†Corresponding author. E-mail: gdqian@zju.edu.cn
*Project supported by the National Natural Science Foundation of China (Grant Nos. 51272237, 51272231, and 51010002) and the China Postdoctoral Science Foundation (Grant Nos. 2012M520063, 2013T60587, and Bsh1201016).
Nitrogen-doped diamond-like carbon (DLC:N) films prepared by the filtered cathodic vacuum arc technology are functionalized with various chemical molecules including dopamine (DA), 3-Aminobenzeneboronic acid (APBA), and adenosine triphosphate (ATP), and the impacts of surface functionalities on the surface morphologies, compositions, microstructures, and cell compatibility of the DLC:N films are systematically investigated. We demonstrate that the surface groups of DLC:N have a significant effect on the surface and structural properties of the film. The activity of PC12 cells depends on the particular type of surface functional groups of DLC:N films regardless of surface roughness and wettability. Our research offers a novel way for designing functionalized carbon films as tailorable substrates for biosensors and biomedical engineering applications.
The regulation of surface and structural characteristics of materials is important for the functionalities of the materials. Carbon based materials as possible candidates for biomedical engineering applications have been studied widely in terms of their chemical inertness, adjustable mechanical properties, and biocompatibility.[1– 6] Among these, diamond-like carbon (DLC) films with high wear resistance, low friction, high hardness, excellent biocompatibility and chemical inertness, and low cytotoxicity and cellular damage show potential as coatings of implantable devices and surgical instruments including orthopedic prostheses, cardiovascular stents, heart valves, and neural implants.[6– 10] When considering the better use of DLC films as semiconductor materials, the controlled variation in electrical conductivity through doping is of primary importance.[11] The acceptable electrical conduction, wide potential window, and low back current of doped DLC-based films also confirm their superiority as microarray electrodes for signal detection in biosystems.[4, 12, 13] Since the cellular response and specific cell functions are related to the microstructure, roughness, and wettability of the materials, and sensitive to the chemical situation of the material’ s surface, [5, 6, 8, 12, 14] different elements are introduced into the carbon skeleton to modulate the structural characteristics of DLC films.[4, 7, 9, 10, 13, 15] Additionally, by immobilizing selected molecules on DLC surfaces via chemical reactions, the DLC films may be given versatile biological functionalities, [3, 6, 16] supporting the desired cell attachment.
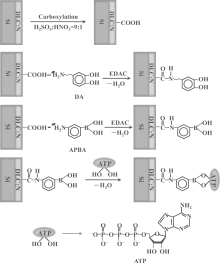
In this study, we attempt to modify nitrogen doped DLC (DLC:N) films by using different chemical molecules with diols or adjacent hydroxyls, such as dopamine (DA), 3-aminobenzeneboronic acid (APBA), and adenosine triphosphate (ATP), via a simple functionalization process (Fig. 1). The effect of surface functionalities on the surface morphologies, compositions, microstructures, and wettability of the DLC:N films is investigated by atomic force microscopy (AFM), x-ray photoelectron spectroscopy (XPS), Raman spectroscopy, and contact angle measurement. Furthermore, our immobilization method fabricates novel surfaces with controllable cell affinity. The capability of the functionalized DLC:N films as electrodes for the detection of electrical signal related to H2O2 released by the stimulated live cells is investigated at a positive potential. A possible mechanism is proposed to describe the effect of various chemical molecules on the cell response. This study may be useful for the design of biomaterials or substrate conditions used in biosensors or neural chips.
The 100-nm-thick DLC:N films were deposited on conductive n-Si (100) substrates by using a filtered cathodic vacuum arc system as described in the literature.[17] Briefly, at a vacuum chamber about 2 × 10− 4 Pa and a negative bias of 800 V, a mixed plasma of nitrogen from 12-sccm nitrogen (99.999 %) and carbon from the graphite target passed through the double-bend off-plane filter and led to the deposition of DLC:N film on the etched Si substrate. The DLC:N substrates were then carboxylated by soaking in a 9:1 mixture of sulfuric acid and nitric acid for 72 h (Fig. 1). The carboxylated DLC:N (DLC:N-COOH) samples were further functionalized by immersion in a mixed solution of 1 mg/mL DA and 1 mg/mL N-Ethyl-N’ -(3-dimethylaminopropyl)-carbodiimide hydrochloride (EDAC) for about 24 h to obtain the DA-functionalized DLC:N (DLC:N-DA). In a similar way, we obtained APBA-functionalized DLC:N (DLC:N-APBA) by immersing the DLC:N-COOH specimens in a mixture of 1 mg/mL APBA and 1 mg/mL EDAC for 24 h. The ATP-functionalized DLC:N (DLC:N-ATP) was further obtained by the reaction of adjacent hydroxyls on ATP molecules and those on APBA molecules of DLC:N-APBA surfaces while immersing the as-prepared DLC:N-APBA samples in a mixture of 1 mg/mL ATP and 1 mg/mL EDAC for 24 h. All surface modification procedures were carried out at room temperature and the obtained films were cleaned by acetone, ethanol, and deionized water respectively, and then dried overnight for use.
