†Corresponding author. E-mail: p.iranmanesh@vru.ac.ir, p.iranmanesh@gmail.com
ZnS nanoparticles are prepared by homogeneous chemical co-precipitation method using EDTA as a stabilizer and capping agent. The structural, morphological, and optical properties of as-synthesized nanoparticles are investigated using x-ray diffraction, scanning electron microscopy, Fourier transform infrared spectroscopy, ultraviolet-visible (UV-Vis) absorption, and photoluminescence spectroscopy. The x-ray diffraction pattern exhibits a zinc-blended crystal structure at room temperature. The average particle size of the nanoparticles from the scanning electron microscopy image is about 50 nm. The ultraviolet absorption spectrum shows the blue shift in the band gap due to the quantum confinement effect. The photoluminescence spectrum of ZnS nanoparticles shows a blue visible spectrum.
Semiconductor nanocrystals, whose electronic and optical properties are tunable, have aroused considerable interest as technologically important materials. As a typical nontoxic wide band gap semiconductor material of the II– VI group, ZnS has been used as optical devices, such as ultraviolet light emitting diodes, [1– 3] flat panel displays, [4, 5] solarcells, [6] and optical sensors.[7] ZnS crystallizes in the zinc-blende (ZB) or wurtzite (WZ) structures at room conditions with a band gap of 3.68 eV or 3.77 eV, [8] respectively.
ZnS with a band gap of 3.68 eV corresponds to ultraviolet radiation for optical interband transition. Wide-band gap semiconductors such as ZnS are ideal materials for the study of discrete states in the gap. Visible luminescence can originate only from transitions involving these localized states. The reported luminescence spectrum and absorption in ZnS both show an emission peak at around 420 nm.[9– 11]
Bulk and nano-scale ZnS crystals have been successfully synthesized by various methods and their properties have been investigated.[12– 15] These general preparation techniques include the precipitation method, sol– gel, reverse-micelle method, microwave method, hydrothermal process, wet-chemical method, spray pyrolysis, etc.
At the nanoparticle sizes of a semiconductor, the energy gap (band gap) increases, and the optical spectrum is shifted toward the short-wavelength region[16] and some of their physical properties differ noticeably from those of the corresponding bulk material. These properties of nanocrystals make them an interesting category of material for optoelectronic applications.
In this work, ZnS nanoparticles are prepared using EDTA as precursor to control the particle sizes by a simple chemical route. The crystallinity and morphology are studied using x-ray powder diffraction (XRD) and a KYKY-EM3200 scanning electron microscope (SEM). The vibrational structure and the changes of the optical properties with particle size are discussed from Fourier transform infrared (FTIR) (Thermo Scientifica-NICOLET iS10) and UV-visible spectra. The photoluminescence (PL) emission properties of the ZnS sample are presented.
Nano-sized particles of ZnS crystals were prepared by co-precipitation reaction using zinc acetate (Zn(Ace)2.2H2O), sodium sulfide (Na2S), and EDTA (Merck, purity > 98%) as source materials. EDTA acted as the capping ligand. All of the reagents were analytical grade in purity and used without further purification, and the deionized water was used as a solvent. The ZnS nanoparticles were prepared in the following sequence: 0.005-mol Zn(Ace)2· 2H2O and EDTA were dissolved in 50 ml of deionized water under continuous stirring, after that the desired amount of NH4OH was used to adjust the PH of the solution to 7 to form clear liquid. Then, 0.1-mol Na2S was added to 50 ml of deionized water. The Na2S solution was then poured into Zn(Ace)2· 2H2O and EDTA solution. After the reaction was completed, a white precipitate formed. Then this mixture was centrifuged at 300 rpm for 30 min and dried at 110 ° C for 24 h in air.
Structural characterization and phase identification of nanocrystalline powder were carried out using XRD experiments with monochromatic Cu– Kα radiation (λ = 1.54056 Å ) in a 2θ range of 10° – 70° in a continuous scan mode in steps of 0.04° to determine the unit cell parameters of the sample under study. Sample morphology was also examined by SEM.
The FTIR analysis of the sample was recorded in a range from 400 cm− 1 to 4000 cm− 1. Potassium bromide (KBr) acting as non-absorbing medium was mixed with solid sample (0.3 wt%– 0.5 wt%) by an agate mortar and pestle to prepare a disk specimen. The UV radiation was performed using a PG Instrument + T80 UV-Vis spectrometer in a wavelength range of 250 nm− 500 nm to obtain the energy of band gap. The PL spectrum of the as-synthesized ZnS nanoparticles was carried out by PL spectrophotometer to investigate the luminescence properties.
The analysis of x-ray pattern (Fig. 1) shows that ZnS sample crystallize in cubic symmetry (F-43M space group) with lattice parameter a = 5.368(4) Å and unit cell volume V = 154.7157(8) Å 3. The three different peaks of the sample correspond to the lattice planes of (111), (220), and (311), which match very well with the cubic zinc blende structure (JCPDS No. 05-0566).
