 h, respectively. With the increase of Ce content, the sintering temperature was decreased correspondingly for optimizing the coercivity. Magnetic properties were measured using a NIM-200C hysteresis graph. The phase constitution, microstructure and chemical composition of the intergranular phase were observed by x-ray diffraction (XRD) spectrum, backscattered electron (BSE) of a scanning electron microscope (SEM), and an energy dispersive spectrometer (EDS), respectively.
XRD patterns show that Re2Fe14B is the main phase for these sintered magnets. Figure
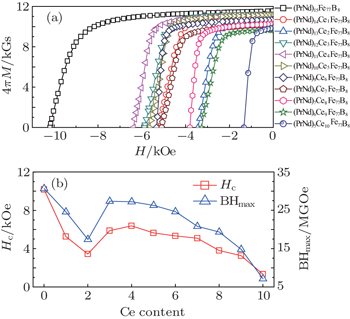 | Fig. 1. (a) The demagnetization loops for the sintered magnets prepared by the dual alloy method, and (b) the variations of coercivity Hc and energy product BHmax with the Ce atomic percent. |
Ce element substitution would result in the decrease of magnetocrystalline anisotropy and coercivity in (NdCe)2Fe14B magnets,[24] but among these sintered magnets, the coercivity is abnormally dependent on the Ce content. Similar experiments on the coercivity variation were observed in Refs. [20], [25], and [26]. Coercivity is dependent not only on the intrinsic magnetic properties, but also on the microstructure. Figure
 | Fig. 2. The surface micromorphology investigated by BSE for the (PrNd)15Fe77B8 (a), (PrNd)11Ce4Fe77B8 (b), (PrNd)9Ce6Fe77B8 (c), and (PrNd)5Ce10Fe77B8 (d), respectively. |
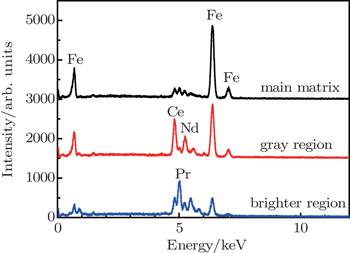 | Fig. 3. The EDS intensities of Pr, Nd, Ce, and Fe for the (PrNd)9Ce5Fe77B8 magnet in the main matrix, intergranular gray, and brighter regions, respectively. |
The nucleation of reversed domain and coercivity are related to the intergranular rare earth-rich phase and grain boundary.[29,30] In order to explore the relationship between the coercivity and the intergranular phase, the element distribution was investigated near the grain boundary by line scanning using EDS. The intergranular phase of (PrNd)15Fe77B8 magnets is rich in Pr and Nd elements (shown in Fig.
 | Fig. 4. The element distribution near the grain boundary for (PrNd)15Fe77B8 (a), (PrNd)13Ce2Fe77B8 (b), (PrNd)11Ce4Fe77B8 (c), and (PrNd)7Ce8Fe77B8 (d) magnets, respectively. |
In the (Nd1−xCex)2Fe14B (x ≤ 0.3) magnets, Ce is predicted to have limited alloying with a preference for larger R(4g) sites due to the atomic size effect, resulting in weak partial ordering.[31] This is consistent with Ce segregation near the grain boundary in this experiment for (PrNd)15−xCexFe79B6 (x ≤ 4). The high c-axis manetocrystalline anisotropy originates from the R(4g) site in the Nd2Fe14B lattice.[32] Ce element aggregation in the intergranular phase and the substitution for R(4g) sites may heavily ruin the magnetocrystalline anisotropy at the grain boundary and decrease the nucleation field,[33] which is likely to result in the dramatic drop of coercivity in (PrNd)14Ce1Fe77B and (PrNd)13Ce2Fe77B8 magnets. The further increase of Ce content leads to an increase of coercivity in the (PrNd)12Ce3Fe77B8 and (PrNd)11Ce4Fe77B8 magnets, but the underlying mechanism remains unclear. Similar results were reported in (Re1−xCex)2(FeCo)14B magnets as x is in the range of 0.20–0.30,[20,25,26] and it proposed that the anomaly was possibly related to the phase separation.[25] Magnetization reversal and coercivity are strongly dependent on the self-interaction between perfect and defect regions at the grain outer layer.[34] As shown in Fig.
For comparasion, the (PrNdCe)2Fe14B magnets were prepared by the conventional powder metallurgy method using the same sintering and annealing process. Figure
In summary, (PrNdCe)15Fe77B8 sintered magnets were prepared using the dual alloy method by mixing (PrNd)5Ce10Fe77B8 with (PrNd)15Fe77B8 powders. The magnetic properties is abnormally dependent on the Ce atomic percent, and the corecivity peak at 6.38 kOe in (PrNd)11Ce4Fe77B8 magnets. In Ce-rich magnets prepared by the dual alloy method, coercivity is higher than that of corresponding magnets prepared by the conventional method. It is expected that these investigations could serve as a reference for optimizing the composition design in preparing high abundant rare earth magnets.
| 1 | |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 | |
| 15 | |
| 16 | |
| 17 | |
| 18 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 23 | |
| 24 | |
| 25 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 31 | |
| 32 | |
| 33 | |
| 34 | |
| 35 |