The surface roughness of functionalized DLC:N films was observed by AFM (CSPM 5500, Benyuan Nano, CHN) with silicon nitride tips (40 N/m) at a cantilever resonant frequency of 300 kHz. The surface topography was imaged in tapping mode with a scanning area of 4 μ m× 4 μ m. The structural characteristics of functionalized DLC:N films were acquired on a Raman microscope (DXR, Thermo Fisher Scientific, USA) with a 633-nm He– Ne laser. The XPS measurement was performed with a VG ESCALAB MkII spectrometer using an unmonochromatized Mg Kα x-ray source (1253.6 eV). The pass energies of the hemispherical analyzer were set to be 50 eV and 20 eV for the wide and the narrow scans, respectively. The peaks were calibrated against the C 1s peak at 284.6 eV. The static contact angle measurement was performed by using a contact angle goniometer (OCA20, GER) at room temperature. 1 μ L deionized water was dropped onto the functionalized DLC:N surfaces and the water droplet was photographed and analyzed via droplet shape analysis software (SCA20). Six tests were applied for each substrate.
Cell adhesion and proliferation were investigated by seeding PC12 cells (rat pheochromocytoma) at a density of 3 × 103 cells/cm2 in a 24-well plate, with each sample (8 mm× 8 mm) set at the bottom of a single well and cultured in Dulbecco’ s modified Eagle’ s medium (DMEM, Neuronbc) containing only 2.5% fetal bovine serum (FBS, Sijiqing), for 1 day, 3 days, and 7 days, respectively. The cells were then fixed with 4% (w/v) paraformaldehyde in a phosphate buffer solution (PBS) for about 15 min at room temperature and stained by 1 μ g/mL DAPI (4’ , 6-diamidino-2-phenylindole, Sigma Aldrich) for 5 min to reveal cell nuclei. The stained cells on eight regions per sample were photographed by a confocal laser scanning microscope (CLSM, fluoview FV1000, Olympus) to calculate the cell density after different proliferation times. Statistical analysis was performed using the two-way analysis of variance (ANOVA) test followed by a Tukey test (LST) to compare the statistical significance between the different groups. The significance levels were set at * p < 0.05. The electrical signal related to H2O2 oxidation was detected at 0.6 V (vs. Ag/AgCl electrode) by using an electrochemical workstation (CHI 660B, USA) with the conventional three-electrode system (DLC:N-based working electrode, Ag/AgCl reference electrode, and platinum foil counter electrode) when the live cells were maintained in the 0.02 M PBS (pH 7.4), and were stimulated to release H2O2 by phorbol 12-myristate-13-acetate (PMA, 10 μ M).
Figure 2 displays the surface morphologies of the original and the functionalized DLC:N films. All films present a homogeneous surface. The root-mean-square roughnesses of the DLC:N, DLC:N-DA, DLC:N-APBA, and DLC:N-ATP are 0.4 nm, 0.8 nm, 0.8 nm, and 0.7 nm, respectively. No significant change in surface topography occurs after chemical functionalization.