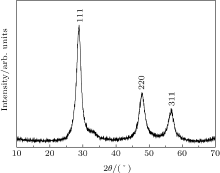 | Fig. 1. The x-ray diffraction pattern of ZnS sample, showing the corresponding miller indices of diffraction planes. |
The significant broadening of the XRD pattern peaks reveals the small particle (crystallite) size and indicates the nanometric particle size.[17] The average particle size is approximately estimated by Hall’ s method[18]
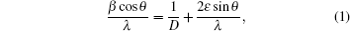
where β is the full width at half maximum (FWHM) of that peak in radian, θ is the diffraction angle of the Bragg peak, λ = 1.54056 Å is the wavelength of the used x-ray, D is the particle size, and ε is the effective residual strain. In this way, D and ε can be estimated from the intercept on the β cos θ /λ axis and the slope of the curve by plotting the β cos θ /λ versus sinθ /λ , respectively. An average particle size of about 5.5 nm is obtained.
A dislocation is a crystallographic defect that strongly affects many properties of zinc sulfide, so the dislocation density is also calculated from the following relation:[18, 19]
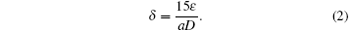
The effective strain and also the dislocation density are estimated to be 6.71 × 10− 3 and 3.41× 1015, respectively.
The SEM image of ZnS nanoparticles is shown in Fig. 2. The SEM microstructural analysis shows that the synthesized ZnS contains mainly the grains of ZnS particles (crystallite) with regular shape. One can see that nearly spherical nanoparticles have an almost homogenous size distribution with a mean size of 50 nm. In the absence of EDTA, a bulk ZnS sample is formed (not shown here). In the synthesis process, the usage of EDTA causes the stabilization of the small particles and the inhibition of this agglomeration. Due to the existence of − COOH group in EDTA molecules which absorbed on the particle surface, EDTA-capped ZnS sample is formed. The maximum particle size does not exceed 70 nm.
The FTIR spectrum of ZnS nanoparticles at room temperature is shown in Fig. 3. This spectrum shows the IR absorption due to the various vibration modes. The characteristic major peaks of ZnS can be observed at about 1124, 998, and 624 cm− 1, which are in good agreement with the reported results in Refs. [20] and [21]. The observed peaks at 1550 cm− 1– 1750 cm− 1 are assigned to the C= O stretching modes, and also the broad absorption peaks in a range of 3100 cm− 1– 3600 cm− 1 correspond to O– H stretching modes arising from the absorption of water on the surface of nanoparticles via − COOH group.[22]
The room temperature UV-Vis spectrum in the absorption mode is measured and shown in Fig. 4. As can be seen, the strongest excitonic absorption peak of the synthesized ZnS nanoparticles appears at around 303 nm. The optical band gap of the nanoparticles is calculated from the UV absorption study, which corresponds to the transition from the valance band to the conduction band, using the following equation:[16, 18]
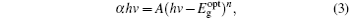
where α is the absorption coefficient, hv is the incident photon energy, A is a constant, and 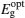
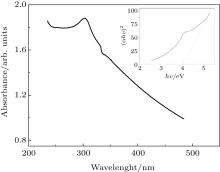 | Fig. 4. UV-Vis spectrum of ZnS nanoparticles. The inset shows the optical band gap of these nanoparticles. |
The value of the optical band gap energy is found to be 4.03 eV (the inset in Fig. 4). A comparison with the value of bulk ZnS of 337 nm (3.68 eV)[8] shows that the band edge is blue-shifted. The absorption spectrum of ZnS nanoparticle shifts towards the lower wavelength side, which can be explained as being due to the quantum size effect. Due to the quantum confinement effect of semiconductor nanoparticles in their electronic structure, which originates from the electron– hole confinement in a small volume, the increasing band gap energy occurred.
The particle size is also calculated from the absorption data using the effective mass approximation relation as follows:[16]
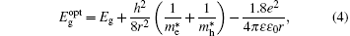
where 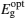
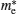
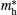
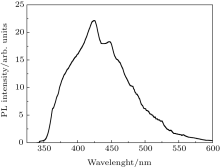
Figure 5 shows the photoluminescence spectrum of as-synthesized ZnS nanoparticles to investigate luminescence properties. Pure ZnS in the form of bulk and nano-crystals shows a main well-known emission band, which is the blue band.[23] Vacancies of sulfur and zinc are equivalent to localized donor and acceptor states, respectively. Owing to these two levels in the energy gap, two optical transitions in the observed UV range can occur. For the pure sample (Fig. 5) two emission peaks are observed at about 428 nm and 448 nm when excited by UV light. These blue fluorescence light emissions are attributed to the recombination from the shallow electron trap states (sulfur-vacancy) to the valence band and conduction band to zinc vacancy trap, respectively.
ZnS nanoparticles are successfully synthesized via the co-precipitation method. The chemical bonding and structural properties of sample are characterized by FTIR and x-ray diffraction to identify the chemical and crystal structure. The x-ray diffraction pattern indicates the very good cubic zinc-blended structure of ZnS nanocrystals. The SEM image reveals that the ZnS nanoparticles have regular shapes with an average size of 50 nm. The UV spectrum of ZnS nanoparticles shows the strong quantum confinement effect with a blue shift in the band gap energy due to the nanocrystalline ZnS particles. The PL spectrum consists of 428- and 448-nm emission bands.
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
| 10 |
|
| 11 |
|
| 12 |
|
| 13 |
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
|
| 18 |
|
| 19 |
|
| 20 |
|
| 21 |
|
| 22 |
|
| 23 |
|