 | Fig. 2. Surface morphologies of differently functionalized DLC:N films: (a) DLC:N, (b) DLC:N-DA, (c) DLC:N-APBA, (d) DLC:N-ATP. |
To further evaluate the surface chemistry of the different DLC:N films, XPS and Raman measurements were performed after they had been functionalized. Figure 3(a) shows a typical XPS overview of differently functionalized DLC:N films. The presence of boron in the DLC:N-APBA film is confirmed by the peak located at 191.1± 0.2 eV, indicating the successful modification of APBA on the DLC:N surface. The two peaks located at 132.4± 0.2 eV and 189.5± 0.2 eV are attributed to the P 2p and P 2s spectra, respectively, due to the further functionalization of the DLC:N-APBA film with ATP molecules. The peaks centered at 284.6± 0.2 eV, 399.2± 0.2 eV, and 532.7± 0.2 eV are caused by photoelectrons excited from the C 1s, N 1s, and O 1s spectra, respectively. The four peaks at 284.6 ± 0.1 eV, 285.7 ± 0.1 eV, 286.7 ± 0.1 eV, and 288.0 ± 0.1 eV for C 1s spectra correspond to the C– C, C– N, C– O, and C= O (or O– C= O) bands, respectively (Fig. 3(b)).[18] A slight enhancement related to the C– N signal in the C 1s spectra and the N– H one in the N 1s spectra, [19] and the increase in nitrogen content for the DLC:N-DA and DLC:N-APBA samples (Table 1) evidence the surface activation of carboxylated DLC:N by the DA and APBA molecules via the EDC/NHS reaction to form the O= C– N(H) bond. The immobilization of C– O and C= O (or O– C= O) bands on functionalized DLC:N films results in a significant increase in the O 1s signal, as reflected by the increase in the ratio of oxygen to carbon (Table 1). The signals of C– B– O and B– O bands in the B 1s spectrum[20] and the P– O one in the P 2p spectrum[21] result from the APBA and ATP groups on the DLC:N-APBA and DLC:N-ATP samples, respectively. Note that the binding energy of P 2s approaches that of B 1s, so the contents of phosphorus and boron in the DLC:N-ATP film cannot be calculated accurately.
| Table 1. Atomic compositions obtained from XPS measurements for differently functionalized DLC:N films. |
The Raman results shown in Fig. 4 also confirm the successful functionalization of DLC:N films by the DA, APBA, and ATP molecules. The peak near 950 cm− 1 is related to the second-order spectrum of the silicon substrate (PSi), and the asymmetric broad peak between 1050 cm− 1 and 1800 cm− 1 is ascribed to the first-order peak of carbon (PC).[22] It is usually believed that a higher intensity of PC and a larger ratio of integral area of PC to PSi (AC/ASi) demonstrate a higher sp2-hybridized carbon film. Our calculated results show an increase in AC/ASi from 5.3 for DLC:N film to 5.7– 6.2 for functionalized DLC:N films, indicating abundant sp2-C surfaces due to the existent carbon rings from DA, APBA, and APBA-ATP molecules after chemical modification. For quantitative analysis, the asymmetric PC is fitted with two Gaussian lines, namely, D peak centered at 1370± 5 cm− 1 and G peak centered at 1545± 10 cm− 1. The G peak, which originates from zone center phonons of E2g symmetry for single crystal graphite, reflects the sp2-bonded sites in both chains and rings, while the D peak, related to K-point phonons of A1g symmetry for disordered graphite, suggests the sp2 sites in aromatic rings. Our fitting results indicate that an increase of the intensity ratio of D and G peaks, ID/IG, from 0.67 to 0.73 for functionalized DLC:N films, represents the evolution of the aromatic sp2 configuration on the film surface after chemical modification.[23]
 | Fig. 4. Raman spectra of differently functionalized DLC:N films. Broad lines are the measurements; dashed lines are fitted D and G peaks. |
The reduced surface contact angles of functionalized DLC:N films further evidence the modified DLC:N films covered by polar hydrophilic groups (Fig. 5). The adjacent hydroxyls on the DLC:N-DA and DLC:N-APBA surfaces contribute greatly to the surface hydrophilicity. The slightly higher contact angle of DLC:N-ATP compared to the DLC:N-APBA sample might be related to the smaller amount of phosphate radical groups immobilized on the film surface due to the space limitation on the monolayer.
Figure 6 shows PC12 cell adhesion on different substrates after 1-day culture. The DLC:N substrate exhibits low cell attachment due to the high hydrophobicity and low polarity on the smooth surface. The incorporation of different surface groups leads to obvious distinction in cell attachment. The APBA and ATP modifications significantly promote cell adhesion to DLC:N substrates by 84% and 75%, respectively, while the DA modification reduces cell adhesion by about 10%. There is no significant difference in surface hydrophilicity and roughness among the DLC:N-DA, DLC:N-APBA, and DLC:N-ATP substrates, indicating that the modification group has an obvious influence on improving the substrate cytocompatibility regardless of the surface hydrophilicity, polarity, and topography. Cell proliferation patterns on different DLC:N substrates are found to be similar in adhesion assays when the cell growth is measured during culture for up to 3 days and 7 days, as shown in Fig. 6. We note that the cells not only prefer to adhere to DLC:N-APBA and DLC:N-ATP surfaces, but also appear to spread out and elongate more when compared to other substrates after 3-day culture (Fig. 7). The cells cultured on all coatings and stained with DAPI show normal nuclear morphologies, without obvious signs of nuclear condensation. As the modified films have almost the same topographies and wettability properties but different surface chemistry, the differences in cell behaviors (adhesion and proliferation) can be correlated to the variation of the surface chemical molecules.
The effect mechanism of different molecules on the material surface on the cell– material interaction is complicated and still under investigation. We suggest that the biochemical performance of molecules might be the crucial factor in controlling the cell behaviors and responses on the functionalized DLC:N films. Our previous study showed that APBA used as a probe molecule could be readily linked to monosaccharide or polysaccharide on cell membranes via condensation reactions without obvious toxicity to cells.[24] Therefore, the APBA molecules with adjacent hydroxyls favor the adhesion of PC12 cells on the DLC:N-APBA surface by condensation reaction between diols on APBA of the film surfaces and those on glycans of the cell membranes. The DA is a classical neurotransmitter which modulates various cell functions and stimulates cell actions.[25] Though the DA molecule has the diol structure, it can cause toxicity by its auto-oxidation to toxic DA-quinone species, which might lead to cellular damage (slow proliferation and easy apoptosis), with the formation of superoxide radicals and hydrogen peroxide at physiological pH.[26] Therefore, the cells on the DLC:N-DA substrate present reduced adhesion and proliferation compared to those on the DLC:N substrate. As the direct source of energy for cell activities, ATP has been found to strengthen cell metabolism activity, activate cell cycle progression, enable cell self-repair, and mediate neurotransmission and hormone secretion, [27] resulting in improved cell proliferation and depressed cell apoptosis. Therefore, the ATP molecules on the DLC:N-ATP surface can favor the adhesion and location of cells by the specific conjugated action between the ATP and the ATP receptor on the cell membranes.
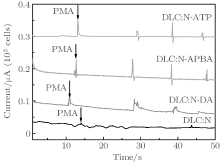
The functionalized DLC:N films are further used for real-time detection of electrical signal of H2O2 (messengers in many cellular functions) secreted by the stimulated live cells. When the PC 12 cells are cultured for 4 h and maintained in 0.02 M PBS (pH 7.4), only the background current can be detected at the positive potential of 0.6 V. After the stimulator PMA is added into the PBS, the cells are stimulated to release H2O2 molecules into the solution. A considerable current of 50 nA is observed at the DLC:N-APBA and DLC:N-ATP electrodes, followed by a gradual decrease of current (Fig. 8). Comparably, the DLC:N-DA and DLC:N electrodes detect a lower signal of H2O2 oxidation. Therefore, the cytocompatibility and electrochemical characters of the DLC:N film are adjustable effectively by the surface modification methods proposed in our research.
The nitrogen-doped diamond-like carbon (DLC:N) films were chemically modified with three chemical molecules including dopamine (DA), 3-aminobenzeneboronic acid (APBA), and adenosine triphosphate (ATP) via the carboxylic acid groups on the carboxylated film surfaces to give covalent binding sites for cell immobilization. The impact of surface functional groups on the composition, microstructure, and wettability was studied. Cell behaviors were related to the chemical molecules fixed onto the DLC:N films regardless of the surface roughness and wettability. Our results highlight the importance of surface chemical molecules on cell– material interactions and suggest mediated bioactions via specific chemical surfaces. The surface modification methods would broaden the application of DLC:N film as an important electrode material for electric devices used in the nervous system.
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
| 10 |
|
| 11 |
|
| 12 |
|
| 13 |
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
|
| 18 |
|
| 19 |
|
| 20 |
|
| 21 |
|
| 22 |
|
| 23 |
|
| 24 |
|
| 25 |
|
| 26 |
|
| 27 |
|